Dr Baowei Fei – A New Technique for Targeted Prostate Cancer Biopsies
Two-dimensional transrectal ultrasound (TRUS) guided biopsy is the standard method for prostate cancer diagnosis. However, the technique is limited in one respect – it can be prone to sampling error. Cancers can be missed, or their severity grossly underestimated. To address this, Dr Baowei Fei, from the University of Texas (UT) at Dallas and UT Southwestern Medical Center, is pioneering a technique that merges positron emission tomography (PET) with TRUS to detect prostate cancer more accurately than before.
Detecting Prostate Cancer
Prostate cancer is one of the most common types of cancer – it affects some one million men globally. It occurs when cells within the prostate gland grow rapidly and abnormally in comparison to normal cells. As is the case with many diseases, early detection is vital. Picking it up early increases survival rates, improves patient outcomes, and helps the patient’s family manage the disease. The problem is, however, that accurate detection of the disease has been, and continues to be, a challenge.
There are a few main types of diagnostic techniques that medical professionals use. For example, when doctors suspect a patient has prostate cancer, they will conduct a ‘prostate specific antigen’, or ‘PSA’ test. This is a test that measures relative PSA levels – a chemical made by both healthy and cancerous prostate cells – in the blood. However, PSA test results can be quite unreliable. In fact, even those with normal PSA levels have been later found to have prostate cancer.
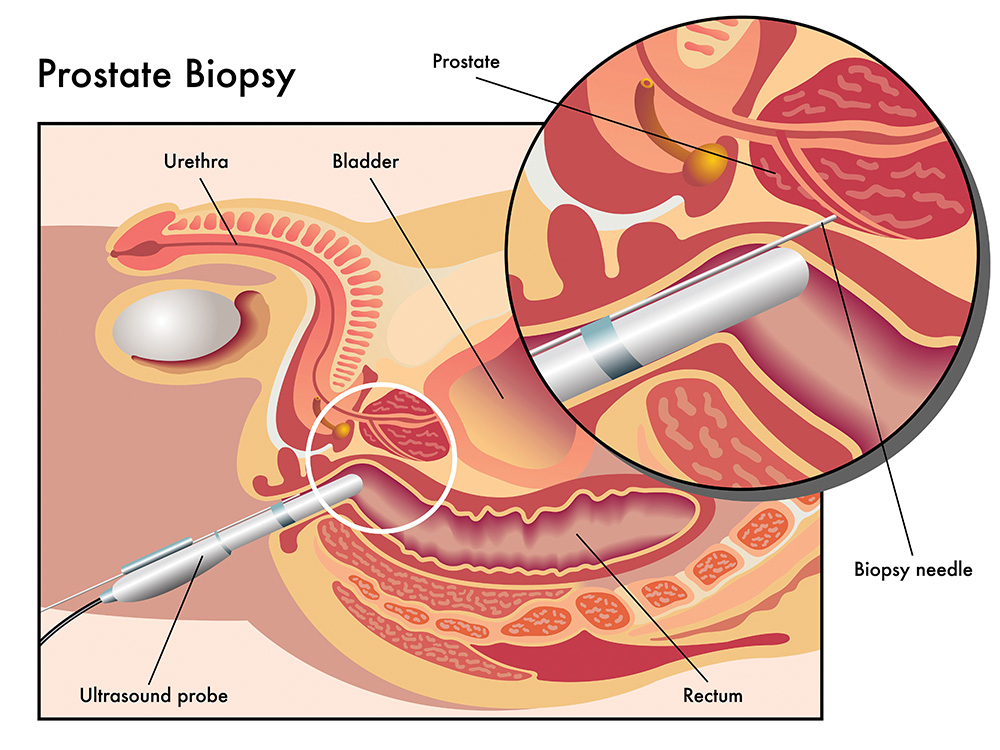
Another diagnostic tool is prostate biopsy. A biopsy removes small pieces of tissue from different parts of the prostate for examination under a microscope. The method used specifically for prostate cancer diagnosis is known as ‘two-dimensional transrectal ultrasound’, or ‘TRUS’, guided biopsy. In this technique, ultrasound imagery is used to guide sampling. However, TRUS guided biopsy also has limitations. It can be misdirected, and thus prone to sampling errors. In other words, the samples taken for biopsy are not representative of the cancer’s severity – or they can even miss the cancer altogether.
So, in summary, there are currently no tests available with sufficient accuracy to screen men for early signs of prostate cancer. Therefore, scientists have been working tirelessly to develop better tests. One such scientist is Dr Baowei Fei, from the University of Texas (UT) at Dallas and UT Southwestern Medical Center, USA. His goal is to enhance the effectiveness of the TRUS guided biopsy technique by incorporating sensitive ‘positron emission tomography’ (PET) imagery.
This multimodal approach produces detailed images of the prostate, and highlights areas of potential cancerous growth. With these images at their disposal, physicians can target biopsies to affected areas. This, in turn, produces more representative samples, and increases the likelihood of a correct cancer diagnosis.
Establishing the Merits of PET/TRUS Guided Biopsy
In research published in 2016, Dr Fei and his colleagues reviewed many of the current techniques for prostate cancer detection. They noted that PET-based techniques can indeed provide a lot of information about the biochemical behaviour of a cancer. They also highlighted that it could be used in combination with ultrasound to guide biopsy sampling to areas of concern. ‘Molecular image-directed, 3D ultrasound-guided biopsy represents a new trend for the diagnosis of prostate cancer and for monitoring of prostate cancer,’ they concluded.
To consolidate this, in 2017, Dr Fei and a colleague conducted a review of the various applications of PET imaging-directed biopsy. They focused their review on cancers in a variety of areas of the body, such as the brain, bones, breasts, abdomen, pelvis, neck and vital organs – as well as glands such as the prostate. From that they concluded, ‘PET-directed biopsy has an increasing role in the diagnosis and staging of various diseases.’ Why? The guidance provided by molecular imagery, they noted, has great potential to increase the detection rate and improve diagnostic accuracy.
PET Molecular Imaging with New Radiotracer
Having proven that PET is effective for detecting disease, and especially cancer, Dr Fei wanted to hone a PET technique for the study of prostate cancer. For this purpose, Dr Fei and his colleagues needed to find a suitable radiotracer. In a medical context, a radiotracer is a compound that emits radiation from within the body of a patient. After adding a radiotracer to a patient’s blood stream, for example, doctors get a map of metabolic activities and patterns. This gives them an insight into the tissue under examination at the molecular level. They can also add these compounds to tumours, glands or organs to survey the metabolism within them.
Dr Fei and his team selected a radiotracer that stood out above others – anti-1-amino-3-18F-fluorocyclobutane-1-carboxylic acid, or 18F fluciclovine (shortened to fluciclovine). This compound had already demonstrated its effectiveness in creating detailed PET imagery of tumour activity, so it was a natural choice for further research. Furthermore, fluciclovine can isolate target areas as it is taken up more readily by cancerous tissue than by normal tissue.
To understand this process better though, Dr Fei and his colleagues examined the specific uptake behaviour of fluciclovine in the prostate. To do this, they introduced fluciclovine to two different sets of lesions – those that were malignant (cancerous) and others that were benign (not harmful). After appropriate uptake time, 3D imaging techniques were used to map the uptake of fluciclovine, and thus determine uptake effectiveness in the two different lesion types. The team confirmed what they has suspected – fluciclovine does indeed accumulate more readily in areas of cancerous growth and is an excellent radiotracer for differentiated imaging techniques involving cancer in the prostate.
How Fluciclovine Helps Doctors
In real terms, they now had a sure-fire way of differentiating between, and producing clear images of cancer-affected areas using fluciclovine. The subsequent biopsy that doctors take, therefore, can be guided to the exact site of the tumour. This addresses the sampling errors common to other techniques, and therefore decreases the likelihood of a cancerous growth being missed.
More targeted biopsies also provide insight into the cancer’s aggressiveness. Suppose a biopsy is taken from an area of minor cancerous growth, while an area of significant or aggressive growth is missed. The biopsy would still confirm that cancer is present. But it would also suggest that the cancer is only minor in nature. If the area of significant growth had been sampled, on the other hand, physicians would have a completely different picture of the disease. This has implications for the subsequent management of the disease, and thus, patient outcomes. So, Dr Fei’s PET/TRUS technique is not just about detecting whether or not cancer is present, but rather, getting a full picture of its progression.
Having selected a robust and powerful technique, the question was – could PET be incorporated with TRUS to produce better imagery of the prostate, and guide biopsy sampling to areas of potential cancerous growth, in a real-world setting? Dr Fei and his team set out to investigate this question by way of a clinical trial.
PET/TRUS Guided Biopsy Proves to Be Effective
The purpose of the clinical trial was to see if using a PET scan and transrectal ultrasound (TRUS) guided biopsy together would detect prostate cancer more accurately than the standard 2D approach, which uses TRUS only to guide the biopsy.
In the trial, which Dr Fei and his team conducted in 2015–2017, the 21 participants were first injected with a solution containing the radiotracer fluciclovine. The patients then underwent abdominal and pelvic scans using PET. Next, an ultrasound probe, of about 2.5 centimetres in diameter, was inserted into the rectum of each patient. Using special apparatus, a 3D ultrasound image was then acquired from each patient’s prostate.
However, when the ultrasound image was processed and combined with the original PET scans, Dr Fei and his colleagues could produce incredibly detailed 3D images of the prostate. And, most importantly, areas of suspected cancerous growth were clearer than ever. In fact, based on these composite images, the researchers could categorise suspicious regions as being 3 (equivocal), 4 (medium) or 5 (high), with regard to their potential to be cancerous. The team then guided the biopsy needle to these suspicious areas. They then processed these samples histologically for pathological examination.
The research team identified a total of 50 suspicious lesions with fluciclovine PET for the 21 patients involved in the study. The likelihood of a positive result using the TRUS guided biopsies that followed increased when the ‘suspicious level’ of the PET identified lesions increased (based on the team’s categorisation system discussed above). In other words, cancerous growths, especially those that had been identified as more significant, were more readily detected when PET targeting was used in conjunction with traditional TRUS guided biopsy.
In summary, their method for targeted biopsy detected cancerous growth in 47.6% of patients, as opposed to 28.6% where typical biopsy methods were used. This increase in cancer detection rates was also achieved using fewer samples. This too is a significant result as their technique may reduce the time needed to treat patients and limit damage done to healthy tissue. Not only that, this study was the first-in-human trial that used PET imaging to direct 3D ultrasound-guided biopsy of the prostate. All round, the research was a great success.
Improving Outcomes for Patients with Prostate Cancer
Many different diagnostic tools are used to detect prostate cancer, such as PSA tests and standard TRUS guided biopsy. However, each one has limitations and many cancers are missed, or their severity is grossly underestimated. As a result, scientists and physicians are working to improve current diagnostic techniques. Dr Fei has been investigating the merits of PET, combined with the traditional TRUS method, as a more targeted method for prostate biopsy. His team’s technique utilises an effective PET radiotracer, namely, fluciclovine. Other radiotracers, such as 68Ga-PSMA, can also be used by this innovative technology.
Dr Fei’s technique essentially combines high-sensitivity PET imagery with real-time information from ultrasound for improved cancer detection. A recent clinical trial, conducted by Dr Fei and colleagues, has demonstrated that this composite PET/TRUS guided biopsy method is more effective in terms of retrieving a representative sample, and making a correct diagnosis for prostate cancer, than the currently used TRUS-only technique.
As was noted at the outset, prostate cancer affects millions of people and has devastating effects on sufferers and their families. Of course, a complete cure for the disease is extremely sought after. In the meantime, however, improved diagnostics and early detection can improve patient treatment, and thus survival rates. By combining PET with the traditional TRUS guided biopsy method, Dr Fei and his team are working to improve prostate cancer detection, thus increasing the chances of a correct diagnosis.
Reference
https://doi.org/10.33548/SCIENTIA367
Meet the researcher

Dr Baowei Fei
Dept. of Bioengineering, University of Texas (UT) at Dallas
Dept. of Radiology, UT Southwestern Medical Center
Dallas, TX
USA
Dr Baowei Fei is Cecil H. and Ida Green Chair in Systems Biology Science, Full Professor of Bioengineering at the University of Texas (UT) at Dallas, and Professor of Radiology at UT Southwestern Medical Center. Before he was recruited to Dallas, Dr Fei was Associate Professor with tenure in the Department of Radiology and Imaging Sciences at Emory University, where he was Director of the Precision Imaging – Quantitative, Molecular & Image-Guided Technologies Program and Director of the Quantitative Bioimaging Laboratory. He was also a faculty member in the Department of Mathematics & Computer Science at Emory. Dr Fei was a Georgia Cancer Coalition Distinguished Scholar, served as a faculty member, and mentored PhD students in the Joint Department of Biomedical Engineering at Emory University and Georgia Institute of Technology. He received his PhD from Case Western Reserve University, Ohio, USA. Dr Fei’s research interests include biomedical imaging, image-guided intervention, medical instrumentation, quantitative Imaging, cancer treatment, hyperspectral imaging, image processing and analysis, artificial intelligence, machine learning and augmented reality. Dr Fei is a Fellow of the American Institute for Medical and Biological Engineering and a Fellow of the International Society for Optics and Photonics (SPIE). In recent times, Dr Fei has been researching the merits of PET/ultrasound guided biopsy as a means of effectively detecting prostate cancer – even taking the technique to clinical trials.
CONTACT
W: https://fei-lab.org/baowei-fei/
KEY COLLABORATORS
Dr David M. Schuster, Emory University School of Medicine
Dr Neil Rofsky and Dr Daniel Costa, The University of Texas Southwestern Medical Center
FUNDING
National Institutes of Health (NIH)
FURTHER READING
B Fei, OA Abiodun-Ojo, AA Akintayo, O Akin-Akintayo, F Tade, PT Nieh, VA Master, M Alemozaffar, AO Osunkoya, MM Goodman, DM Schuster, Feasibility and Initial Results: Fluciclovine PET/Ultrasound Fusion Targeted Biopsy of Recurrent Prostate Cancer, Journal of Urology, 2019. DOI: 10.1097/JU.0000000000000200
OA Abiodun-Ojo, AA Akintayo, O Akin-Akintayo, F Tade, PT Nieh, VA Master, M Alemozaffar, AO Osunkoya, MM Goodman, B Fei, DM Schuster, 18F-Fluciclovine Parameters on Targeted Prostate Biopsy Associated with True Positivity in Recurrent Prostate Cancer, Journal of Nuclear Medicine, 2019. DOI: 10.2967/jnumed.119.227033B Fei, DM Schuster, PET Molecular Imaging–Directed Biopsy: A Review, American Journal of Roentgenology, 2017, 209, 255.
B Fei, PT Nieh, VA Master, Y Zhang, AO Osunkoya, DM Schuster, Molecular Imaging and Fusion Targeted Biopsy of the Prostate, Journal of Clinical and Translational Imaging, 2016, 5, 29.


Creative Commons Licence
(CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
More articles you may like
Dr David Hansen – Abhishek Juneja | Coming of Age: Active Balancing in Adolescence
Adolescence, the transitional stage between childhood and adulthood, comes with challenges as well opportunities for growth and learning.
Jella van de Laak | Could Tumour- Targeting Bacteria Enhance Cancer Immunotherapy?
Cancer immunotherapy has transformed treatment outcomes, yet many patients still fail to respond. At Maastricht University, Jella van de Laak, working with Professor Ludwig Dubois and Professor Jan Theys, is exploring how tumour-targeting bacteria could improve immune responses and expand treatment success.
Distinguished Professor Michael Zhdanov | Mapping Magma and Drilling for Oil: New Methods for Geophysical Modelling
Geophysicists use a variety of different methods to peer beneath the Earth’s surface. Seismic activity, gravitational fields , and magnetic fields each offer their own windows into the world underground, but, in isolation, are incapable of giving us the full picture. Combining data from distinct geophysical surveys, however, is its own challenge. For a number of years, a team of researchers led by Professor Michael Zhdanov has worked to develop a mathematical framework capable of generating detailed geophysical models from multiphysics data.
Through application to modelling magma chambers underneath Yellowstone and searching for oil deposits in the Barents Sea, they demonstrate that their approach can produce robust and accurate predictions
Professor Deanna Sellnow | How Strategic Communication Can Save Lives When Crisis Hits
Professor Deanna Sellnow from Clemson University argues that crisis and risk communication research must evolve beyond simply describing and deconstructing problems to actively empowering people protect themselves and their communities. In an era of increasing globalisation, she proposes that researchers adopt learner- and learning-centered approaches to ensure communication does not simply inform people about dangers, but also teaches them how to respond effectively (e.g., to reduce risks, mitigate harms, improve safety, save lives) and why.




