Dr Youzhong Guo | Pioneering Understanding of Cell Membrane Components
Cell membranes are critical for cellular life. The effective extraction of proteins and lipids from cell membranes is a necessity for research, but traditional methods may damage the membrane components and limit the accuracy of data. Dr Youzhong Guo at Virginia Commonwealth University has recently developed a revolutionary method for the extraction of membrane components in the format of native cell membrane nanoparticles to allow in-depth structural studies of membrane proteins whilst preserving functionality and limiting damage to vital mechanisms. This exciting work is driving forward the understanding of the structure, function and protein-lipid interactions of membrane protein.
Membrane Proteins: Critical for Cellular Life
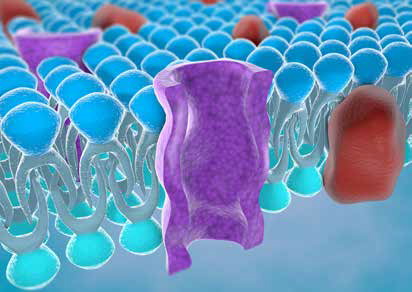
Cell membranes are an essential constituent of living organisms. Not only do cell membranes protect and organise cells, but they underpin a range of vital functions ensuring survival across species. For example, in humans, cell membranes in the brain are responsible for memory and consciousness, underscoring their importance to life itself. Given the critical role of cell membranes, it is unsurprising that research in this field is captivating to the scientific community and beyond, as evidenced by the history of Nobel prizes awarded to researchers elucidating the crucial and fascinating ways in which cell membranes work.
Research into the components of membranes, and the interactions between these components, provides critical insight into their structure and function. The two fundamental elements of cell membranes are proteins and lipids, which form a diverse and complex system connecting the membrane to the wider cell. Additionally, native lipid environments are vital in the maintenance of protein structure and function.
Traditionally, detergents have been used in the extraction of membrane proteins and lipids. However, these can induce structural damage and alter functionality which may hinder meaningful research. Protein-lipid interactions are critical in many biological systems, including targeted drug delivery and the development of vaccines – key concerns in medical science. Effective study of these interactions is dependent upon the presence of membrane proteins. The destruction of cell membranes by detergents may result in the removal of protein-associated lipid molecules, thus methods that successfully solubilise cell membrane proteins whilst retaining the lipid components are needed. Extraction of membrane proteins into lipoprotein particles using membrane-active polymers offers a possible substitute for detergent-based procedures and is now emerging as an important, viable alternative in the study of membrane protein function and structure.
Structure of plasma membrane of cell
Revolutionising the Extraction of Membrane Proteins in Native Lipid Environments
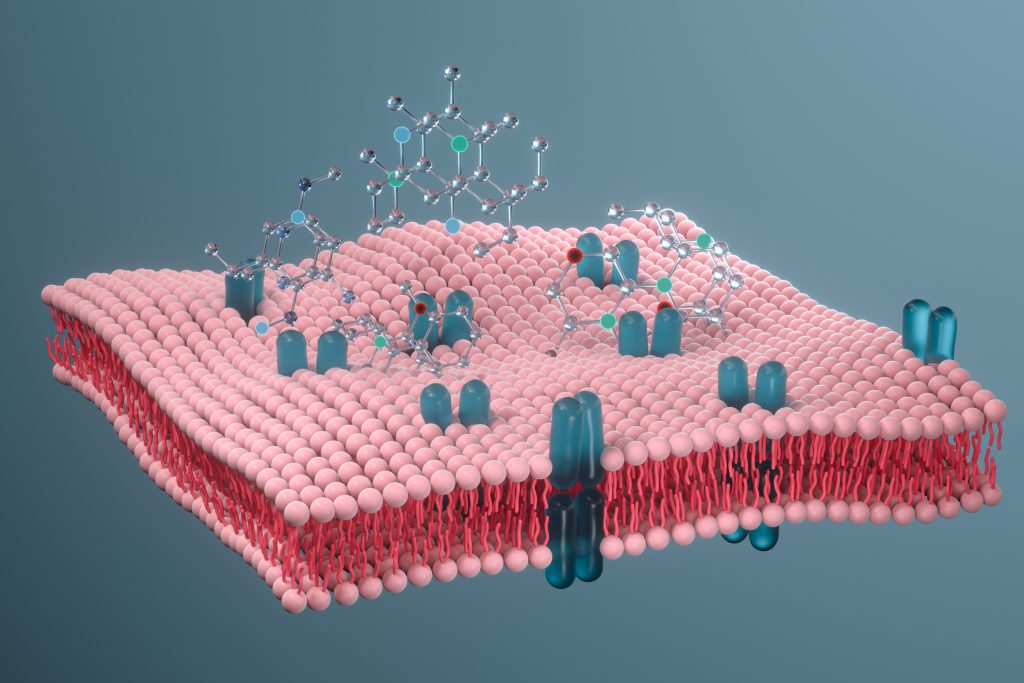
Recently, Dr Youzhong Guo and his team at Virginia Commonwealth University’s Department of Medicinal Chemistry in Richmond, USA, have revolutionised the extraction of cell membrane components using detergent-free processes and developed a unique technique to produce native cell membrane nanoparticles (NCMN).
In addition to the novel membrane co-polymer system invented by Dr Guo, a series of complementary linked libraries have been developed, comprising a comprehensive polymer library, a library of tailored preparation and extraction protocols, and a library of analysis protocols. This system has resulted in the production of detailed high-resolution structural models of various membrane proteins complex from bacteria, fungi, plants and animals and humans, contributing to the knowledge of protein-lipid interactions and allowing the overall functionality of cell membranes to be elucidated.
Discovery of a Structurally Preserved Lipid Bilayer
In order to accurately determine the structure of a membrane-embedded multidrug exporter, and to investigate its mechanism of active transport, Dr Guo and colleagues employed their ground-breaking detergent-free extraction protocol to prepare proteins for biochemical analysis, resulting in the discovery of a distinct lipid bilayer within the exporter structure.
Following extraction, the membrane proteins were purified and snap-frozen for analysis using electron microscopy. A 3D reconstruction of the resulting images revealed that each structural unit exists in one of three states, namely, ready for binding (loose), substrate-bound (tight) or substrate-released (open). Further analysis revealed that the transmembrane region was surrounded by a disordered lipid belt, and the central cavity of the structure contained an organised lipid bilayer with a hexagonal pattern and triangular double-layered shape. The layers were comprised of an inner and outer leaflet with distinct molecular patterns; the inner leaflet contained tightly packed lipid molecules with straight tails, whereas those in the outer leaflet were more loosely packed with curvier tails. Furthermore, several specific protein-lipid interactions were identified within the central cavity, including via protrusions and bonds.
It is recognised that, as proteins undergo conformational changes, lipid bilayers can adapt due to their fluid nature. Dr Guo proposed that the central lipid bilayer structure has an important role in the mechanism of action of multidrug exporters by acting as a mediator of these conformational changes and promoting drug extrusion via the transmembrane transporter, as well as providing structural support. The optimal environment for membrane proteins is most certainly within the native membrane, and the completely detergent-free extraction method developed by Dr Guo does appear to facilitate the preservation of this environment, which confers several advantages over other methods.

Characterisation of Membrane Protein Channels and Assembly of Nanodevices
A particularly challenging aspect of membrane protein research includes the characterisation and reconstitution of integral components such as channels and transporters, especially when protein-degrading detergents are used in the process of extraction. Regardless of whether the membrane protein structure is maintained, functionality is often compromised, and reconstitution of the proteins into proteoliposomes is required if functional characteristics are to be determined. Proteoliposomes mimic the cell membrane environment, which not only allows the study of membrane protein structure and function, but also analysis of the mechanisms of drug delivery devices.
Using the previously described NCMN system, Dr Guo and colleagues studied a number of integrated membrane protein channels which aid in the rapid expulsion of molecules from within the cell. The researchers found that NCMN particles can be used to directly reconstitute the channels into liposomes, suggesting that this method may be a feasible option for the routine production of functional channel proteins for use in membrane research.
Channels were constructed to optimise protein expression and function, transformed into cells of interest, and grown in culture, followed by induction of protein expression, further culture and cell harvesting. For the preparation of nanoparticles, the resulting cell membrane proteins were mixed with NCMN buffer and NCMN polymer to achieve a pre-determined protein concentration. Following purification of the sample, analysis using electron microscopy and reconstitution into proteoliposomes was possible.
For electron microscopy image acquisition, an aliquot of sample was absorbed onto a copper grid, followed by a series of drying and washing steps. Images were captured and recorded using a camera attached to the microscope. Analysis of the images confirmed the high quality of the resulting nanoparticles. The reconstitution of proteins using NCMN polymers commenced with the drying of the lipid solution, followed by rehydration of the lipids in specific buffers or sucrose solution to form liposomes. NCMN protein particles were then added for reconstitution according to experimental requirements. The reconstitution of functional channels using rehydration with buffers was successful with some polymers, reinforcing the notion that NCMN particles are a viable option for the study of reconstituted membrane channels. Additionally, in contrast to detergent-based methods, channel particles remained stable when refrigerated and retained functional viability for several months.
Next, Dr Guo and his team proceeded to determine whether modified reconstituted channels might be assembled into functional nanodevices which represent gated membrane-embedded valves with the ability to deliver specific substances into cells residing in low-pH environments, such as within inflamed or malignant tissue. The simplified reconstitution protocol described above was utilised, with the addition of a fluorescent dye to the mixture. The fluorescent signal of the solution was monitored for 5 minutes, the pH was lowered, and the fluorescent signal monitored for a further 30 minutes. The release of dye from the nanodevices was confirmed with an 80% increase in fluorescent signal over the monitoring period, indicating that functionality was indeed preserved following assembly. This has the potential to significantly advance the development of bespoke drug delivery nanodevices.
A Comparison of Membrane Protein Extraction Systems
Recently, Dr Guo conducted a concise but thorough mini-review evaluating a selection of the available methods for the detergent-based and detergent-free extraction of membrane components. A comparison of the techniques with regard to the extent of protein and lipid survival and successful reconstruction was made. Interestingly, the most commonly used detergent-free extraction methods did not fare better than detergent-based methods regarding the preservation of native cell membrane lipids, or when determining membrane structure. In contrast, the NCMN system developed by Dr Guo and his team demonstrated superior preservation of native cell membrane lipids associated with the membrane proteins and produced particles of sufficient quality to perform high-resolution structural analysis.
Building on Success: The Wider Context
Detergents have indisputably contributed to advances in the study of membrane proteins. However, the associated destruction of the lipid bilayer may have implications for both structural and functional analysis. Research by Dr Guo revealing the structure of the lipid bilayer within a multidrug exporter and its interaction with the surrounding environment has driven forward the understanding of mechanisms of transport across cell membranes, while the continual expansion of the NCMN polymer library has enabled many more new membrane proteins and complexes to be investigated and has permitted the unique properties of a wide variety of membrane proteins to be accommodated. Using the NCMN system in the reconstitution of proteoliposomes is a relatively straightforward process and applicable to a variety of integral membrane constituents, representing a powerful new tool in nanobiotechnology.
Detergent-free extraction methods confer significant advantages over detergent-based methods in terms of maintaining stability during longer-term storage and retention of functionality upon reconstitution. Whilst the implementation of detergent-free protocols for extracting cell membrane proteins and lipids is still relatively recent, there is much potential surrounding the development and application of the methods employed.
The research carried out by Dr Guo and his team is revolutionising the understanding of membrane protein biology and there is plenty more to come. Dr Guo has a plethora of manuscripts awaiting publication detailing the ongoing work of his team. This exciting area of biological research may be in its infancy but holds much promise for the future of cell membrane studies. Dr Guo and his laboratory will undoubtedly be at the forefront with their pioneering technologies and insights.
SHARE
DOWNLOAD E-BOOK
REFERENCE
https://doi.org/10.33548/SCIENTIA880
MEET THE RESEARCHER

Dr Youzhong Guo
Department of Medicinal Chemistry
Institute for Structural Biology, Drug Discovery and Development
School of Pharmacy
Virginia Commonwealth University
Richmond, VA
USA
Dr Youzhong Guo is an Assistant Professor and also a member of the Institute for Structure Biology, Drug Discovery and Development at Virginia Commonwealth University. Having been awarded his PhD by the University of Texas at Austin in 2010, Dr Guo took up a postdoctoral position at Columbia University before accepting his current position. Dr Guo has authored and co-authored more than 20 published papers, several of which appear in highly acclaimed peer-reviewed journals, including Science, PNAS and Nature Communications. As an expert in the structure biological of membrane proteins, Dr Guo’s ground-breaking work in the development of novel native cell membrane nanoparticles system has resulted in patented technologies, with several more pending. He is the recipient of several high-profile project grants and has presented his research at conferences all over the world. In addition to his extensive teaching and mentoring duties at Virginia Commonwealth University, Dr Guo is the organiser of the International SMALP Conference and Co-Director of SMALP.NET. He is also a founder of NCMN Bio.
CONTACT
W: https://www.ncbi.nlm.nih.gov/myncbi/1fMGq6ZwVF6Q7/bibliography/public/
KEY COLLABORATORS
Dr Wayne A. Hendrickson, Columbia University, USA
Dr Joachim Frank, Columbia University, USA
Dr Ann E. MacDermott, Columbia University, USA
Dr Paul Blount, UT Southwestern Medical Center, USA
Dr Mark Yeager, University of Miami, USA
Dr Amedee des Georges, City College New York, USA
FUNDING
National Institute of General Medical Sciences (R01GM132329)
Virginia Commonwealth University
FURTHER READING
W Qiu, Y Guo, Analysis of the oligomeric state of mycobacterial membrane protein large 3 and its interaction with SQ109 with native cell membrane nanoparticles system, Biochimica et biophysica acta. Biomembranes, 2022, 1864(1), 183793. DOI: https://doi.org/10.1016/j.bbamem.2021.183793
C Catalano, D Ben-Hail, W Qiu, et al., Cryo-EM Structure of Mechanosensitive Channel YnaI Using SMA2000: Challenges and Opportunities, Membranes, 2021, 11, 849. DOI: https://doi.org/10.3390/membranes11110849
Y Guo, Detergent-free systems for structural studies of membrane proteins, Biochemical Society Transactions, 2021, 49, 1361–1374. DOI: https://doi.org/10.1042/BST20201080
L Yang, C Catalano, Y Xu, et al., A native cell membrane nanoparticles system allows for high-quality functional proteoliposome reconstitution, BBA Advances, 2021, 1, 100011. DOI: https://doi.org/10.1016/j.bbadva.2021.100011
KG Kroeck, W Qiu, C Catalano, et al., Native Cell Membrane Nanoparticles System for Membrane Protein-Protein Interaction Analysis, Journal of Visualized Experiments, 2020, 161, e61298. DOI: https://doi.org/10.3791/61298
W Qiu, Z Fu, GG Xu, et al., Structure and activity of lipid bilayer within a membrane-protein transporter, Proceedings of the National Academy of Sciences of the United States of America, 2018, 115(51), 12985–12990. DOI: https://doi.org/10.1073/pnas.1812526115

REPUBLISH OUR ARTICLES
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence (CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
SUBSCRIBE NOW
Follow Us
MORE ARTICLES YOU MAY LIKE
Dr JoLee Sasakamoose – Dr Mamata Pandey | Empowering Indigenous Health: The Indigenous Wellness Research Collaborative in Saskatchewan
The Indigenous Wellness Research Collaborative is a transformative alliance dedicated to advancing health systems and service delivery for Indigenous communities across Saskatchewan. Founded a decade ago and co-led by Dr Mamata Pandey, a research scientist at the Saskatchewan Health Authority, and Dr JoLee Sasakamoose (M’Chigeeng First Nation), Canadian Institute of Health Research Chair in Indigenous Wellness and Health Equity at the University of Regina, their team’s work is rooted in a commitment to Indigenous leadership and community-defined wellness goals. Guided by the Cultural Responsiveness Framework, the Collaborative prioritises creating ethical spaces that serve as a middle ground for respect, reciprocity, and authentic partnerships. The team employs a strengths-based approach to health research, centering Indigenous methodologies that respect the interconnectedness of spiritual, mental, emotional, and physical well-being.
Professor Jaya Krishnan | Revolutionary Gene Therapy Helps Hearts Regenerate After Heart Attacks
Myocardial infarction, commonly termed as a heart attack, is a major cause of death and poor health worldwide. Regenerating heart tissue is an exciting and promising concept that can have significant benefits in myocardial infarctions and related diseases, but this has not yet been achieved in real-life clinical treatments. In a collaboration between Goethe University Frankfurt and Goethe University Hospital, Professor Jaya Krishnan and colleagues address this by controlling pathologic genes involved in the development of heart failure that develops after heart attacks. The researchers demonstrate a new way of treating heart disease by aiding in the division and regrowth of heart cells after a heart attack.
James J. Driscoll, MD, PhD | Immunoproteasome Activation Enhances the Recognition of Tumour Cells and Boosts Anticancer Immune Responses
The correct functioning of the human immune system depends on its ability to recognise danger, such as tumour cells, viruses, and bacteria. Scientists are learning how immunoproteasome activation can overcome the mechanisms by which cancer cells escape immune responses. Immunoproteasomes are small high molecular weight protein-degrading machines that signpost abnormal proteins made by cancer cells, directing the immune system against them. Dr James Driscoll at University Hospitals Cleveland Medical Center is using novel proprietary small molecules to selectively boost the catalytic activity of immunoproteasomes, which increases the tumour killing (or cytotoxic) effect of a group of white cells called T-cells. These findings provide a strong rationale for developing personalised therapeutics that target immunoproteasomes, for cancer and other immune-mediated conditions.
New Approaches to Treating Alzheimer’s Disease
Alzheimer’s disease is a devastating condition that strips away people’s memory, thinking, and independence. By 2050, it is expected to affect over 100 million people around the world, making it a high priority for scientific and medical research. Researchers are now exploring the potential for mechanical and light-based stimulation of the brain and nervous system to treat Alzheimer’s disease symptoms. At the University of Minho in Portugal, Francisca Monteiro is developing a PhD project supervised by a multidisciplinary set of experienced researchers, who have reviewed the evidence behind these approaches, including whole-body vibration, auditory stimulation, transcranial ultrasound stimulation, and photobiomodulation. The team aims to synthesize the evidence to support these treatments and understand what further work is needed.