Dr. Celia Chen – Mercury in Seafood: What the Madhatter didn’t know
A childhood spent between smokestacks and alongside beaches led Dr. Celia Chen to a long and distinguished career investigating marine ecosystem pollution.

Tell us about yourself, what brought you into this field?
I wanted to be an environmental scientist since I was very young. I grew up along the metropolitan corridor of New York City and New Jersey, where there were lots of industrial facilities spewing air and water pollution. It made a very deep impression on me. At the same time, the beaches of New Jersey gave me my love of marine ecosystems. As a result, I came to be interested in contaminants in aquatic ecosystems, particularly at the margin between freshwater and saltwater, i.e. estuaries.
How did you end up in the Toxic Metals Superfund Research Program?
In the year that I finished my Ph.D., a group of Dartmouth scientists got together to develop and submit a program proposal for the Superfund Research Program. We were awarded the program grant in 1995 and have had it ever since. The program requires a combination of biomedical and non-biomedical projects, and I became the Co-leader of a non-biomedical project on metals in aquatic ecosystems.
The program has scientists from many different backgrounds. How do you feel this affects the research you can do?
I believe that in order to solve the complex environmental problems in our world, we need to take interdisciplinary approaches, not just within the sciences but the social sciences as well. It definitely expands my range of knowledge and allows me to examine my own scientific questions in different ways. Even within my own project, I work with biogeochemists and trace metal chemists who have taught me so much about the physical/chemical environment of the estuaries we work in and the intricacies of metal speciation.
You are also the leader of the Research Translation Core. Can you tell us a bit about this role?
As the leader of the Research Translation Core, I am responsible for communicating and translating the research in our Toxic Metals Program to our stakeholders (governmental and non-governmental entities) and the public. This requires that I become a “translator of science” in fields of study outside of my own; the other projects in our program involve epidemiology, plant genetics, and molecular biology – all far from my expertise in aquatic ecology. Some of our main Research Translation projects have involved making short films for the public on arsenic in drinking water and mercury in fish. We have also brought together mercury scientists to summarize and synthesize mercury science relevant to policymakers and the recently signed Minamata Treaty. We are now doing the same for the science associated with arsenic in food.
What should society be doing to minimize methylmercury pollution?
The only thing we can do is to reduce sources, both atmospheric sources (especially coal-fired power plant and artisanal small-scale gold mining) and industrial point sources. The former requires technology for removal of mercury (Hg) from power plant emissions, and the latter involves educating the artisanal small-scale gold miners found mostly in the developing world. The Minamata Treaty, when fully ratified, will help to reduce Hg sources.
Where would you like to take your research from here?
I think there is still a lot of work to do on predicting the effects of climate change on the fate and bioavailability of Hg, particularly the interactions of environmental factors (nutrient loading, temperature, salinity, pH). I am also interested in the co-occurrence of contaminants (e.g. organic contaminants and mercury) in fish and the benefits and risks of eating fish. For the latter, I am interested in the factors that control the fatty acid composition of fish.
If you could perform one experiment, regardless of cost or difficulty, what would it be?
I would conduct a large mesocosm experiment (Ed: a mesocosm is a controlled section of the natural environment) on the interactive effects of pH, temperature, and organic carbon on the production, bioaccumulation, and trophic transfer of MeHg. It would require a larger mesocosm system than we have available and would likely need to run for the better part of a summer. These larger-scale controlled experiments are important since they capture environmental realism but allow for controlling important environmental factors as well.
And lastly, every mercury-related article needs a question involving the Mad Hatter…so why do you think a raven is like a writing desk?
Ha ha! I like the answer “Poe wrote on both of them”. The manner in which mercury enters, travels through and accumulates in the food web is of vital importance.
Mercury Rising
The Dartmouth Toxic Metals Superfund Research Program comprises an interdisciplinary group of experts investigating the effect arsenic and mercury have on the environment and human health.
 Be it mad as a hatter, suffering from the Danbury Shakes, or showing severe mercurialism, humanity has known for many years that mercury poisoning has long-reaching dangers. But mercury is no longer the concentrated poison which drove hat-makers mad, it now occurs in a more dangerous form – concentrated in aquatic food chains, and is now known to have a wider range of health effects.
Be it mad as a hatter, suffering from the Danbury Shakes, or showing severe mercurialism, humanity has known for many years that mercury poisoning has long-reaching dangers. But mercury is no longer the concentrated poison which drove hat-makers mad, it now occurs in a more dangerous form – concentrated in aquatic food chains, and is now known to have a wider range of health effects.
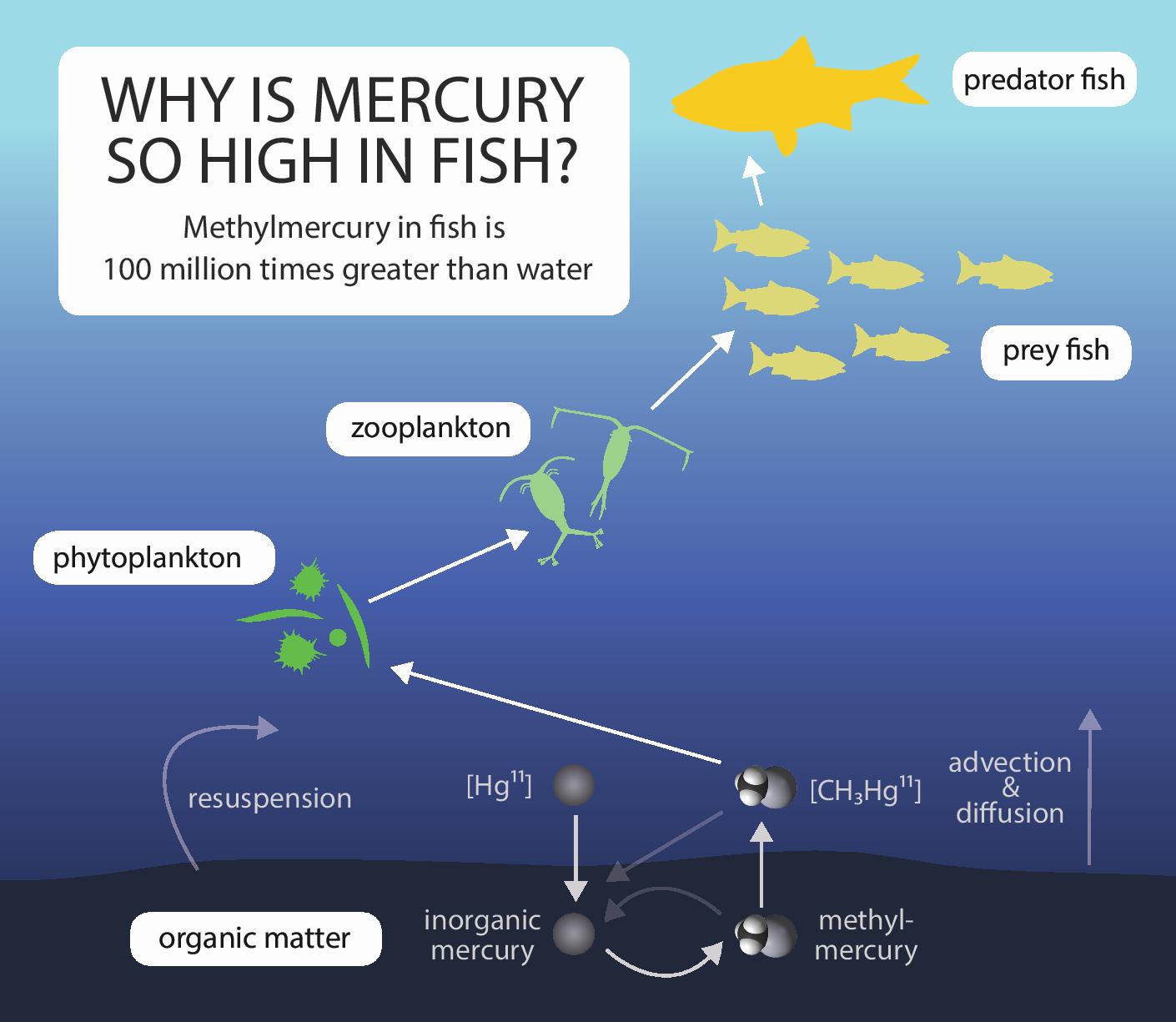
Mercury is particularly problematic in the marine food web, as the highly bioavailable methylmercury (MeHg) is readily taken up by marine organisms such as plankton in a process known as bioaccumulation. These organisms are then eaten by predatory fish, who in turn are fed upon by higher predators, each, in turn, accumulating the mercury already present in their prey. Thus, high-level predators such as tuna and swordfish will eventually end up with potentially harmful levels of mercury, a process known as biomagnification. This, in turn, means that humans who feed frequently upon these higher predators can give themselves mercury poisoning, with all of the neurological symptoms that this implies.
This is important because fisheries and farmed fish provide tens of millions of tonnes of protein to consumers worldwide, protein which in many cases cannot be replaced by other sources. In addition, seafood has important nutrients that are also not found in other foods. The accumulation of mercury in both wild and farmed species is thus a significant problem for public health, and thus, understanding the manner in which mercury enters, travels through, and accumulates in the food web is of vital importance.
Methylmercury is both toxic and easily bioavailable, and enters marine ecosystems via several sources. Estuaries sit at the interface of freshwater rivers and saltwater coastal systems, making them an intersection between land-based sources and ocean sources of pollution. Because of this central location, estuaries tend to collect mercury from contaminated sites on land, from river inflows, and from the ocean itself. Despite this knowledge, very little research has been performed on how exactly estuarine food webs and mercury sources actually link together.
So what do we know? Methylmercury, the toxic form of mercury, is formed in underwater sediments, from which it can diffuse out into the water – here it joins MeHg brought in by river inflows or ocean currents. In the water, it is taken up by microscopic plants and animals. Sediment MeHg can also be picked up during feeding by benthic infauna (e.g. worms and clams burrowing through the sediment). Transfer of MeHg from sediments into the water is particularly important, although the nature of this process in coastal and estuarine systems is not well understood. Further factors complicate this process: for example, organic carbon molecules can bind to Hg and reduce its availability to bacteria and fish, while other factors like temperature will encourage the uptake of Hg by certain organisms.
MERCURY STUDIES IN THE FIELD
To shine further light on this process, Dr. Celia Chen of Dartmouth College, and her collaborators have recently been examining the amount of MeHg present in estuaries across the northeast US coastline. Essentially, they have been measuring sources and stores of mercury within individual estuaries in the region, attempting to determine which environmental factors (temperature, organic carbon, salinity) influence MeHg production and bioaccumulation and thus provide a possible conceptual map of mercury transformation and movement.
Interestingly, they did not observe any correlation between the amount of methylmercury in sediments where most of the mercury is stored and that found in fish. However, there was a strong link between the amount of mercury in the water, be it completely dissolved or as particulate matter, and the amount found in the fish themselves – and MeHg levels were noticeably lower in fish which fed in the sediment. This all suggested that the direct source of mercury contamination in the food web was coming from the water, and that this pollution was coming not just from the sediments in the estuary.
So where was it coming from? The group performed further research to try and answer this question, showing that water methylmercury levels were not related to sediment mercury levels across a range of sites– completely surprising given the amount of mercury stored in sediments. Estuaries are intersections of multiple different mercury sources, and it seemed that a number of complex external sources were interacting to create the final polluted mixture of mercury in the water and sediments.
MERCURY STUDIES IN THE LABORATORY
One challenge facing researchers in this field is the sheer complexity of the problem – when dealing with an entire ecosystem, how do you decide which variables are the most important? One way in which complexity can be simplified is via the use of model systems, for example in the use of cultured cells to help answer questions covering an entire human patient. Part of Dr. Chen’s research involves the development of model systems for mercury bioaccumulation, in particular, the use of killifish and amphipods in the laboratory.
Killifish, specifically the gloriously named Mummichog (Fundulus heteroclitus), are an exceptionally hardy species, living happily in waters with differing salt, temperature and oxygen levels and in pristine to polluted ecosystems. Due to this, their widespread distribution, and their position towards the bottom of the food web, killifish are a good model for examining the accumulation of mercury within an ecosystem.
Dr. Chen’s research has already shown the value of these fish, with recent work having shown that higher water temperatures are linked to increased mercury accumulation in the mummichog both in the tank and in the wild. This is a concerning discovery, as the steadily increasing global temperatures are thus likely to be matched by an increase in mercury within the ecosystem – bad news for those who enjoy tuna frequently.
The group plans to use model organisms and their field research to further identify these complex sources and pathways of mercury. This is particularly important nowadays when other environmental conditions are changing with climate and the international community has recently committed to control global mercury sources through an international treaty, the Minamata Convention on Mercury. Though there is as yet no real way to extract methylmercury from the environment once it is there, their work holds out the hope of understanding these puzzling processes. And from understanding, we can predict how, when and where mercury pollution will end up in our seafood.
Researcher Profile

Celia Y. Chen, Ph.D.
Research Professor
Department of Biological Sciences
Dartmouth College
Dr. Celia Chen has spent the last 20 years unravelling the ways in which metal pollutants travel through and contaminate freshwater and marine food webs. Using both laboratory and field studies, her work on biological accumulation of mercury forms a core component of the Toxic Metals Superfund Research Program at Dartmouth College. Alongside her research she also works to communicate the program’s discoveries to both the wider community and government agencies.
CONTACT
T: +1 603 646 2376
W: http://www.dartmouth.edu/~toxmetal/ about/research-team/faculty/Celia.html
W: http://www.dartmouth.edu/~toxmetal/ index.html
W: http://www.dartmouth.edu/~toxmetal/ mercury-source-to-seafood/index.html
KEY COLLABORATORS
Dr. Brian Jackson, Department of Earth Sciences, Dartmouth College
Dr. Kate Buckman, Department of Biological Sciences, Dartmouth College
Dr. Vivien Taylor, Department of Earth Sciences, Dartmouth College
Dr. Robert Mason, Department of Marine Sciences, University of Connecticut
Dr. Nicholas Fisher, School of Marine and Atmospheric Sciences, Stony Brook University
Dr. Charles Driscoll, Department of Civil and Environmental Engineering, Syracuse University
FUNDING
Superfund Research Program of the National Institute of Environmental Health Sciences
National Institute of Environmental Health Sciences of the National Institutes of Health
New York State Energy Research and Development Administration
US Forest Service

