Dr Spencer Proctor – A Novel Antibody with Vaccine-like Properties to Treat Heart Disease in Diabetes
Patients with diabetes mellitus face an increased risk of developing heart disease. The high levels of sugars, lipid and cholesterol associated with diabetes cause thickening of the blood vessels, increasing the incidence of coronary heart disease and stroke. The pioneering team from the Centre of Molecular Immunology, Cuba, in collaboration with Dr Spencer Proctor from the University of Alberta in Canada are testing an antibody that targets the walls of the blood vessels, preventing the binding and accumulation of cholesterol.
An Important Collaboration Between Two Countries
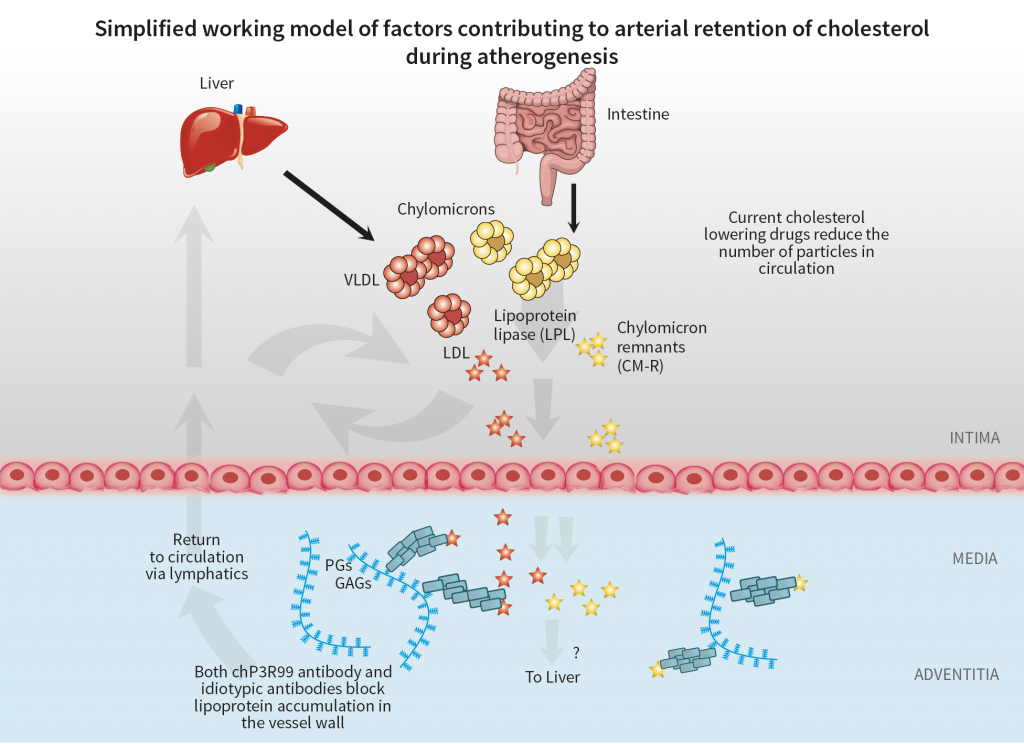
The accumulation of lipids and cholesterol in arterial vessels leads to the formation of obstructive deposits, known as plaques, that limit the vital flow of oxygenated blood to organs and tissues. This condition, known as atherosclerosis, affects millions of individuals each year, particularly those with insulin resistance and diabetes. The damage resulting from atherosclerosis depends on the location of the affected arteries. If the obstructions limit the blood flow to the heart, they may cause chest pain, breathlessness and heart attacks. Similarly, when the plaques form within the carotid arteries, they can cause a reduction or blockage of the blood supply to the brain, leading to a stroke. Atherosclerosis can affect any artery in the body and cause an array of conditions, such as kidney disease, erectile dysfunction and even numbness in the limbs.
Atherosclerosis is traditionally treated by reducing the amount of cholesterol-dense proteins, known as LDL or ‘bad cholesterol’, in the bloodstream. Dr Spencer Proctor (Canada), Dr Sylvie Marleau (Canada), Dr Ana Maria Vazquez and Dr Yosdel Soto (Cuba) have joined forces in a historical collaboration between the two countries offering a novel approach to treat atherosclerosis.
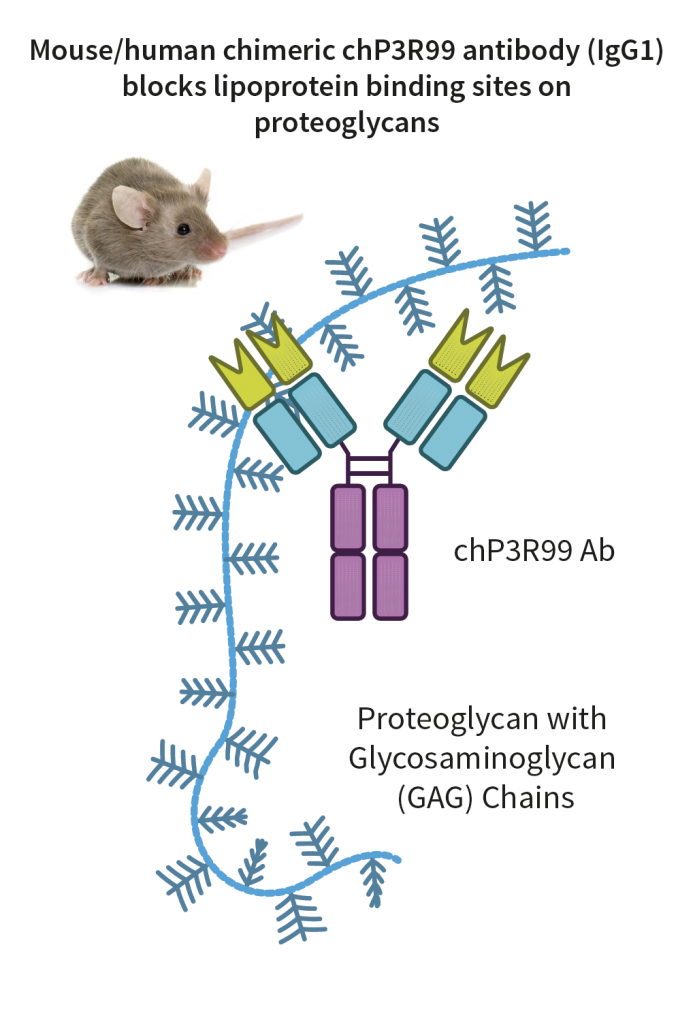
A Radically Different Approach
LDL cholesterol is produced by the liver and is the main target of current cholesterol-lowering medications. Dr Proctor and Dr Soto, however, are leading to also decrease the impact on heart disease by another type of ‘bad’ cholesterol, known as remnant cholesterol, which is derived from the diet and secreted by the intestine. The accumulation of remnant cholesterol is significantly increased during diabetes. To support this concept, it is worthy of note that the European Union and Canada have begun measuring remnant cholesterol and other non-fasting lipids as a tool to identify groups at risk of developing atherosclerosis.
These clinical outcomes have prompted Dr Proctor and Dr Soto to devise new ways of targeting atherosclerotic plaques. Rather than targeting the production of LDL, Dr Proctor and Dr Soto aim to block cholesterol from all sources from binding to specific anchor-like points in the blood vessels known as glycosaminoglycans (GAGs). GAGs are sugar side chains in the artery walls that act as sticky regions for cholesterol particles. There is currently no drug that specifically targets the GAGs on the blood vessels, preventing both LDL and remnant cholesterol from attaching.
The pioneering efforts of the Centre for Molecular Immunology (CIM) in Cuba have developed an antibody known as chP3R99. The centre has accumulated 25 years of achievements in developing monoclonal antibodies that have proved effective in the treatment of cancer and other chronic diseases. The chP3R99 antibody specifically binds to the sugar side chains, effectively preventing cholesterol particles from all sources from forming plaques on the blood vessels. This mechanism of action is of great relevance to conditions such as insulin resistance and diabetes, where the increased risk of cardiovascular disease is partly due to a greater abundance of GAGs on the arterial wall.
Pioneering the Development of a Unique Antibody
Scientists at CIM in Cuba first developed chP3R99 in mice and eventually engineered what is known as a ‘chimeric’ mouse/human antibody, a protein that has been expressed by combining the genetic material from the two different species. The team at CIM in collaboration with Dr Sylvie Marleau (University of Montreal) tested the antibody on arterial tissue from rabbits and mice, demonstrating in several publications that chP3R99 causes a significant decrease in the formation of atherosclerotic plaques. The decrease was caused by the preferential binding of the antibody to the sugar chains on the artery walls rather than reducing cholesterol levels.
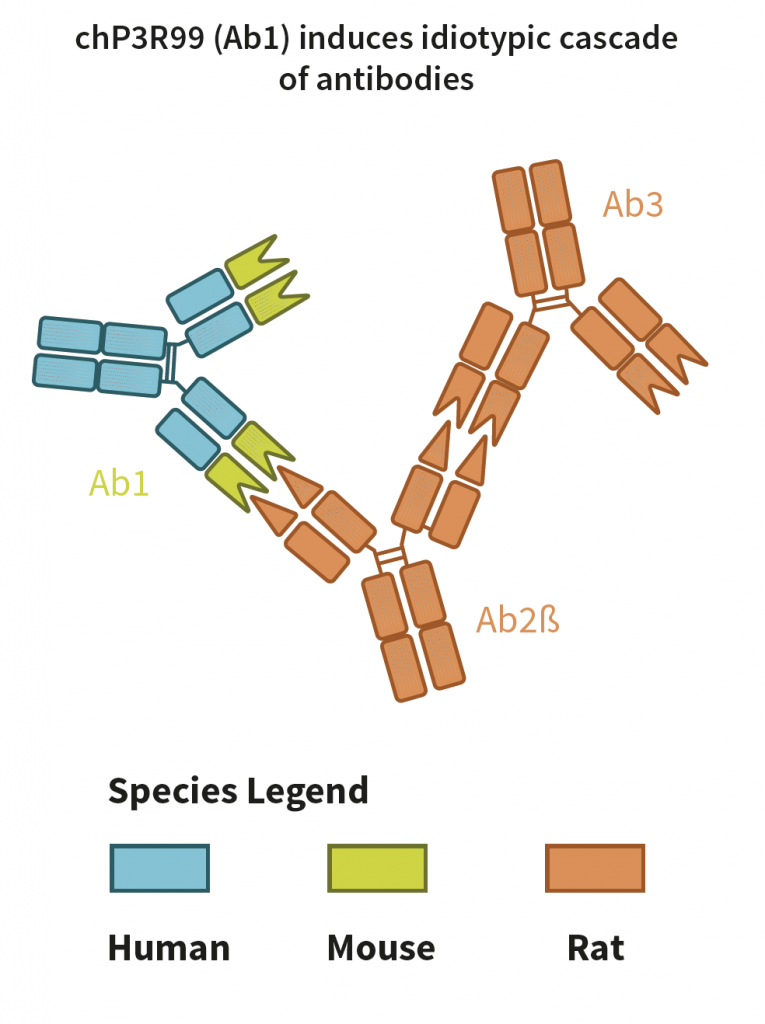
Notably, the antibody has a dual mechanism: not only does it bind to the GAGs sugar chains, but it does so in a vaccine-like fashion, which means that it does not need to be injected repeatedly in successive multiple doses to maintain its effect. Once injected, it will produce a cascade of secondary antibodies, which will continue to exert the protective action against the accumulation of cholesterol. The treatment has shown promising results in preclinical models of atherosclerosis. What sets apart the drug developed at the CIM from other compounds in preclinical studies is that other treatments show benefits in the early stages of atherosclerosis, while the chP3R99 antibody also halts plaque progression and stimulates regression in advanced atherosclerotic lesions.
Using the Antibody to Target Remnant Cholesterol
Dr Proctor is an expert in the field of metabolism of lipids in health and disease, particularly in relation to the interaction of lipids with arterial vessels during atherosclerosis and insulin resistance. Dr Proctor’s team has previously demonstrated that insulin resistance and diabetes correlate with an increase in the binding of remnant cholesterol to the arterial wall, causing the formation of plaques. The group has contributed to the understanding that diabetic atherosclerosis can accelerate vascular disease. This is due to the fact that insulin resistance causes an extensive re-modelling of the architecture of the arterial wall, leading to an increased production of sugar side chains in GAGs, which in turn will increase the capacity for cholesterol binding. The group have been able to publish their findings by using mice, rats and swine animal models.
In collaboration with Dr Soto and the team at CIM, Dr Proctor has gathered compelling evidence that shows that the novel antibody is able to inhibit the binding of remnant cholesterol to arterial tissue, proving the significant contribution that all forms of cholesterol and not just LDL have on atherosclerosis in animal models of insulin resistance.
The project is conducted in the state-of-the-art facilities at the University of Alberta, which hosts research excellence centres such as the Alberta Diabetes Institute and the Group on the Molecular and Cell Biology of Lipids (MCBL). The researchers there have mastered the use of fluorescent imaging techniques and confocal microscopy to trace the interaction of lipids and cholesterol with the arterial walls. In order to to complete pharmacological and toxicological studies with the chP3R99 antibody, the production process of its latest version is under development to scale up its production at pilot plant (pre-commercial) scale.
In their collaboration, the two scientists and their teams have collected numerous preliminary data that confirm the effectiveness of the antibody against cardiovascular disease in higher-order mammals. They are confident that the treatment will prove to be effective against human arterial tissue.
The Vaccine-like Properties of chP3R99
The preliminary data from the ambitious project by the Canadian and Cuban scientists show that the antibody can be used successfully both as an acute dose treatment and as a series of injections. When administered acutely, the antibody is given as a high dose intravenous injection to insulin-resistant animals with a highly re-modelled arterial wall architecture. When the arterial tissue of the treated animals is analysed ex vivo, it displays significantly reduced binding of both remnant cholesterol and LDL cholesterol to the arterial wall when compared with untreated control animals.
Interestingly, when used in experiments as a vaccine, the drug can be injected just under the skin, in multiple small doses, over several weeks. The animals treated this way are able to develop their own secondary (and tertiary) antibodies against the GAGs of the arterial wall. These ‘generational’ antibodies are in turn capable of displacing the binding of both remnant and LDL cholesterol, reducing plaque formation when compared to the control group. Although unpublished, these data show proof of principle that the novel compound can successfully be used as a vaccine for the treatment of atherosclerosis in insulin-resistant, higher-order mammals. If confirmed in humans, these exciting results offer a new and effective therapeutic strategy to treat heart disease in diabetic patients.
Future Developments
The collaborating teams hope to corroborate their findings with in vitro studies, to map in detail the mechanism of binding of the novel antibody to GAGs, specifically under conditions of insulin resistance. They aim to confirm that the antibody inhibits both remnant and LDL cholesterol and to ascertain whether there is a competition between the two forms of cholesterol for the binding sites on the arterial wall matrix. They also plan to use discarded donor human blood vessels to measure the protective effect of the antibody against cholesterol retention and atherosclerotic plaque formation in humans.
All the data obtained so far make the two teams confident that the novel antibody will be effective when tested in human-derived specimens. If this is confirmed, the drug will undergo phase 1 clinical trial, offering new hope of long-time protection for millions of individuals with cardiovascular disease.
For more inforamtion visit: www.cardiovacc.ualberta.ca
Short on time? watch this animation
Reference
https://doi.org/10.33548/SCIENTIA648
Meet the researchers

Dr Spencer Proctor
Division of Human Nutrition
Director, Metabolic and Cardiovascular Diseases Laboratory
Molecular Cell Biology of Lipids Group, Women & Children’s Health Research Institute, Alberta Diabetes and Mazankowski Heart Institutes
University of Alberta
Canada
Dr Spencer Proctor is full professor at the University of Alberta and a principal member of the Alberta Diabetes Institute. Dr Proctor trained as a physiologist and cardiovascular scientist in both Australia and Canada. In 2004, he founded the Metabolic and Cardiovascular Diseases Laboratory (MCVD) at the University of Alberta. Dr Proctor and the MCVD Laboratory are contributing to the link between nutrition and dietary-related chronic disease such as obesity and diabetes to increased risk of cardiovascular disease. Among Dr Proctor’s most important findings is the development of a model of over-production of intestinal derived remnant cholesterol particles during insulin resistance/diabetes that is thought to contribute to cardiovascular disease risk.
CONTACT
E: spencer.proctor@ualberta.ca
Twitter: @ProctorMCVDLab
KEY COLLABORATORS
Dr Ana Maria Vazquez (Centre for Molecular Immunology, Cuba)
Dr Sylvie Marleau (Faculty of Pharmacy, University of Montreal, Canada)
FUNDING
Canadian Institutes for Health Research
Heart and Stroke Foundation of Canada

Dr Yosdel Soto López
Department of Immunobiology, Head
Division of Immunology and Immunotherapies
Centre for Molecular Immunology
Havana, Cuba
Dr Yosdel Soto obtained his PhD in Biological Sciences from the University of Havana, Cuba. Since 2016 he has acted as Head of the Project on Atherosclerosis and is currently the Head of the Department of Immunobiology at the Centre for Molecular Immunology in Cuba. He has authored several publications that demonstrate the therapeutic potential of chP3R99 against atherosclerosis and has been instrumental in the development and characterisation of the latest version of the antibody enriched in arginine residue in a strategic position in the variable region of the antibody, therefore increasing binding effectiveness. In collaboration with Dr Proctor, he is working on the use of the antibody as a vaccine for the treatment of atherosclerosis in diabetes. Dr Soto has received numerous prestigious awards for his work on developing this vaccine and is a Member of the Cuban Academy of Sciences.
CONTACT
E: yosdel@cim.sld.cu
Twitter:
KEY COLLABORATORS
Dr Sylvie Marleau, Faculty of Pharmacy, University of Montreal, Canada
FUNDING
Centre for Molecular Immunology
Heart and Stroke Foundation of Québec
Heart and Stroke Foundation of Canada
International Atherosclerosis Society
FURTHER READING
R Sarduy, V Brito, A Castillo, et al, Dose-Dependent Induction of an Idiotypic Cascade by Anti- Glycosaminoglycan Monoclonal Antibody in apoE−/− Mice: Association with Atheroprotection, Frontiers in Immunology, 2017, 8, 232.
V Brito, K Mellal, KF Zoccal, et al, Atheroregressive Potential of the Treatment with a Chimeric Monoclonal Antibody against Sulfated Glycosaminoglycans on Pre-existing Lesions in Apolipoprotein E-Deficient Mice, Frontiers in Pharmacology, 2017, 8, 782.
Y Soto, N Mesa, Y Alfonso, et al, Targeting arterial wall sulfated glycosaminoglycans in rabbit atherosclerosis with a mouse/human chimeric antibody, mAbs, 2014, 6(5), 12–11.

Want to republish our articles?
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence
(CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
More articles you may like
Dr Shigetaka Hayano | The Rubber Revolution: Cracking the Code for Tire Recycling!
Traditionally, rubber waste was nearly impossible to recycle due to crosslinked sulphur bonds. But a team of researchers led by Dr Shigetaka Hayano from Zeon Corporation, in Japan, have achieved a groundbreaking feat in rubber recycling. Using mild conditions for the reaction, scientists have overcome the unfavourable cross-linked structure and have achieved recovery of rubber’s original monomers. This process restores cyclopentene monomers with 90% efficiency, allowing old tires and industrial rubber waste to be chemically recycled into high quality materials. If scaled up, this innovation could revolutionise waste management, reduce environmental pollution, and enable a circular economy for rubber production.
Prof Doron Lancet | Reproducing lipid micelles permit early Darwinian evolution
The origin of life has puzzled science and philosophy for thousands of years. While the prevailing scientific narrative is of an ‘RNA world’ or ‘polymers first’ approach, Prof Doron Lancet’s group of the Weizmann Institute of Science in Rehovot, Israel, present an alternative, describing a ‘lipid world’ scenario as a plausible origin for life, in which lipid micelles (simpler structures than cell-like vesicles) could have been nanoscopic protocell precursors. Core to the theory is that these micelles could exhibit catalysis and compositional self-reproduction, passing information through the mixture of their lipid components rather than through a genetic sequence.
Dr Sebastian Rabien | Making Membrane Mirrors for Future Space Telescopes
Mirrors play a key role in space telescopes, but to keep increasing the scale of this technology, mirrors need to be light and compact, so they can be transported in spacecraft, but also able to be adaptively corrected and controlled to ensure their accuracy. Dr Rabien and his colleagues from the Max Planck Institute for Extraterrestrial Physics, in Germany, have developed a technique to make extremely thin and lightweight mirrors, which can then be controlled with adaptive optics, making them a potential solution for larger space telescopes.
Dr Alyson J McGregor | Uncovering the Gender Gap in Medical Research: How Sex Differences Impact Healthcare Outcomes
Medical research has historically focused predominantly on male subjects, leading to dangerous gaps in our understanding of how diseases and treatments affect women. Dr Alyson McGregor from University of South Carolina School of Medicine Greenville has devoted her career to addressing this critical issue, highlighting how biological sex differences impact health outcomes. Her work demonstrates that ignoring these differences can have life-threatening consequences and advocates for more inclusive research practices to ensure safe and effective healthcare for everyone.




