Professor Danny Reinberg – Dr Gary LeRoy | Confronting a Rare and Deadly Cancer in Children
A diagnosis of cancer is never welcome but is particularly devastating when a child is found to have diffuse intrinsic pontine gliomas – a rare but particularly aggressive and treatment-resistant malignancy in the brain. In this article, we focus on the contributions of Professor Danny Reinberg and Dr Gary LeRoy of the Sylvester Comprehensive Cancer Center at the University of Miami Miller School of Medicine to understanding what makes these tumours so dangerous and, importantly, to developing much-needed approaches to improve the outcomes for these children.
A Deadly Tumour
Oncology, the medical speciality dealing with the treatment of cancers, has seen remarkable progress during the last decade. Promising novel therapies against tumours that would have been previously untreatable are prominent in the news. Scientists are honing their skills to identify the biological changes that cause certain cells to divide uncontrollably, resulting in the appearance of cancers.
Learning about exactly what makes a cell take a wrong turn can lead to strategies for tackling the onset of cancer. Indeed, this valuable information about what has gone wrong can lead to the intelligent design of novel drugs to target those biological pathways that have become corrupted. Such novel treatments can be effective and life-sustaining, often with bearable side effects.
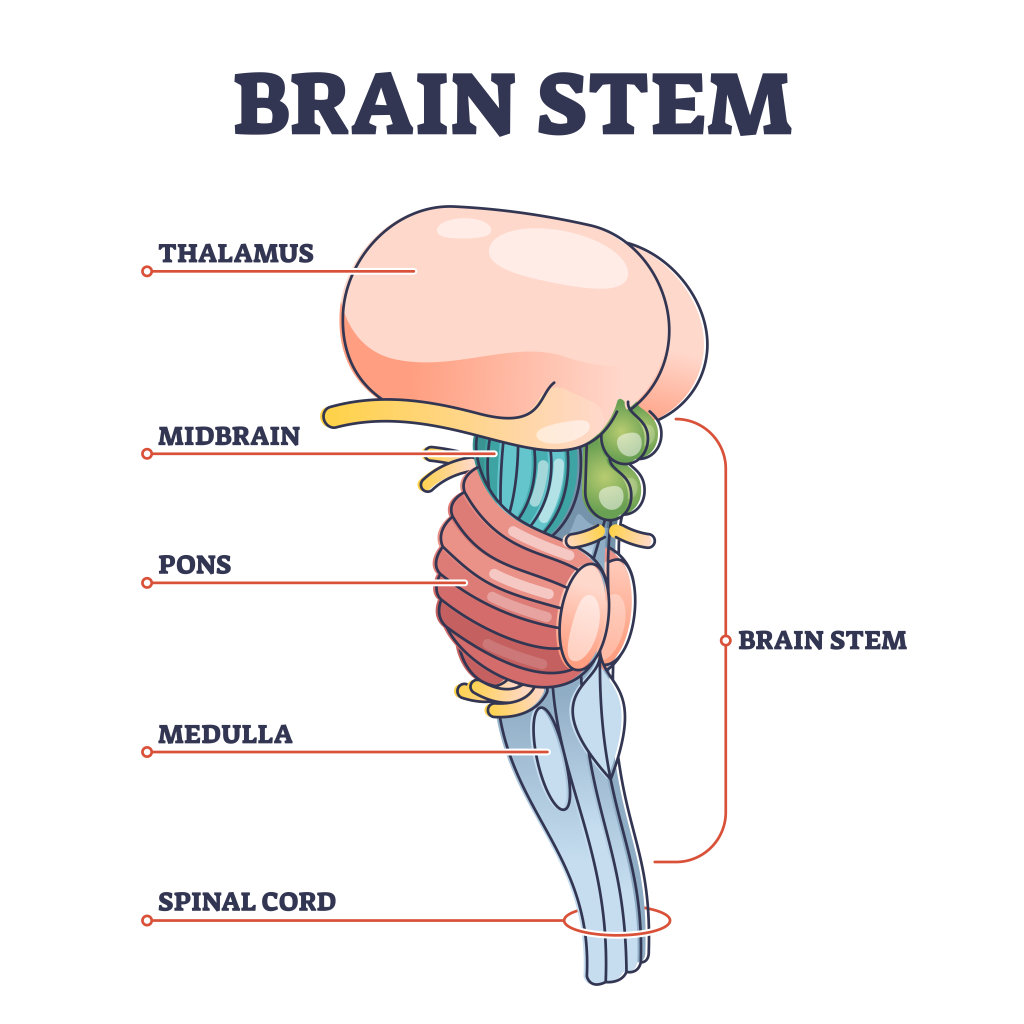
Yet, despite some advances, certain types of tumours are far more challenging, and progress in combating these diseases has been much slower. One such case involves diffuse intrinsic pontine gliomas (DIPGs). Although not commonly known, DIPGs manifest themselves in the brain stems of young children, typically between the ages of 5 and 7 years, and, unfortunately, are refractive to currently available therapies. Most of these very young patients die within 9 months of diagnosis, and the likelihood of one of them surviving more than 5 years is less than 2%. These dire statistics make DIPGs the deadliest brain tumours in children.
Genetics for the Uninitiated
To understand the work of Professor Danny Reinberg and Dr Gary LeRoy (previously at the New York University School of Medicine and now at the Sylvester Comprehensive Cancer Center at the University of Miami Miller School of Medicine), we need to briefly recap on some biology.
The DNA inside a cell contains all the instructions necessary for life. In humans, it contains around 40,000 such instructions – genes – that provide the cell with the molecules it needs for survival. Some of these genes are involved in ‘housekeeping’ functions that all cells require for survival. But other genes are tapped into during development – these genes push a cell to have specific properties and give rise to the different tissues that comprise our body. We inherit our genes from our parents, and any aberrancies in some of these genes can make us more susceptible to certain diseases. Also, some of these genes can incur aberrancies during our lifespan, leading to susceptibility to certain diseases.
It is important to understand that the chromosomes of our cells contain the DNA containing these genes. One type of cell may allow a gene to be available, while another type has this gene silenced. Whether a gene is available or silenced depends on the extent to which the DNA containing this gene is compacted in a given cell type. And, the extent to which this DNA is compacted is tightly regulated so that a lung cell has DNA available for lung-specific tasks, but its heart-specific tasks, for example, are silenced. How this DNA gets compacted involves the chemical modification of a set of histone proteins that interact with the DNA. Some chemical modifications of the histone proteins lead to segments of the DNA being physically compacted such that the genes are unavailable. Other chemical modifications of the histone proteins lead to a flexible region of DNA, and those genes are available.
So, what makes a heart cell different from a brain cell involves making genes available for a given cell type (e.g., heart cells) while silencing the genes needed for another cell type (e.g., lung cells). When this ‘programme’ malfunctions, the cell loses its personality. The cell may find ways to survive, but it is no longer normal. Indeed, an aberrancy in one of the histone modifications that silences DNA is at the crux of DIPGs.
Starting Cancer
The histone modification that is relevant to DIPGs is the tri-methylation (me3) of histone H3 (H3) at its 27th amino acid, which is a lysine (K) (designated: H3K27me3). This modification is placed by a large protein complex named PRC2. This histone modification would normally give rise to compacted DNA and, thus, silenced genes. A notable publication showed that DIPG cells actually have a mutant H3 gene and that its mutation is present at exactly the K27 position where normal histone H3 would be trimethylated by PRC2.
An Important Sign
During his training as a biologist, Dr LeRoy became interested in whether changes in histones could explain the development of certain tumours. At the time Dr LeRoy was studying this process, he developed methods to measure how well the PCR2 complex performs its task of trimethylating histone H3. The discovery that DIPG cells contain histone H3 mutant at the position where PRC2 would normally modify the histone was very exciting. While all cells have multiple copies of histone H3 genes, all of which produce histone H3, the previous publication showed that the DIPG mutation in histone H3 occurred in only one of these genes, and yet, the DIPG cells are badly disrupted in their normal functioning.
Dr LeRoy and his supervisor, Professor Reinberg, recognised that this mutant histone H3 must somehow affect how PRC2 normally places this histone modification not only on the mutant H3 but also on the normal histone H3 in the cell. The mutant must somehow corrupt PRC2.
The Devil is in the Detail
As Dr LeRoy tells us, the way PRC2 works is conceptually quite simple. PRC2 binds to histone H3 protein, and once it is trimethylated by PRC2, one end of the PRC2 complex ‘reads’ this new modification, making PRC2 super-active such that its other end enthusiastically places more of the same modification on other histone H3 proteins. This straightforward process creates a long stretch of compacted and silenced DNA.
In carefully conducted experiments, Professor Reinberg and Dr LeRoy demonstrated that when the PRC2 complex binds to the abnormal histone H3 found in the DIPG tumour cells, they interacted too strongly, and this wound up inactivating PRC2 even after it got released from the mutant. Now, PRC2 was defective and could no longer trimethylate even the normal histone H3 in the cell. As a result, the DNA that should have been compacted was instead available, causing the cell to be abnormal. But instead of these abnormal cells dying, the inappropriate, available genes manage to keep the cells alive, but at the cost of becoming cancerous.
A Stoke of Good Luck
So, how does this incapacitation of PRC2 in DIPGs lead to inappropriate DNA becoming available? Dr LeRoy started to explore differences in the chemical modifications of the histone H3 proteins when cells have the mutant H3. The mutant cells responded to the absence of trimethylated histone H3 at appropriate genes by placing another version of histone H3 that was di- or trimethylated at a different site, H3K36me2/3.
Such H3K36me2/3 supports gene availability. The substitution of H3K27me3 that would normally compact the DNA now has H3K36me2/3, making the wrong DNA available. When he carefully examined DNA containing H3K36me2/3 in normal cells, he found that two proteins, LEDGF and HDGF2, bind to H3K36me2/3. These proteins normally help sustain the gene when H3K36me2/3 is at the proper DNA sites. But now, because H3K36me2/3 cropped up at DNA sites erroneously in DIPG cells, LEDGF and HDGF2 support the availability of the wrong DNA in DIPG cells, fostering their cancerous properties.
Dr LeRoy went on to reason that by lowering the levels of the LEDGF and HDGF2 proteins in DIPG cells, the cells would be relieved from inappropriate DNA being available. Indeed, when he depleted experimentally LEDGF and HDGF2 from DIPG cells in culture, the cells stopped dividing uncontrollably, and most died.
A Plan Emerges
Encouraged by their initial experiments, Professor Reinberg and Dr LeRoy developed a plan to further explore the role of LEDGF and HDGF2 in the growth of DIPG tumours. Included in this plan is NSD1. While PRC2 manages the levels of H3K27me3, NSD1 manages the levels of H3K36me2/3. So, inhibiting NSD1 and/or LEDGF2 and HDGF2 might eliminate the negative impact of H3K36me2/3 in DIPGs. In addition, the scientists want to understand which genes are no longer silenced because of the mutant histone in DIPGs.
The next step is to test if reducing the levels of one or more of these H3K36me2/3-related molecules, NSD1, LEDGF, and HDGF2, can inhibit the rampant growth of the majority of and potentially all DIPG tumours. As Dr LeRoy points out, the method used to deplete these molecules in cells involves intricate genetic approaches that are impossible to achieve in humans. Thus, Dr. LeRoy’s goal is to identify molecules/drugs that can inhibit combinations of these proteins (NSD1, LEDGF, and HDGF2) to ultimately treat children with this arsenal in hopes of killing off their DIPG intrusion.
The Ultimate Test
Sadly, not all interventions that target tumour cells under experimental conditions work when tested. Thus, as final proof, the team is now planning to test if compounds that inhibit NSD1, LEDGF, and HDFG2 will stop the growth of DIPG tumours in pre-clinical trials. Furthermore, if any tested potential drugs show promise, they intend to evaluate their side effects before clinical trials begin.
Of course, Professor Reinberg and Dr LeRoy are fully aware that this is an ambitious programme. Nonetheless, their encouraging early results give them hope that one day, they will be able to remove DIPGs from the list of the deadliest tumours in children. For the benefit of the affected children and their families, we sincerely hope that they will succeed.
SHARE
DOWNLOAD E-BOOK
REFERENCE
https://doi.org/10.33548/SCIENTIA1016
MEET THE RESEARCHERS

Professor Danny F. Reinberg
Sylvester Comprehensive Cancer Center at the University of Miami Miller School of Medicine
Miami, FL
USA
Professor Danny Reinberg studied at the Albert Einstein College of Medicine in New York, completing his PhD in 1982. He gained postdoctoral experience at the University of California Berkeley, the Albert Einstein College of Medicine, and Rockefeller University. After working at the State University of New York, Stony Brook, he moved to the Department of Biochemistry, University of Medicine and Dentistry of New Jersey. Professor Reinberg became an Investigator at the Howard Hughes Medical Institute. Following roles at the Robert Wood Johnson Medical School in New Jersey and New York University Langone School of Medicine, he was appointed Distinguished Professor in the Sylvester Comprehensive Cancer Center at the University of Miami Miller School of Medicine in 2023.
CONTACT
E: dxr1274@miami.edu
W: https://umiamihealth.org/sylvester-comprehensive-cancer-center/research/labs/reinberg-lab
X: @ReinbergDanny

Dr Gary T. LeRoy
Sylvester Comprehensive Cancer Center at the University of Miami Miller School of Medicine
Miami, FL
USA
Dr Gary LeRoy initially studied toxicology at Rutgers University. He then completed his PhD in biochemistry at Rutgers Medical School in 2000. After this, he gained extensive experience in biochemistry, molecular biology and proteomics as a postdoctoral research scientist in various laboratories at the Department of Molecular Biology, Princeton University between 2000 and 2012. He worked as a Senior Scientist at the State University of New York Medical School from 2013, where he investigated the role of the PRC2 complex and identified novel components of this complex and the role of the H3K27M histone mutation in paediatric brain tumours. In 2023, he relocated to the Sylvester Comprehensive Cancer Center at the University of Miami Miller School of Medicine.
CONTACT
E: leroygt@gmail.com
W: https://umiamihealth.org/sylvester-comprehensive-cancer-center/research/labs/reinberg-lab
CURRENT FUNDING
Making Headway Foundation
St. Baldrick’s Foundation
Hyundai Hope on Wheels
Alex’s Lemonade Stand Foundation
FURTHER READING
TM Escobar, A Loyola, D Reinberg, Parental nucleosome segregation and the inheritance of cellular identity, Nature Reviews Genetics, 2021, 22, 379–392. DOI: https://doi.org/10.1038/s41576-020-00312-w
J Yu, G LeRoy, D Bready, et al., The H3K36me2 writer-reader dependency in H3K27M-DIPG, Science Advances, 2021, 7(29), eabg7444. DOI: https://doi.org/10.1126/sciadv.abg7444
G LeRoy, O Oksuz, N Descostes, et al., LEDGF andHDGF2 relieve the nucleosome-induced barrier to transcription in differentiated cells, Science Advances, 2019, 5(10), eaay3068. DOI: https://doi.org/10.1126/sciadv.aay3068
M Stafford, C-H Lee, P Voigt, et al., Multiple modes of PRC2 inhibition elicit global chromatin alterations in H3K27M pediatric glioma, Science Advances, 2018, 4(10), eaau5935. DOI: https://doi.org/10.1126/sciadv.aau5935
O Oksuz, V Narendra, CH Lee, et al., Capturing the Onset of PRC2-Mediated Repressive Domain Formation, Molecular Cell, 2018, 70, 1149–1162. DOI: https://doi.org/10.1016/j.molcel.2018.05.023

REPUBLISH OUR ARTICLES
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence (CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
SUBSCRIBE NOW
Follow Us
MORE ARTICLES YOU MAY LIKE
How Food Environments Shape Our Eating Habits
How we eat dramatically impacts our health, yet millions of Americans live in ‘food deserts’ – areas with limited access to fresh, nutritious food. Recent research reveals that solving this crisis requires looking beyond just physical access to food to understand how our entire community environment shapes our dietary choices. Through a series of pioneering studies, Dr Terrence Thomas and colleagues at North Carolina A&T State University have been investigating how different aspects of our food environment influence what we put on our plates. Their findings suggest that creating lasting change requires reimagining how communities engage with food at every level.
Dr Jim Wu | Ziresovir Offers New Hope for Treating Respiratory Syncytial Virus Infections
Respiratory syncytial virus (RSV) causes respiratory tract infections in children and adults. While for many patients the outcomes of infection are mild, for others, infection can prove fatal, and there is a lack of effective treatments. Dr Jim Wu from the Shanghai Ark Biopharmaceutical Company in China carries out his vital research to develop new, safe, and effective treatments to tackle this killer.
Dr Sandra Grumelli | The Importance of the Choline in Chronic Lung Infections
People with chronic lung conditions like COPD and cystic fibrosis are vulnerable to lung infections caused by the bacterium Pseudomonas aeruginosa. These infections are often difficult to treat and can cause sudden worsening of symptoms, known as flare-ups or acute exacerbations. While we know P. aeruginosa triggers inflammation and damage in the lungs, much less is understood about how exactly it causes these flare-ups, or how it survives in such a harsh environment. Dr Sandra Grumelli from the Center of Investigations of Respiratory Diseases in Argentina, has explored the role of a common molecule called choline which is released during infection. Using a combination of mouse models and laboratory experiments, she has discovered that choline not only makes breathing harder, it also helps P. aeruginosa adapt to and persist in the lungs. Her research opens up new possibilities for tackling chronic infections by targeting the bacteria’s energy use and the way it responds to its environment.
Can Your Personality Shield Your Mind From Ageing? How being open to new experiences might protect against cognitive decline as we age
Many of us have witnessed the troubling effects of ageing on the mind in older friends or family members – the forgotten names, the misplaced keys, the struggle to solve problems that once seemed simple. For decades, scientists have accepted cognitive decline as an inevitable part of growing older. But what if our personality could protect us from some of these changes? A remarkable 25-year study by Dr David Sperbeck, a neuropsychologist at North Star Behavioral Health Hospital in Alaska, has uncovered compelling evidence that certain personality traits might act as a shield against age-related cognitive decline.





