Dr Delphine Muriaux | Shining New Light on Human Immunodeficiency Virus Assembly Mechanisms
People living with human immunodeficiency virus (HIV) now have very effective treatment options to allow them to live long lives but the need for new and improved therapeutics remains. Dr Delphine Muriaux from Le Centre national de la recherche scientifique (CNRS) in Montpellier, France, researches HIV infection and replication utilising advanced state-of-the-art microscopy. This super-resolution imaging has led to new findings on the importance of the HIV-1 Gag proteins and the cellular host co-factor IRSp53, a membrane curving protein, and how they interact with host cell membranes.
Human Immunodeficiency Virus: Risks and Replication
Human immunodeficiency virus (HIV) is a pathogen that infects and destroys cells in the immune system. Because it is found in certain bodily fluids, HIV can be passed on through sex, sharing needles and pregnancy or breastfeeding. Initially, an infection may present with flu-like symptoms for a couple of weeks and then seemingly disappear for years. However, if left untreated, HIV eventually leads to acquired immune deficiency syndrome (AIDS), where the immune system becomes increasingly damaged and minor additional infections can prove severe or even fatal.
Diagnosing HIV can be achieved through a blood or saliva test and fortunately, there are now very effective treatments for those who are HIV positive. Antiretroviral drugs are taken every day to prevent the virus from replicating so that the immune system can repair itself. Thanks to these lifesaving therapeutics and other interventions, new HIV infections have been dropping since 1997, but there are still an estimated 37.7 million people living with the virus worldwide.
In order to continue to improve available therapeutics and quality of care for HIV patients, it is important to study the virus and all its complex activities. Dr Delphine Muriaux has built a ‘Membrane Domains and Viral Domains’ team of dedicated scientists to do just this at the Institut de Recherche en Infectiologie de Montpellier (IRIM). This institute sits within Le Centre national de la recherche scientifique (CNRS) in Montpellier, France.
The mechanisms by which HIV infects cells are extensively researched, but there remains much to discover about its molecular activities. The virus invades immune cells called CD4+ T helper cells, which are a type of white blood cell that help to activate other aspects of the immune system as well as suppress immune reactions where necessary. HIV can also infect types of white blood cells called macrophages and dendritic cells.
To enter a target host cell, a glycoprotein on the HIV particle binds to a receptor on the CD4+ cell. These two structures have a strong affinity (attraction) to each other. Once attached, a viral nucleocapsid containing its genetic material and certain enzymes enters the cell. These enzymes including reverse transcriptase, protease and viral integrase, are used to synthesise the new genome and integrate viral DNA into the host genome. From there, the host’s own DNA replication machinery is used to produce HIV proteins to be transported to the cytoplasm and assembled at the inner leaflet of the plasma membrane. The new HIV particle then assembles at the membrane and buds off to repeat the process, killing the host immune cell. This is why a patient’s immune system can be devastated by an uncontrolled HIV infection.
Shining New Light on Viral Assembly
Although this process of viral assembly has been understood for decades, the timeframe in which it occurs has only more recently been laid out. This is thanks to advancements in technology that allow scientists to observe biological activities on a molecular level, in real-time. Previous techniques were limited by the diffraction of light, which minimises the resolution possibilities for a microscope image. The diffraction barrier prevents a microscope from distinguishing between two objects that are less than half the wavelength of light used apart from each other. These objects can be singular molecules, which means that the molecular details of an HIV assembling within a host cell could not be viewed.
New solutions have overcome this barrier to enable super-resolution imaging of endless biological occurrences and structures. These visualisations often utilise microscopic molecules called fluorophores or fluorescent tags, which absorb specific wavelengths of light and emit a longer wavelength which is seen as colour. They can be designed to attach to specific biological structures so that different structures can be clearly observed in different colours. Super-resolution microscopy either involves selective deactivation of fluorophores or single-molecule detection and localisation.
In addition to the improvements in fluorescent tags, super-resolution microscopy has assisted new research from Dr Muriaux. After co-authoring a review on these advanced techniques and the results they have facilitated, she went on to utilise them in her own studies of the assembly of HIV in CD4 T cell plasma membrane. She believes that elucidating invasion and replication mechanisms on a never-before-seen molecular level will allow the entire infection process to be seen in a new light.

Dr Muriaux in the bioSafety level 3 laboratory at CEMIPAI CNRS Montpellier. Credit. Delphine Muriaux
Using Super-resolution Microscopy
One important aspect of HIV infection that Dr Muriaux could see in more detail was the activity of a protein called Gag. It is the main structural protein of HIV and other retroviruses – responsible for the assembly of viral particles. When HIV particles have replicated and budded from the host cell, the composition of their outer lipid membrane is very different from the plasma membrane of the host cell. It contains a notably higher concentration of types of phospholipids called phosphoinositides, including one major called phosphatidylinositol 4,5-bisphosphate lipid, or PI(4,5)P2 for short, as well as some cholesterol. The lipids (fats) in the HIV membrane are also highly ordered and densely packed.
These unusual differences led Dr Muriaux to question how the structures of an HIV particle such as Gag interact with a host plasma membrane to create such a specific environment. Is Gag targeted at existing lipid domains in the plasma membrane or does it somehow create these lipid compositions to help new viruses form? Dr Muriaux, with the help of Dr Cyril Favard from CNRS in Montpellier, France, and Drs Jakub Chojnacki and Christian Eggeling from the University of Oxford (UK), investigated these questions using super-resolution microscopy in living CD4+ T cells that were either infected with HIV or were not infected but were still expressing Gag. To visualise the movement of the lipids, they were tagged with fluorophores, as were the Gag proteins.
Tracking these different molecules in HIV-infected cells revealed that the virus immobilises PI(4,5)P2 and cholesterol but does allow sphingomyelins to move throughout the host cell. The cells that were not infected but expressed Gag showed the same characteristics, restricting the movement of the lipids at HIV assembly sites. This means that Dr Muriaux, her team and collaborators demonstrated that HIV-1 utilises its Gag proteins to selectively trap PI(4,5)P2 and cholesterol in host cells to produce their own specialised lipid membrane for particle assembly.
Illustration of HIV in blood
Facilitating Human Immunodeficiency Virus Budding
Dr Muriaux continued to investigate HIV Gag and plasma membrane deformation in a subsequent study. This time, she and her colleagues focused on membrane curvature, which is the bending of the membrane to accommodate the shape of organelles or biological processes. These can include endocytosis (taking an outside particle into a cell) and vesiculation (exiting the cell via formation, integration with the membrane and release of a vesicle). These mechanisms are important for HIV assembly and release. Previously, scientists thought that Gag was the sole driver of plasma membrane curvature for HIV particle formation, but Dr Muriaux has revealed that there is a more detailed mechanism.
She studied an I-BAR protein, IRSp53, which binds to membranes in a PI(4,5)P2-dependent manner with high affinity to generate a negative membrane curvature, meaning the membrane protrudes outwards. She chose IRSp53 due to her previous observations that it is implicated in a signalling pathway that promotes the release of HIV particles. These factors made it a prime candidate for being important in HIV Gag assembly and viral bud formation. She utilised single-molecule localisation microscopy to observe IRSp53 and its activities within cells.
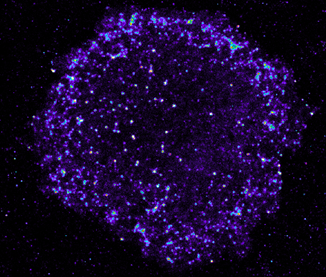
T cells infected by HIV. Each dot is an HIV assembly platform at the cell surface of the infected CD4+ T lymphocytes. Each dot surrounding the cell is a single virus. Credit. MDVA research team, IRIM
Montpellier, France.
The results from these observations revealed that IRSp53 forms a complex (a bound structure) with Gag within cells, next to the cell plasma membrane. Therefore, through its membrane-bending abilities and interactions with Gag, IRSp53 is an essential factor for optimal HIV particle formation and replication. Proper and efficient HIV membrane curvature and complete assembly at the cell plasma membrane require IRSp53 and Gag to work together.
The importance of IRSp53 in viral budding was also demonstrated in a test whereby the gene that produces the IRSp53 protein was knocked down, meaning much less of the protein is produced. This resulted in a decrease in overall viral particle production and HIV budding was stopped halfway through the process. By discovering and documenting the replication mechanisms of HIV, scientists can better understand where and how to target them with drugs to prevent the virus from reproducing.
The research of Dr Muriaux, with her team at CNRS and collaboration with national and international academia laboratories as well as biotechnology companies, has allowed us to understand HIV infection in a new, much more detailed way. Her work is on track to inform the development of novel therapeutics for HIV and also pandemic viruses such as influenza viruses and coronaviruses.
SHARE
DOWNLOAD E-BOOK
LISTEN TO THE AUDIO
REFERENCE
https://doi.org/10.33548/SCIENTIA855
MEET THE RESEARCHER

Dr Delphine Muriaux
Institut de Recherche en Infectiologie de Montpellier (IRIM)
Le Centre national de la recherche scientifique (CNRS)
Montpellier
France
Dr Delphine Muriaux completed her PhD in molecular and cellular biology from Pierre et Marie Curie University in Paris before postdoctoral training in virology at the National Cancer Institute (National Institutes of Health) in the USA. As a Research Director, she went on to establish a team at the Institut de Recherche en Infectiologie de Montpellier (IRIM) within Le Centre national de la recherche scientifique (CNRS) in France. Dr Muriaux and her colleagues investigate membrane domains and viral assembly, focusing on the molecular and cellular mechanisms of virus replication. She is also the head of the CEMIPAI laboratory which assists academics and private companies in antiviral drug screening and nano-biological object detection and characterisation using super-resolution microscopes in a level 3 biosafety laboratory. She also trains students by providing workshops in scientific national schools and mentoring PhD students from the University of Montpellier Woman in Science Mentoring programme.
CONTACT
E: delphine.muriaux@irim.cnrs.fr
W: https://www.irim.cnrs.fr/index.php/recherche/equipes/domaines-membranaires-et-assemblage-viral and https://www.cemipai.fr
Twitter: @MuriauxD @VirAssemblyLab
KEY COLLABORATORS
Dr Cyril Favard, IRIM CNRS Montpellier France
Dr Jakub Chojnacki, University of Oxford, Oxford, UK (now at IrsiCaixa, Barcelona, Spain)
Prof Dr Christian Eggeling, University of Oxford, UK (now at Jena University, Germany)
Dr Patricia Bassereau, Institut Curie, Paris, France
Prof Dr Jonhson Mak, Griffith University, Australia
Prof Philippe Roingeard, Tours University, France
FUNDING
ANRS (Agence national de recherche sur le SIDA)
Sidaction
CNRS
ANR (Agence National de Recherche)
FURTHER READING
K Inamdar, F-C Tsai, R Dibsy, et al., Full assembly of HIV-1 particles requires assistance of the membrane curvature factor IRSp53, eLife, 2021, 10, e67321. DOI: https://doi.org/10.7554/eLife.67321
C Favard, J Chojnacki, P Merida, et al., HIV-1 Gag specifically restricts PI(4,5)P2 and cholesterol mobility in living cells creating a nanodomain platform for virus assembly, Science Advances, 2019, 5(10), eaaw8651. DOI: https://doi.org/10.1126/sciadv.aaw8651
K Inamdar, C Floderer, C Favard, D Muriaux, Monitoring HIV-1 Assembly in Living Cells: Insights from Dynamic and Single Molecule Microscopy, Viruses, 2019, 11(1), 72. DOI: https://doi.org/10.3390/v11010072
A Thomas, C Mariani-Floderer, MR López-Huertas, et al., Involvement of the Rac1-IRSp53-Wave2-Arp2/3 Signaling Pathway in HIV-1 Gag Particle Release in CD4 T Cells, Journal of Virology, 2015, 89(16), 8162–8181. DOI: https://doi.org/10.1128/JVI.00469-15


REPUBLISH OUR ARTICLES
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence (CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
SUBSCRIBE NOW
Follow Us
MORE ARTICLES YOU MAY LIKE
Stroke: A Global Challenge in Need of Innovation
Stroke is the third leading cause of both death and disability worldwide, but unfortunately, current diagnostic tools are unsatisfactory. Dr Roustem Miftahof and Dr Alexander Hermann at Advanced Biosimulation Technologies LLC, USA, are overcoming the limitations of existing diagnostic tools with the development of the Neuro-Glia-Vascular Unit Engine. This transformative tool for clinicians and biomedical researchers provides patient-specific health insights with the potential to improve care not only in stroke but across a range of neurological disorders.
Dr Alyson J McGregor | Uncovering the Gender Gap in Medical Research: How Sex Differences Impact Healthcare Outcomes
Medical research has historically focused predominantly on male subjects, leading to dangerous gaps in our understanding of how diseases and treatments affect women. Dr Alyson McGregor from University of South Carolina School of Medicine Greenville has devoted her career to addressing this critical issue, highlighting how biological sex differences impact health outcomes. Her work demonstrates that ignoring these differences can have life-threatening consequences and advocates for more inclusive research practices to ensure safe and effective healthcare for everyone.
How Food Environments Shape Our Eating Habits
How we eat dramatically impacts our health, yet millions of Americans live in ‘food deserts’ – areas with limited access to fresh, nutritious food. Recent research reveals that solving this crisis requires looking beyond just physical access to food to understand how our entire community environment shapes our dietary choices. Through a series of pioneering studies, Dr Terrence Thomas and colleagues at North Carolina A&T State University have been investigating how different aspects of our food environment influence what we put on our plates. Their findings suggest that creating lasting change requires reimagining how communities engage with food at every level.
Dr Jim Wu | Ziresovir Offers New Hope for Treating Respiratory Syncytial Virus Infections
Respiratory syncytial virus (RSV) causes respiratory tract infections in children and adults. While for many patients the outcomes of infection are mild, for others, infection can prove fatal, and there is a lack of effective treatments. Dr Jim Wu from the Shanghai Ark Biopharmaceutical Company in China carries out his vital research to develop new, safe, and effective treatments to tackle this killer.