Dr Mary Whitman – From Nerve Development to Vision: A Bumpy Ride
The eye movement disorders strabismus (characterised by eye misalignment) and nystagmus (characterised by involuntary oscillation of the eyes) together affect up to 5% of the population and have a detrimental impact on vision. These disorders also impact on facial appearance and social interaction, which may, in turn, lead to psychological difficulties. Dr Mary Whitman, at the Boston Children’s Hospital, USA, is working to understand the genetic causes and neurological mechanisms underlying eye movement disorders to improve treatment and, ultimately, prevent their onset.
Single-vision Lens Versus Bifocals
Accommodative esotropia is one of the most common types of strabismus observed in children. It is characterised by an inward deviation of the eyes when trying to focus. Early detection is essential for successful improvement and lack of appropriate treatment can result in severe and long-term damage to vision. Children with a specific subtype of accommodative esotropia have traditionally been treated with bifocal glasses to correct both distance and close vision. They are more expensive than single-vision glasses and are often more difficult for young children to get used to.
Dr Mary Whitman at the Boston Children’s Hospital, USA, along with her colleagues called this approach to treatment into question. In a paper published in 2016, Dr Whitman reported having retrospectively studied 180 children, comparing the improvement of the esotropia and development of depth perception after treatment with single-vision (only distance correction) or bifocal glasses. She observed similar outcomes in both groups, and a lower risk of surgery in children using single-vision lenses. This study was the first to indicate that single-vision lenses represent the preferable option due to reducing the cost and complexity of care, as well as improving treatment outcomes. On the basis of these findings, the American Academy of Ophthalmology has now recommended single-vision rather than bifocal glasses for the treatment of accommodative esotropia in children.
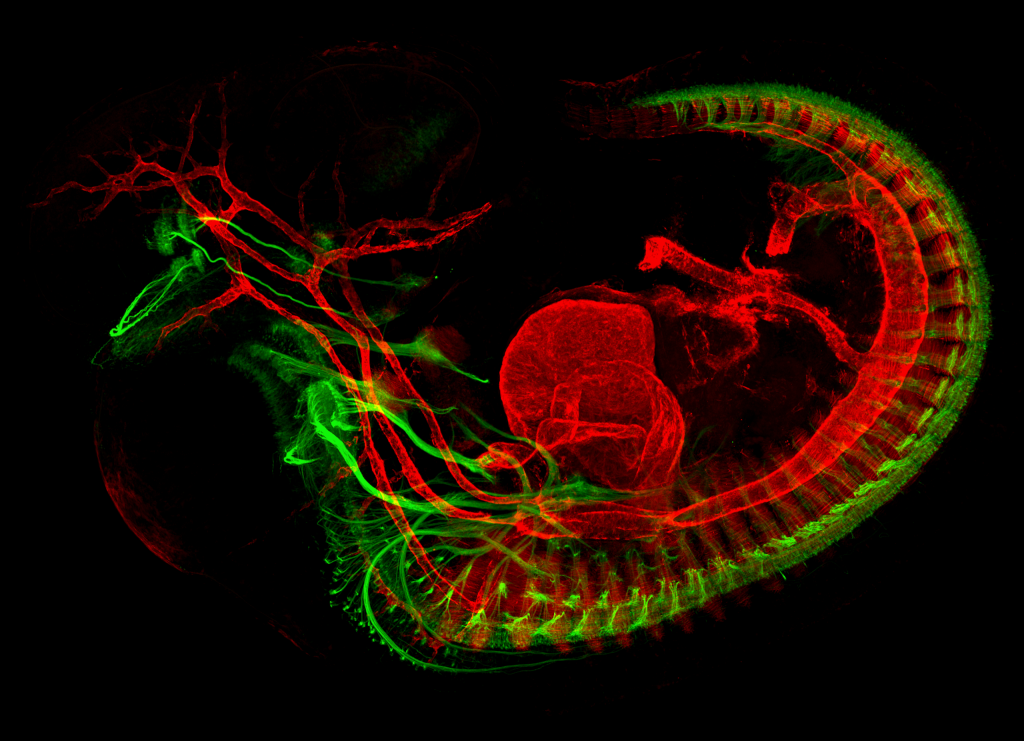
Developing mouse embryo. Motor nerves are in green and muscles are in red. Credit: Mary Whitman.
The Heritable Component of Comitant Strabismus
Strabismus alone affects 4% of the population. Noncomitant strabismus occurs where the deviation of the eye depends on the direction of the gaze, whereas comitant strabismus occurs where the deviation remains the same in all directions. Comitant strabismus is also highly associated with psychological difficulties, and the study of its genetic patterns is required to understand its cause and pathophysiology. As early as 400 BC, Hippocrates described the familial clustering of comitant strabismus and was the first to propose the possibility of parent-children transmission. Since then, observations of families and twins have confirmed a genetic contribution.
Dr Whitman contributed to an important genome-wide association study published in 2018 that sought to identify the potential associations between comitant esotropia and genetic regions. Such approaches allow scientists to determine which genetic region is most likely to be associated with an observed disease. Genome alterations can be very small variations of the genome (called SNP, standing for single nucleotide polymorphism), or larger alterations, such as the deletion or insertion of genetic material.
The DNA of large cohorts of people from the United States, the United Kingdom, and Australia was meticulously studied. A significant association between comitant esotropia and an SNP of the gene WRB located on chromosome 21 was identified in this work. The WRB gene is widely expressed in foetal and adult tissues, and codes for a protein playing a critical role in cellular function. Consistent with these findings, it had previously been demonstrated that genetically modified zebrafish lacking the WRB gene are blind and deaf.
Dr Whitman and her colleagues have very recently conducted a genetic association study in which more than 2,000 patients with esotropia were compared with an even larger sample of control participants. The researchers identified that three rare, recurrent DNA duplications (repeated stretches of DNA) increase the risk of esotropia. This important work is a step forward in understanding the pathophysiology of strabismus, although the functional consequences of these duplications, and the genes and regulatory regions involved in them require further exploration.

The Brain Circuitry Behind Sharp Vision
Although a high level of control over eye motion is necessary for sharp vision, little is known about the mechanisms responsible for the development of the ocular motor system. Information has come from the study of ocular congenital cranial dysinnervation disorders (CCDDs). Patients with CCDDs are unable to move one or both eyes in certain directions, and can have involuntary movements of the eyes, the eyelids or the face, sometimes leading to abnormal head or chin positioning. CCDDs were originally thought to be caused by muscle abnormalities, but studies primarily from Dr Elizabeth Engle’s group at Boston Children’s Hospital have demonstrated that CCDDs are mainly caused by altered muscle innervation rather than muscle abnormality. This represented a breakthrough for researchers in this field and offered new insights into normal development. Whilst some genetic causes of CCDDs have been identified, the development of the complex brain circuitry controlling eye movements remains much of a mystery.
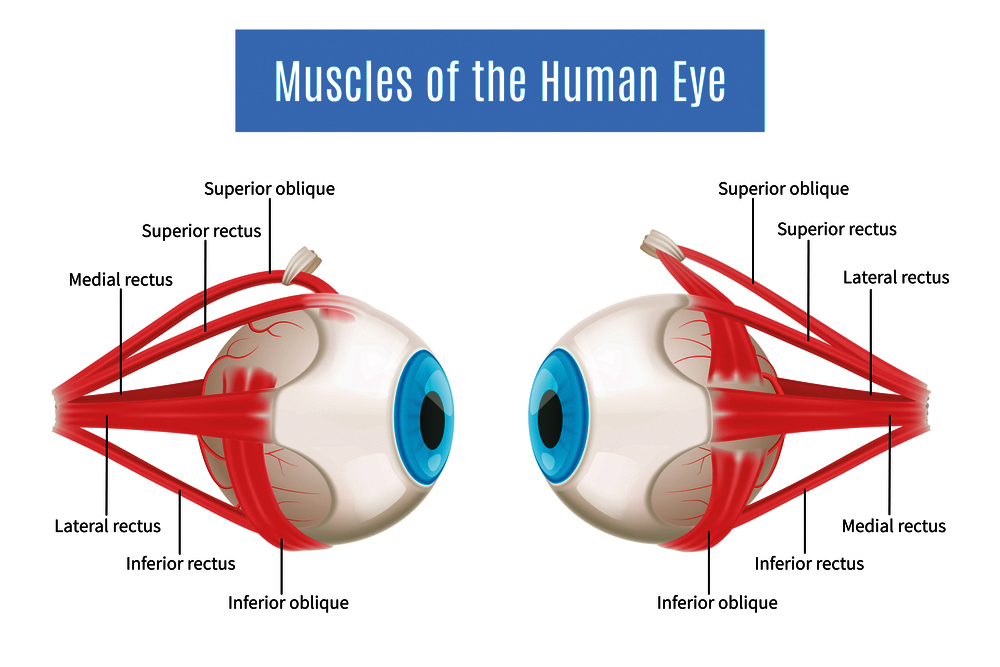
Eye movements are controlled by six extraocular muscles (EOMs) that are innervated by three cranial nerves (CN3, CN4 and CN6), and eyelid opening controlled by a seventh muscle, innervated by CN3. Typically, the neuron axons that are responsible for the transmission of the signal emerge from the brainstem; assembling into a nerve that stretches across the brain to reach the orbit and its corresponding EOM. Unfortunately, this is not a straightforward path and many disruptions can occur along the way, for example, during neuron specification, axon growth, or the trajectory development. Previous studies had already suggested a potential role for EOMs in this process, triggering Dr Whitman’s curiosity.
Together with her team and published in 2017, Dr Whitman generated a new line of genetically modified mice that lacked EOMs and expressed fluorescent protein in the ocular motor nerves (responsible for motor action of the eyes and eyelids), allowing scientists to temporally and spatially follow the nerves development in healthy mice embryos and those lacking EOMs. Orbital dissections and imaging techniques provided precise images. Both healthy and mutant embryos had similar ocular motor nerve trajectories from the brainstem to the orbit. However, a difference appeared in their terminal trajectories – nerves from mutant embryos were not able to correctly develop terminal branches to reach the targeted EOM. Embryos lacking EOM showed thin and blunt terminal nerve branches whilst healthy embryo images revealed strong and defined terminal branches.
Most prior studies did not permit the visualisation of the terminal branching, but Dr Whitman highlighted the local role of EOMs in guiding the three ocular motor nerves to finalise their trajectory to the correct EOM. Whether EOMs act through direct contact or are mediated through diffusible cues remained to be determined and motivated Dr Whitman and her team to pursue their research further.
Guiding the Nerves from the Brainstem to the Orbit
Although innervation of the EOMs relies on only three cranial nerves, complex mechanisms allow cranial nerves to reach their designated muscle. Along their trajectories, cranial nerves are guided by several signals that influence their direction. Up until now, studying nerve guidance has been hampered by technical challenges. Traditional in vitro assays remove nerves from their microenvironment which may influence the trajectory of the nerves, and in vivo assays are not appropriate for screening tests.
To overcome these limitations, Dr Whitman and her colleagues developed an embryonic slice culture technique allowing time-lapse imaging of the developing nerve while maintaining an intact microenvironment. Embryos expressing fluorescent protein (GFP-positive) in the oculomotor nerve (CN3) were extracted from the uterine horn and cut into slices containing both the oculomotor nucleus and the orbit. The slices were then grown on cell culture inserts and pictures of the slice were taken every 30 minutes for 3 days. ‘The first GFP-positive oculomotor axon reached the orbit/eye over the next 18 to 24 hours, and then began branching to their final targets. We find this basic timing to be recapitulated in slice culture’ says Dr Whitman.
In a research paper published in 2018, Dr Whitman and her colleagues studied the CXCR4/CXCL12 signalling influencing oculomotor nerve (CN3) development. CXCR4 is a receptor and CXCL12 its ligand, previous research already identified their role in the nervous system formation, neuron migration, and axon guidance. Adding a CXCR4 inhibitor in slice culture media led CN3 to grow dorsally (toward the back) from the oculomotor nucleus rather than ventrally (toward the front), in the direction of the orbit. These observations were confirmed in vivo, where genetically modified mice lacking CXCR4 in oculomotor neurons showed a misrouting similar to the one observed in the slice culture assay. Furthermore, loss of CXCR4 also caused EOMs to be innervated by aberrant nerves such as CN5, normally not involved in EOMs innervation. ‘It is remarkable that the loss of a single guidance factor causes such a profound change in axon trajectory and suggests there may be a repulsive factor ventrally or an attractive factor dorsally that is normally overcome by CXCR4/ CXCL12 signalling. The identity of such a factor is a subject for future study’ states Dr Whitman.
Most recently, in a study published in 2019, Dr Whitman studied a consanguineous (meaning closely related) family and linked a unique form of oculomotor synkinesis (involuntary movement of the eyes or eyelids when attempting a different movement) with an alteration of the ACKR3 gene. Interestingly, CXCR4/CXCL12 signalling is also regulated by ACKR3. Upon binding of CXCL12, the receptor ACKR3, a scavenger receptor, internalises and degrades CXCL12. Dr Whitman showed that when ACKR3 is altered, the balance of CXCR4/CXCL12 in the nerve’s surrounding is disturbed and leads to similar misrouting of oculomotor nerves observed after CXCR4 loss. Together with CXCR4/CXCL12, ACKR3 is crucial for the proper development of oculomotor nerves.
Dr Whitman remains motivated to discover the developmental mechanisms of the ocular motor system. Always innovative, her research opens doors to ground-breaking medical innovation. Cranial nerves can also regenerate after injuries, aneurysms or tumours. ‘As the nerves regenerate, they can form aberrant connections that cause debilitating synkinetic symptoms for patients. Understanding the cues that guide initial cranial nerve trajectories could lead to treatments to prevent or alleviate aberrant regeneration’ summarises Dr Whitman.
Reference
https://doi.org/10.33548/SCIENTIA559
Meet the researcher

Dr Mary Whitman
Assistant Professor
Harvard Medical School
Boston, MA
USA
Dr Mary Whitman obtained her PhD in Neurobiology from Yale University in 2008 rapidly followed by her medical degree from Yale School of Medicine. After she interned at New York University, Dr Whitman became an ophthalmology resident at Colombia University and New York Presbyterian Hospital. She further specialised in pediatric ophthalmology at Boston Children’s Hospital in 2014. In addition to being a dedicated ophthalmologist, Dr Whitman is also an Assistant Professor at the renowned Harvard Medical School. Her research focuses on understanding the mechanisms of development of the ocular motor system and the genetic causes of strabismus and ocular congenital cranial dysinnervation disorders. Using genetically modified mice models and complex imaging techniques she is a pioneer researcher in the field. Dr Whitman recently received the Early Career Clinician Scientist Award from the Association for Research in Vision and Ophthalmology.
CONTACT
E: mary.whitman@childrens.harvard.edu
W: https://eye.hms.harvard.edu/marywhitman
FUNDING
Knights Templar Eye Foundation
National Institute of Health
Massachusetts Eye and Ear
Children’s Hospital Ophthalmology Foundation
Massachusetts Lions Foundation
Boston Children’s Hospital
FURTHER READING
MC Whitman, SA Di Gioia, W-M Chan, et al, Recurrent Rare Copy Number Variants Increase Risk for Esotropia, Investigative Ophthalmology & Visual Science, 2020, 61(10), 22.
MC Whitman, N Miyake, EH Nguyen, et al, Decreased ACKR3 (CXCR7) function causes oculomotor synkinesis in mice and humans, Human Molecular Genetics, 2019, 28(18), 3113–3125.
S Shaaban, S MacKinnon, C Andrews, et al, Genome-wide association study identifies a susceptibility locus for comitant esotropia and suggests a parent-of-origin effect, Investigative Ophthalmology & Visual Science, 2018, 59(10), 4054–4064.
MC Whitman, EH Nguyen, JL Bell, et al, Loss of CXCR4/CXCL12 signaling causes oculomotor nerve misrouting and development of motor trigeminal to oculomotor synkinesis, Investigative Ophthalmology & Visual Science, 2018, 59(12), 5201–5209.
SM Michalak, MC Whitman, JG Park, et al, Ocular motor nerve development in the presence and absence of extraocular muscle, Investigative Ophthalmology & Visual Science, 2017, 58(4), 2388–2396.
MC Whitman, EC Engle, Ocular congenital cranial dysinnervation disorders (CCDDs): insights into axon growth and guidance, Human Molecular Genetics, 2017, 26(R1), R37–R44.


Want to republish our articles?
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence
(CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
More articles you may like
Prof Candis M. Morello – Prof Jan D. Hirsch | Recent innovations in pharmacy education
A pioneering research team from the Skaggs School of Pharmacy and Pharmaceutical Sciences, University of California, San Diego, United States, has been instrumental in developing innovative techniques for teaching pharmacy students. The Next Generation of Pharmacist Educators (NextGen-RxEd) programme is a new method of training the next generation of pharmacist educators and academics. To help pharmacists and pharmacy students visualise the complex issues experienced by their patients, the team led by Professors Candis Morello and Jan Hirsch developed an innovative educational tool, called the Medication Therapy Management (MTM) Spider Web.
Professor Michael Yarus | How RNA Started the Conversation That Built Life
The genetic code stores the instructions for building proteins, yet how it first arose remains unclear. It likely did not appear fully formed, but instead emerged step by step from simple chemical interactions. In this study, a team led by Professor Michael Yarus at the University of Colorado Boulder shows how early ribonucleic acid (RNA) molecules could bind specific amino acids, creating the first coding relationships. These early interactions are then refined through evolutionary processes such as duplication and merging of partial codes. By combining the experimental data with computer simulations, the work provides a testable pathway from prebiotic chemistry to the modern genetic code.
Identifying Nutritional Risk in Early Childhood: Insights from NutriSTEP®
Early childhood is a critical period for growth and development, yet many young children face nutritionrelated risks that can go unnoticed. Professor Janis Randall Simpson and colleagues have developed NutriSTEP®, validated and reliable screening tools that help identify potential nutritional concerns in toddlers and preschoolers. Their large-scale analysis of Canadian data reveals patterns in diet, behaviour, and food access that could help guide early interventions and support healthy development.
Professor Tian Yu Cao | Twistor Theory: A New Framework for Quantum Gravity
At Boston University, Professor Tian Yu Cao is rethinking the foundations of modern physics. His work builds on twistor theory which demonstrates that spacetime is secondarily derived from twistor constructions, but goes further to highlight the most important implication of the Penrose transform in that the primary physical agents can only be mathematically described by elements of cohomology with the defining feature having roots in spin. This view of primary agents combines with Cao’s other major claim that quantum behaviour itself may arise from the physical property of spin leads to a new consistent framework of quantum gravity in which long-standing puzzles in black holes (evaporations) and cosmology (transitions between cycles of cosmos) can be adequately addressed, with the crucial help from the on-going development of operator product expansion formular defined on twistor space.




