Dr Lisa Burden | Dr Daniel Burden – Monitoring and Controlling the Delivery of Single Molecules through Nanopores
Monitoring and controlling molecules as they are transported in and out of nanometre-sized compartments is no easy task. Dr Lisa Burden, Dr Daniel Burden and their colleagues at Wheaton College have made significant contributions to understanding these processes by developing nanoscale components inspired by cell membranes. They are also creating methods to interpret the signals that arise during transportation. These methods can be exquisitely sensitive, revealing even the tiniest signatures created by individual molecules.
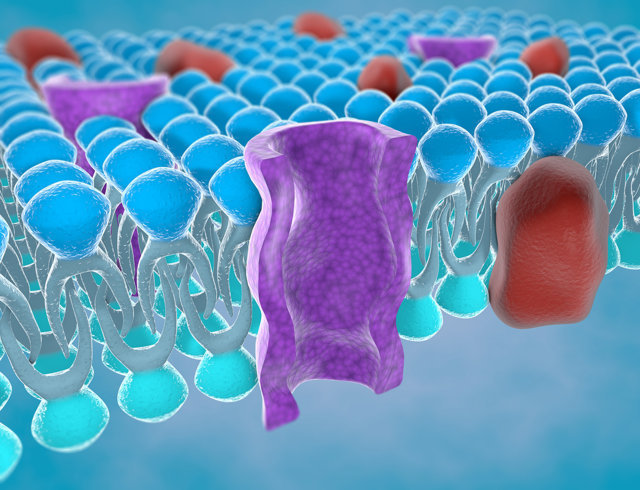
Protein ion channels embedded within cell membranes offer a route for molecules to enter and exit the cells in our bodies. The biological purpose of these channels is to deliver and remove substances essential for many central life processes, including nerve signal propagation and protein synthesis. The type of molecule and how much it interacts with the walls of these ion channels allows our bodies to monitor and control many complex and intricate chemical reactions.
The question of how to observe and control these tiny molecular events in the macroscopic realm that is accessible to us presents a persistent challenge. If scientists could have complete control over molecular transport in and out of cells, then they could design efficient chemical sensors inspired by the properties of these membrane ion channels. Sensors based on cell membranes have a wide variety of applications in different fields, such as medical diagnostics, monitoring targeted drug delivery, determining the rate of cellular drug uptake, and even measuring environmental pollutants.
In a membrane-based sensor, these channels are typically called ‘nanopores’, which run transversely through a lipid membrane. To increase the probability that a nearby small molecule will pass through a nanopore to the other side of the membrane, an external voltage is applied. Once energised, the electrostatic attraction between the molecule and cell membrane promotes rapid molecular transport through the pore.
Dr Daniel Burden, Dr Lisa Burden and their colleagues at Wheaton College are focused on designing controllable membrane-based tools for the detection and delivery of small molecules that move between fluid-filled compartments. ‘Our research attempts to harness existing cell-membrane proteins from a variety of microorganisms, understand their function, and implement strategies for chemical modification that will transform the protein from its native function to a desired chemical sensing, or transport control application,’ explains Dr Lisa Burden.
Nanopore Sensors
To develop an efficient membrane-based sensor for DNA sequencing applications, the rate of interaction between moving molecules and the nanopore walls needs to be controlled. Towards this end, DNA chains that contain the genetic information can be labelled with additional molecules, such biotin (a small vitamin), fluorescent compounds that glow, or streptavidin (a protein that binds biotin). When biotin- or streptavidin-bound DNA approaches an ion channel, applying an external voltage can determine whether the DNA strand enters this channel or is driven away into the surrounding solution. When driven into the pore, the extra size of the attached label helps slow, or even stop, DNA transport. This makes the DNA easier to detect with electricity. By adding labels that glow, researchers can capitalise on secondary optical signatures that also characterise the transportation event. Together, both electrical and optical measurements yield more information about the nature of the DNA interaction with the inner walls of the pore and can potentially unlock sequence information that is hidden within the DNA structure itself.
The Burden lab proposed a new method to simultaneously characterise the optical and electronic properties of a membrane-based sensor, taking advantage of the controllable behaviour of biotin- and streptavidin-bound DNA. Through the combined use of fluorescence microscopy and electrical current measurements, the team was able to quantify the electrical and optical signals associated with a fluorescently labelled DNA molecule and the nanopore.
When an external voltage is applied to the system, the distribution of electric charge (or polarity) on the membrane changes. This change in polarity then drives the labelled DNA strand to enter the channels. When the streptavidin-bound DNA enters a channel, the labelled molecule starts to fluoresce and the team can detect this fluorescence signal. If the DNA strand leaves the ion channel and goes back into the main solution, the fluorescence response is switched off and can no longer be detected. The correlation between the fluorescence activity and membrane polarity allows the team to calculate diffusion constants, which describe rates of molecular movement. This dual optical and electrical approach explored by Drs Lisa and Daniel Burden can also be used to study cellular processes at microsecond time resolution, thus allowing the scientists to directly observe and study the transportation of molecules into a cell.
Taking a Nanopore’s Temperature
The ‘potential energy surface’ of a molecule describes the energy difference between its stable and unstable geometries. When the energy difference is less than the available thermal energy, temperature changes can reconfigure a molecule’s shape in favour of high-energy (unstable) and more reactive geometries. To probe unstable geometries that occur quickly, these temperature changes need to take place very rapidly, with exact spatial precision, and in a way that can be simultaneously probed. Traditional solution heating and reaction monitoring technologies cannot achieve this aim.
In collaboration with investigators at the National Institute of Standards and Technology (Gaithersburg, MD), the Burden group helped develop a new method for rapidly heating a fluid, which allows a nanopore to measure the geometrical change of single molecules located near the pore opening. The new method involves locally heating a tiny volume of solution – a yoctolitre, or 10−24 litres – using a laser heat source that is focused by metallic nanoparticles. In the group’s first work, the team attached gold nanoparticles (with diameters of 40 nanometres) to the top portion of a single nanopore. By irradiating the gold particles with a laser, ‘surface plasmons’ become excited. Surface plasmons are quantum phenomena – arising due to the oscillating charges of surface electrons. Exciting them causes the local temperature and viscosity of the solution to quickly change. Single target molecules located within this tiny volume can then be thermally driven into rare high-energy shapes, which are detectable when they are subsequently transported through the nanopore.
As a function of time and laser power, the group measured the consequent change in the conductivity of the surrounding solution. By measuring the conductivity change in the solution, the team was able to infer the extent of the temperature change and detect shape changes in single polymer molecules residing in the heated zone.
Their approach to measure optically induced temperature changes near nanopores has only been demonstrated with polyethylene glycol molecules thus far. However, this method has potential to be used for detecting conformational changes in proteins that undergo unusual and rapid folding processes and other complex changes, as well as studies aimed at unravelling the DNA double helix.
Simulating Membrane-Based Sensors
Some sensing measurements proposed for nanopores are experimentally complex. Furthermore, a vast number of different compounds might be detectable using nanopores. In efforts to narrow the immense investigative scope, computer simulations offer guidance to experimentalists who seek to discover and implement new applications. Simulations are able to predict current, force and temperature changes that occur inside nanopores, and to some extent, they can also predict similar changes in the surrounding solution. This enables researchers to predict the likelihood that a given molecular target will get drawn into the pore, where it can be characterised.
The voltage applied across a nanopore provides the driving force for entry of a target molecule, in what can be referred to as a molecular-capturing event. As a molecule in solution approaches the nanopore entrance, two kinds of forces are at work. Unidirectional ‘drift producing forces’ encourage targeted molecules to flow into the pore, like driftwood captured by a storm water drainpipe. However, at the nanometre scale, random directional forces, arising from frequent collisions with the target’s molecular neighbours, become very significant. The forces arising from these impacts tend to push the target away from capture, competing against unidirectional drift through the channel. The relative size of the two opposing factors determines which type of motion dominates and also governs the likelihood of a capture event taking place. Interestingly, the relative balance of forces experienced by a target molecule depends on its distance from the nanopore and the strength of the applied voltage.
Drs Lisa and Daniel Burden have constructed a theoretical model to calculate the magnitude and direction of both the drift producing forces and the randomising forces for molecules as they approach and move through a nanopore. To do this, they combined a finite element analysis (FEA) method, in which a series of differential equations are used to calculate properties inside and outside the channel, with a Brownian motion thermal simulation. Using this approach, the team was able to plot force maps of unidirectional and randomising forces that a target molecule experiences as a function of location and applied voltage. By also looking at the erratic movement of the simulated target molecules, the team could calculate how likely a molecular capturing event would be for a given set of conditions.
Their results identified several critical limits that dictate the rates associated with molecular capture. These included the channel shape and size, the strength of the applied voltage, the salinity of the solution, and the size and charge of the molecule. The team’s simulations demonstrated that under commonly used analysis conditions, random motion can disrupt directional flow, rendering a molecular capture event less probable. Significantly, their research ruled out certain types of complex sensor configurations, where the voltage required for high-probability capture is currently outside of realistic experimental tolerances. The simulations carried out by Drs Daniel and Lisa Burden offer a valuable resource for predicting the experimental conditions required to design functional sensing devices for molecular capturing events.
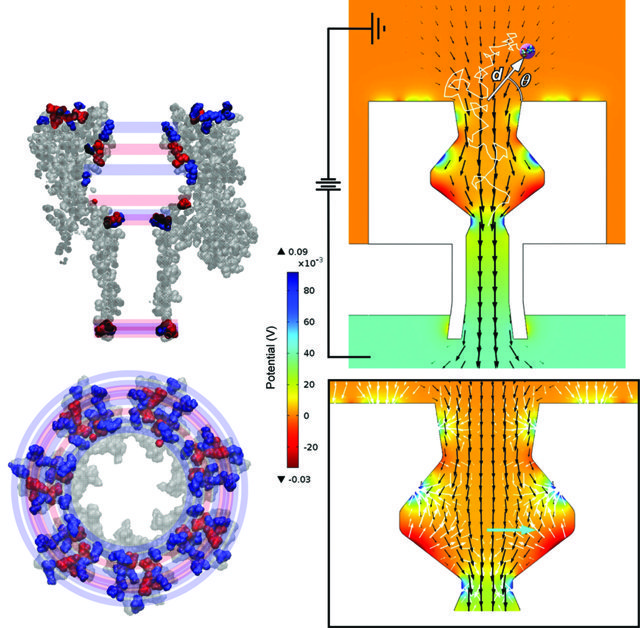
Reprinted with permission from J. Phys. Chem. B, 2015, 119 (33), 10448–10455. Copyright 2015 American Chemical Society.
Future Directions for the Burden Group
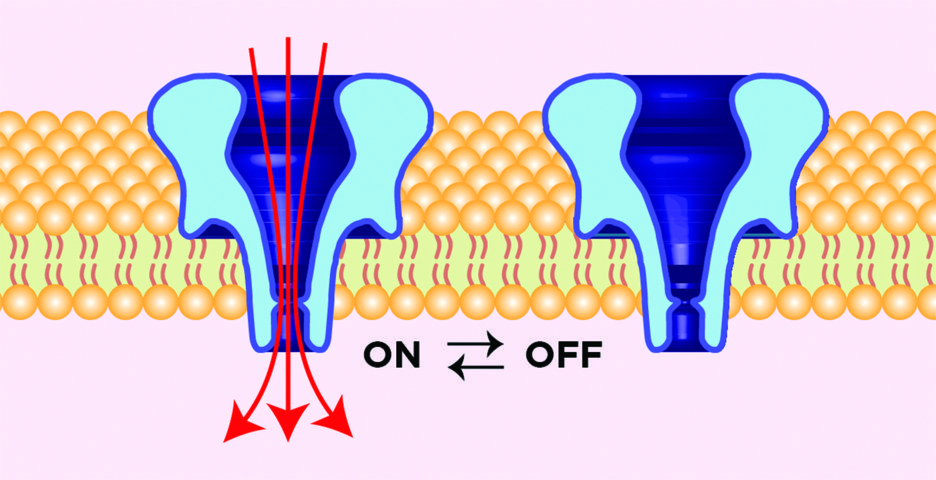
The team now intends to collate their extensive experience with membrane-based sensors and work towards designing new controllable nanovalves. Their intention is to design tiny chemical valves that can be completely switched ‘on’ and ‘off’ on command, in a completely reversible and repeatable fashion, without the need for an externally applied voltage. Currently, the best ion channel candidates for valve operation only prohibit a small percentage of fluid flow, or are not reversible. So, the team is designing strategic chemical modifications to adapt channels for this application. Dr Daniel Burden states that the outcome will enable modified channels to operate ‘much like a mechanical valve controls the flow of fluid in a pipe’. ‘Just as today’s macroscale manufacturing requires numerous types of mechanical valves to control the direction and type of fluid flow in complex machines, tomorrow’s nanoscale manufacturing will require similar components that control molecular transport between tiny compartments,’ he explains. These new nanovalves, with differing pore sizes and channel shapes, could be incorporated into small liquid-filled chambers, or even into the membranes of living cells to control what molecules enter and exit.
The Burden group’s recent nanovalve work has shown that chemically modified channels can be stimulated to control bulk aqueous flow using reversible chemical interactions. Early work is being performed in both model lipid membranes and the more complex environment of biological cells. The team’s results suggest that nanovalves embedded into cell membranes can control whether a cell lives or dies, by adding appropriate chemical triggers that open the valve to kill the cell, or close the valve to let it live. Furthermore, reversible molecular flow through the valve may be permitted over many ‘on’ and ‘off’ cycles.
This new type of nanoscale valve system also drives the need for new metrologies to gauge transport through the pore. Traditionally, researchers estimate the volume of molecular flow through its correlation with ionic current. However, this cannot account for the movement of neutrally charged compounds, chemical filtering effects induced by the channel’s interior, or the potential cooperative effects of nanovalve aggregation. Thus, the team seeks to develop new optical approaches that will allow direct monitoring of materials moving through chemically modified nanopores. The new type of data promises to uncover relationships between chemical modification, channel shape and molecular size, which can then be used for optimising future nanovalve designs.
‘The type and level of flow controlled by these nanovalves could create new horizons for many areas of science and engineering, such as enhanced chemotherapies, novel chemical sensors, new approaches to nanoscale manufacturing, and enable aqueous-based molecular electronic devices,’ says Dr Daniel Burden. If the team achieves their concept of designing controllable nanovalves, the potential contribution that this technology would make is extensive.
Meet the researchers

Dr Daniel Burden
Department of Chemistry
Wheaton College
Wheaton, IL
USA
Dr Daniel Burden graduated with a PhD in Analytical Chemistry from Indiana University in 1997. Following a postdoctoral position in biophysics at the National Institute of Standards and Technology in Gaithersburg, MD, Dr Burden undertook a position at Wheaton College, where is he is currently Professor of Analytical Chemistry. Dr Burden’s research includes methods to measure small molecules embedded into lipid membranes and observe their chemical and physical behaviours.
CONTACT
E: daniel.burden@wheaton.edu
T: (+1) 630 752 5065
W: http://burdenlab.org

Dr Lisa Burden
Department of Biology
Wheaton College
Wheaton, IL
USA
Dr Lisa Burden graduated with a PhD in Biochemistry from Indiana University in 1996. She then undertook two postdoctoral positions at Indiana University and National Institutes of Health, USA before starting her current position as Visiting Associate Professor of Biology at Wheaton College in 2000. Dr Burden’s primary research focus includes the study of toxin proteins produced by common bacteria and the chemical and biological interactions of these toxins with cellular lipid membranes.
CONTACT
E: lisa.burden@wheaton.edu
T: (+1) 630 752 7268
W: http://burdenlab.org
KEY COLLABORATORS
Andreas Geiger, former student at Wheaton College
Jared Ellingsen, former student at Wheaton College
Abigail Williams, former student at Wheaton College
Yi Yen Heng, current student at Wheaton College
Nathan Taylor, current student at Wheaton College
Emma DiBernardo, current student at Wheaton College
Dr John Kasianowicz, National Institute of Standards and Technology, Gaithersburg, MD
FUNDING
Camille and Henry Dreyfus Foundation
Research Corporation
Petroleum Research Fund
National Institute of Standards and Technology
National Science Foundation
Wheaton College Alumni Foundation
REFERENCES
ED Pederson, J Barbalas, BS Drown, MJ Culbertson, LM Burden, JJ Kasianowicz and DL Burden, Proximal Capture Dynamics for a Single Biological Nanopore Sensor, The Journal of Physical Chemistry B, 2015, 119, 10448–10455.
JE Reiner, JW Robertson, DL Burden, LK Burden, A Balijepalli, JJ Kasianowicz, Temperature sculpting in yoctoliter volumes, Journal of the American Chemical Society, 2013, 135, 3087–94.
EL Chandler, AL Smith, LM Burden, JJ Kasianowicz, DL Burden, Membrane surface dynamics of DNA-threaded nanopores revealed by simultaneous single-molecule optical and ensemble electrical recording, Langmuir, 2004, 20, 898–905.