SFB 1027 – Cell Physics: Understanding How Biological Matter Self-Organises
The Collaborative Research Centre SFB 1027 at the Saarland University in Saarbrücken and Homburg is an interdisciplinary research team that aims to achieve a quantitative understanding of the physical mechanisms at work when biological matter self-organises into complex structures. Such self-organisation allows biological systems to perform dynamic functions including cell migration and polarisation, cell-cell adherence and synaptic transmission, biofilm formation and tissue growth.
The sum is more than its parts – this old saying also holds true on all scales of living matter: atoms form large proteins that perform as molecular motors, pumps and channels; proteins, DNA and membranes form cells that can migrate, adhere and proliferate; cells form tissues and organs; tissues and organs form organisms. In the same way that one cannot predict the different phases of water by simply looking at a single water molecule, one cannot understand the emergent functions of proteins or cells, for example, by studying their constituents in isolation.
In recent years, so-called ‘active matter’ systems, especially those found in biology, have rapidly become the focus of many research groups. In contrast to passive matter, which responds passively to external forces, pressure or temperature changes, active matter can move, pump, copy, and even proliferate. Just like a ‘smart’ material, active matter can adopt to a changing environment. This ability depends crucially on the use of energy, which is the reason why these processes are inherently out of equilibrium. Most systems found in nature are actually out of equilibrium: they are not stationary and are continuously subjected to a flux of matter and energy. Nearly all dynamic processes occurring in living cells consume energy in the form of ATP and are commonly denoted as ‘active’ processes.
Prominent examples include ATP-driven molecular motors within cells, which on one side carry cargo around along filaments of the cytoskeleton and on the other side exert forces between the cytoskeletal filament network. The collective action of molecular motors changes the cell’s shape and pulls it forward on surfaces and in three-dimensional environments. Other examples on a larger scale are swarms of self-propelled bacteria in liquid that seek out nutrients, or a collection of migrating immune cells searching for target cells, such as tumour cells. On a smaller scale, examples include ATP-driven molecular ion pumps, which transport particles against a concentration gradient from one cell compartment to another or into the extracellular space, and the delicate ATP-driven molecular machinery that fuses the membranes of vesicles carrying neurotransmitters or toxic material with the cell’s plasma membrane during exocytosis (expulsion of molecules into the extracellular space).
One of the foremost institutions for the study of collective behaviour of biological matter is the Collaborative Research Centre SFB 1027 ‘Physical Modelling of Non-Equilibrium Processes in Biological Systems’ at the Saarland University in Saarbrücken and Homburg. The SFB 1027 aims to achieve a quantitative understanding of the physical mechanisms at work when biological matter self-organises into complex structures to perform dynamic functions such as those described above. With this goal in mind, SFB 1027 scientists analyse the ways in which large biological molecules and cells interact physically, exert forces, move each other, and self-organise into complex functional patterns on all scales, ranging from proteins, lipid membranes and cells to biofilms and tissues.
This multidisciplinary collaboration is made up of researchers from physics, medicine and biology, and involves associated external groups from Dresden, Göttingen, Erlangen and Geneva. Combining their expertise in a range of disciplines, SFB 1027 researchers employ a two-pronged approach – studying simplified model systems, on the one hand, and whole organisms on the other. They perform theoretical and experimental modelling of medically relevant biological processes on the molecular, cellular and inter-cellular levels. Since its recent founding in 2013, SFB 1027 has published numerous findings, a few of which will be detailed below.
Illuminating the Function of Immune Cells
The immune system is our body’s means for defending against disease and decay, fighting invading pathogens and removing dead or damaged cells to keep us healthy. The innate immune system is the first line of defence against disease, providing generalised responses that slow the progress of potential disease-causing agents and helping to clear out damaged tissue at the site of an injury. The adaptive immune system is more nuanced – it works out strategies to shut down novel pathogens and develops memories of past infections so that it is better prepared to handle similar pathogens much more quickly if it encounters them again.
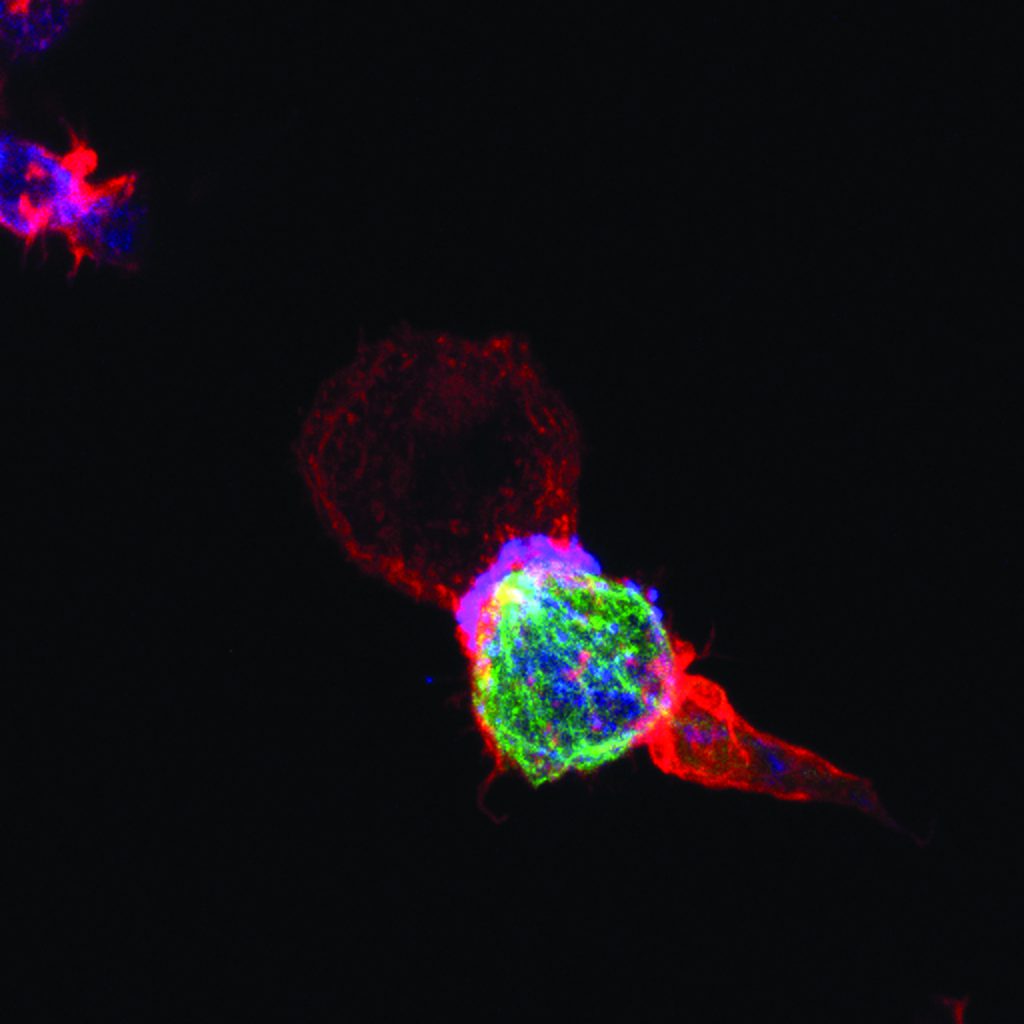
Two of the most important components of the innate and adaptive immune systems are natural killer cells and T cells. T cells and natural killer cells are the main killer cells of the human body that eliminate pathogen-infected or tumorigenic cells. CD4+ T helper cells are crucial for activating killer cells and allowing them to proliferate. These helper cells also regulate the immune response by releasing substances called cytokines.
By combining theory and experiment, SFB 1027 researchers Markus Hoth, Bin Qu, Franziska Lautenschläger, Aránzazu del Campo and Heiko Rieger aim to understand the biophysical aspects of this killing process. To be efficient, killer cells need to use optimal search strategies in complex cellular environments to locate target cells and kill them quickly. Being serial killers, they should not waste too much of their killing capacity on a single target so as to avoid inefficient killing of subsequent targets.
The researchers are working to define these killer cell search strategies in both 2D and 3D environments. Their main hypotheses are that killer cells work cooperatively and their search strategies are heavily influenced by the environment generated by other cells and metabolites. As part of this investigation, the team is analysing the transport of vesicles (bubbles in the cell that move substances around) to and from the immunological synapse (the interface between a killer cell and its target cell). They are also investigating the expression of motor proteins (proteins that derive energy from ATP to do work), and look at how the ratios of these proteins help to control cytokine transport and release at the immunological synapse and away from it.
Ultimately, their research will lead to a comprehensive understanding of how a swarm of killer cells operates cooperatively to destroy tumour cells. The efficiency of the immune response is of great importance to cancer therapy, and this was recently highlighted by the great success of immune checkpoint therapy in cancer treatment. The researchers now want to identify searching and killing parameters that increase the efficiency of tumour cell killing, as such insight could trigger new cancer therapies in the future.
Understanding Intracellular Transport
When an activated T cell or a natural killer cell has found a target cell and established an immunological synapse, so-called lytic granules (small vesicles containing cell-killing material) need to be transported to the synapse where they are released towards the target cell. Since vesicles are too heavy to be transported by mere diffusion, they are instead moved along the filaments of the cell cytoskeleton with the help of molecular motors. Molecular motors are proteins that can move along cytoskeletal filaments in specific directions. Intracellular cargo can either be transported by single motors or by teams of molecular motors.
Despite extensive research in the past years, several fundamental questions related to motor-driven cargo transport remain unanswered. This is particularly true for two-way cargo-transport, where most studies have been carried out in vivo, i.e., in a rapidly changing environment where it is difficult to disentangle the properties of the motor-cargo complex and the influence of the environment on cargo dynamics.
SFB 1027 researchers Ludger Santen, Stefan Diez and Reza Shaebani analyse cargo transport driven by teams of molecular motors using a combined experimental and theoretical approach. They study the cooperation of molecular motors and their response to varying external control parameters using in vitro assays. Using these setups, the researchers investigate how cargo is transported by teams of kinesin-1 and mammalian cytoplasmic dynein motors (present at defined ratios) under well-controlled experimental conditions. They then use theoretical models to characterise the contributions of individual motors in different motor-cargo complexes. Ultimately, their results allow models to be developed for two-way cargo transport along microtubules on a large scale to understand transport in neurons.
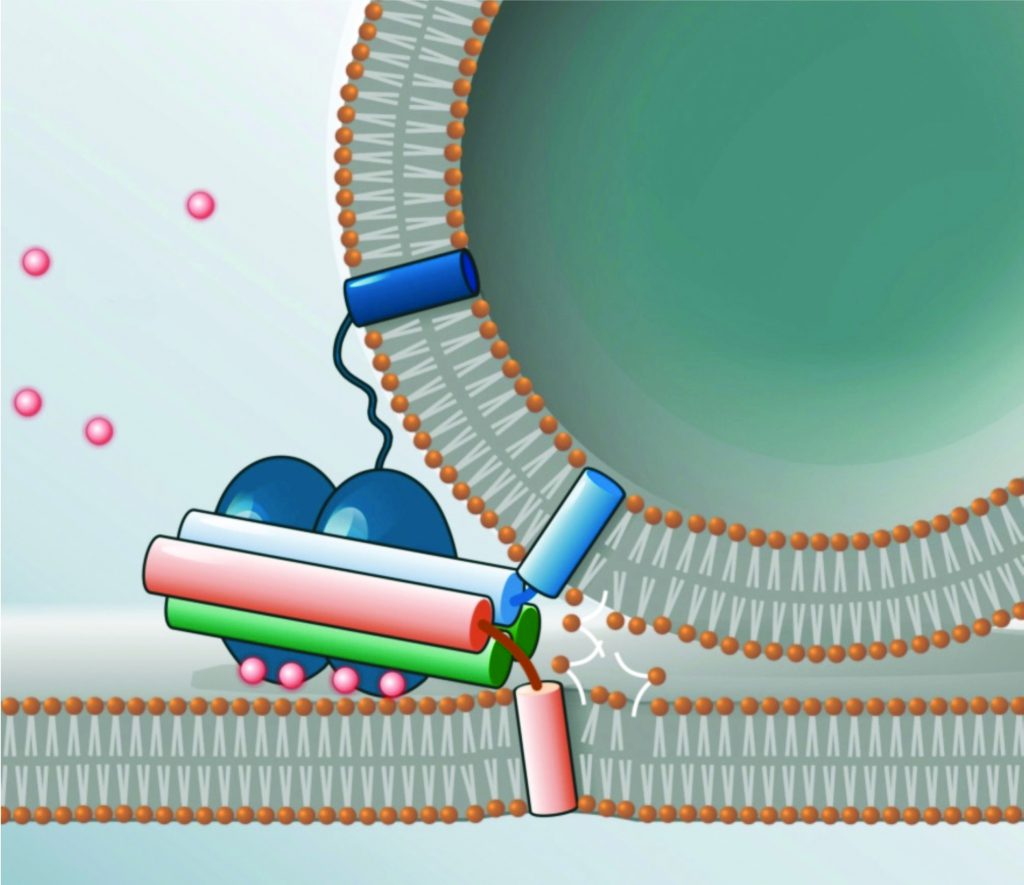 Understanding Exocytosis Machinery
Understanding Exocytosis Machinery
Once the lytic granules arrive at the immunological synapse, they must release their cell-killing contents through the cell membrane towards the target cell. This process is called exocytosis, and is a form of active transport in which a cell expels molecules through an energy-dependent process. Exocytosis and the opposite process, endocytosis, are used by all cells, because most important chemical substances are large hydrophilic (water loving) molecules that cannot pass through the hydrophobic (water hating) portion of the cell membrane without the input of energy. As well as being a process employed by killer cells, exocytosis is also the means by which neurotransmitters are released from nerve cells, to pass information across synapses in the nervous system.
In exocytosis, vesicles are carried from within the cell to the cell membrane, and their contents (hydrophilic molecules) are secreted into the extracellular environment. This secretion is possible because the vesicle transiently fuses with the outer layer of the cell membrane. Membrane fusion requires a lot of energy, and so cannot happen spontaneously. Therefore, delicate molecular machinery, made up of so-called SNARE proteins, mediates this vesicle fusion by pulling the vesicle towards the cell membrane and then opening a pore.
SFB 1027 scientists Dieter Bruns, Ralf Mohrmann, Ralf Seemann, Jean-Baptiste Fleury and Rainer Böckmann study this SNARE machinery and membrane fusion events – in vivo, in vitro and in silico – (i.e. using computer simulations). A challenging goal of their research is to build an artificial synapse, and then introduce SNARE proteins in order to study membrane fusion. With these studies, the team aims to explore the role of the flexibility of SNARE proteins, and the possible curvature and flexibility of the cell membrane they are inserted in.
A further goal is to extend their investigations from controlled vesicle fusion, which constitutes the first step of synaptic transmission (neurotransmitter release), to the activation of receptors on post-synaptic nerve cells. In this context, they will use their artificial synapse to study the trafficking properties of neurotransmitter cargoes that are released from individual synaptic vesicles and which subsequently interact with post-synaptic receptors. By exploring the transport of neurotransmitters as a function of vesicle and receptor density, the team is able to observe different types of neurotransmitter transport properties, from ‘free-flow’ to ‘traffic jams’, with implications for understanding various neurological diseases. Ultimately, this artificial synapse will be generalised to study similar processes at the immunological synapse, where during exocytosis toxic material is expelled towards the target cell.
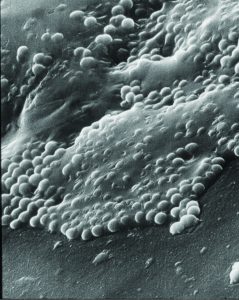 Loosening the Grip of Biofilms
Loosening the Grip of Biofilms
Another class of cell-to-cell interactions that carry strong consequences for health take place within biofilms. Biofilms are aggregates of bacteria that can form on any surface, such as dental plaque, and are often problematic to remove. Many biofilms evade the immune response, develop antibiotic resistance and can be incredibly difficult to treat when they are pathogenic. Biofilms are of particular concern in hospital settings, where they can grow on the surfaces of medical implants or catheters.
Traditionally, researchers have focused on trying to remove biofilms from surfaces, but SFB 1027 scientists, including Karin Jacobs, Markus Bischoff and Matthias Hannig opted to take a unique approach, and look at how these bacterial colonies bind to surfaces in the first place. They combined bacterial experiments with sophisticated modelling techniques to better understand what exactly allows the bacteria to bind so tightly to a surface. They found that attachment is much different than anyone thought – the bacteria manipulate proteins on the surface of their cell wall to form much tighter surface bonds than would be expected.
The SFB 1027 team found that manipulating the cell wall proteins through chemical treatments was effective in reducing the bonding efficiency. This work is a step towards the development of cleaning solutions that are extremely effective against biofilm formations.
The Future of Cell Research
The SFB 1027 team is engaged in novel and exciting cell biophysics research that could revolutionise our understanding of immune function, neural processes and bacterial behaviour. Through their multidisciplinary approach, the team is shining light on molecular processes in and between cells that could lead to new treatments and better healthcare worldwide.
Meet the Researchers

SPEAKER
 Professor Heiko Rieger
Professor Heiko Rieger
Professor Heiko Rieger is the Speaker of SFB 1027 on ‘Physical Modelling of Nonequilibrium Processes in Biological Systems’. In 1989, he received his PhD in Theoretical Physics from the University of Cologne, Germany, after which he worked as a postdoctoral researcher at the University of Maryland at College Park and the University of California at Santa Cruz, USA. From 1995 until 1999, he was a Heisenberg fellow at the Research Centre Jülich, Germany. Since 1999, he has been a Full Professor for Theoretical Physics at the Saarland University in Saarbrücken, Germany, where he works in the areas of statistical physics, theoretical biophysics and computational physics. His current research interests include the biophysics of the immune system, the physics of cancer and collective phenomena in active matter. He has published over 200 papers and is editor for Europhysics Letters and Journal of Statistical Mechanics and executive editor for European Physical Journal B.
E: h.rieger@physik.uni-saarland.de
STEERING COMMITTEE
Professor Karin Jacobs
Condensed Matter Physics, Saarland University
E: k.jacobs@physik.uni-saarland.de
Professor Markus Hoth
Biophysics, Saarland University
E: markus.hoth@uks.eu
Professor Bin Qu
Biophysics, Saarland University
E: bin.qu@uks.eu
Professor Volkhard Helms
Bioinformatics, Saarland University
E: volkhard.helms@bioinformatik.uni-saarland.de
PRINCIPAL INVESTIGATORS
Professor Markus Bischoff
Microbiology, Saarland University
E: markus.bischoff@uks.eu
Professor Rainer Böckmann, Rainer
Computational Biology, University Erlangen-Nürnberg
E: rainer.boeckmann@fau.de
Professor Ivan Bogeski
Cardiovascular Physiology, Georg-August-University Göttingen
E: ivan.bogeski@med.uni-goettingen.de
Professor Dieter Bruns
Physiology, Saarland University
E: dieter.bruns@uks.eu
Professor Niels de Jonge
Experimental Physics, Leibniz Institute for New Materials
E: niels.dejonge@leibniz-inm.de
Professor Aránzazu del Campo Bécares
Biomaterials, Leibniz Institute for New Materials
E: aranzazu.delcampo@leibniz-inm.de
Professor Stefan Diez
Bioengineering, TU Dresden
E: stefan.diez@tu-dresden.de
Dr Jean-Baptiste Fleury
Experimental Physics, Saarland University
E: jean-baptiste.fleury@physik.uni-saarland.de
Professor Matthais Hannig
Dentistry and Periodontology, Saarland University
E: matthias.hannig@uks.eu
Professor Volkhard Helms
Bioinformatics, Saarland University
E: olkhard.helms@bioinformatik.uni-saarland.de
Professor Karsten Kruse
Theoretical Physics & Biochemistry, University Geneva
E: karsten.kruse@unige.ch
Professor Franziska Lautenschläger
Experimental Physics, Saarland University
E: f.lautenschlaeger@physik.uni-saarland.de
Professor Ralf Mohrmann
Physiology, Saarland University
E: ralf.mohrmann@uks.eu
Professor Barbara Niemeyer
Biophysics, Saarland University
E: barbara.niemeyer@uks.eu
Professor Albrecht Ott
Experimental Physics, Saarland University
E: albrecht.ott@physik.uni-saarland.de
Professor Ludger Santen
Theoretical Physics, Saarland University
E: l.santen@mx.uni-saarland.de
Professor Manfred Schmitt
Molecular and Cell Biology, Saarland University
E: mjs@microbiol.uni-sb.de
Professor Ralf Seemann
Experimental Physics, Saarland University
E: r.seemann@physik.uni-saarland.de
Dr Reza Shaebani
Theoretical Physics, Saarland University
E: shaebani@lusi.uni-sb.de
FUNDING
German Research Foundation DFG
WEBSITE
www.sfb1027.uni-saarland.de
zbp.uni-saarland.de