PrognostX Health | A New Test to Identify Chronic Kidney Disease – A Costly and Silent Killer
Chronic kidney disease is a common but irreversible condition with an increasing worldwide prevalence. The significant patient morbidity and mortality are accompanied by an unmet clinical need for more effective testing methods to identify affected patients and patients at high risk at the early stages of the disease, before it becomes irreversible. Dr. Aaron Carrithers and Dr. Stephen Carrithers at PrognostX Health have developed a new test to help reduce the number of patients progressing to late-stage chronic kidney disease and end-stage renal disease, aiming to improve human health and reduce the financial burdens on our pressured healthcare systems.
Chronic Kidney Disease: A Global Epidemic
Your kidneys help to remove waste products from the body, maintain balanced electrolyte levels, and regulate blood pressure. Chronic kidney disease (CKD) is a condition characterised by a gradual loss of kidney function. One way of assessing this is to calculate the ‘eGFR’ which is the estimated glomerular filtration rate of the kidneys. This is based on a patient’s serum creatinine level (which shows how well your body is performing its job of filtering waste from the blood) and currently requires the patient’s age, sex and race.
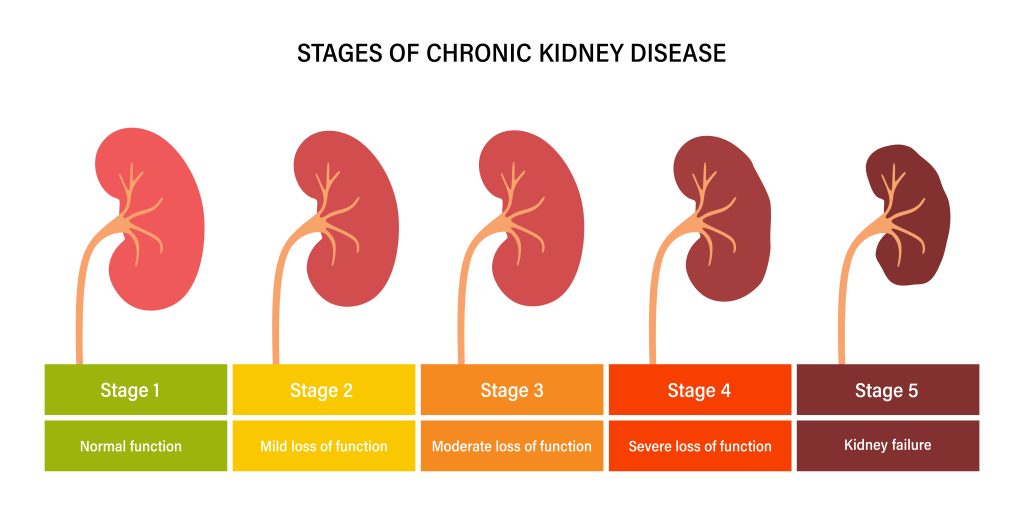
Using eGFR, a stage can be assigned to reflect the severity of disease in patients. Stage 1 indicates that the damage to kidneys is very mild, whereas Stage 5 is assigned to patients whose kidneys have lost almost all of their function and typically requires life-sustaining dialysis treatments three times per week. This is known as end-stage renal disease (ESRD), which leads to many more added medical complications, increased expenses to both the patient and the healthcare system, decreased quality of life, and a high likelihood of early death. However, Stage 3 CKD is considered the point at which the disease is irreversible and due to the subtle and underlying symptoms associated with this disease stage, over 90% of individuals are unaware of their chronicity. Thus, progression to dialysis and ESRD is likely imminent without early intervention.
Kidney disease is currently recognised as a global epidemic affecting nearly 850 million people worldwide, and approximately 700 million of these patients have CKD. Many are undiagnosed, untreated, and unaware of their irreversible condition. As such, CKD is associated with significant patient morbidity and mortality, placing a huge financial burden on already struggling healthcare systems.
Hypertension, diabetes and acute kidney injury (including patients with kidney conditions resulting from COVID-19 infection) are the major predisposing conditions for developing CKD, accounting for more than three in every four new cases. Adverse outcomes include an increased risk of developing cardiovascular and cardiorenal disease, Alzheimer’s disease, depression, and progression to end-stage renal disease, leading the patient to require 2x–3x weekly dialysis treatment and/or kidney transplantation.

A Silent Killer
CKD is clinically diagnosed in about one in seven people – but many people are unaware that they have it. Among adults with CKD, the percentage aware of their disease state was fewer than 10% for patients in Stage 3 and fewer than 60% in Stage 4. CKD is therefore often referred to as a ‘silent killer’, being generally asymptomatic until very the late stages of illness.
This means that there are large numbers of people with later-stage CKD who remain undiagnosed and untreated, resulting in a considerably shorter lifespan than if it had been detected earlier. Poor rates in diagnostic efficacy and low patient awareness of CKD are secondary to the inherent limitations of the current diagnostic standard of care. Indeed, current methods require multiple tests and take no less than three months to even diagnose a patient with CKD, and for many patients, the process could take one year. Furthermore, due to this untimely diagnostic method, about 60% of patients are lost to follow-up, which contributes to the rising incidence of dialysis and ESRD. Despite efforts made to raise diagnostic efficiency and patient awareness, substantial improvements have yet to be seen.
Novel and Effective Interventions: Need for Timely Diagnosis
Novel medications have been shown to effectively slow CKD progression and help prevent end-stage renal failure, and have received both U.S. (Food and Drug Administration) and European approval for patient use. These critical advances point to the urgent need for accurate and early diagnostic tests if we are to identify patients with the disease and thus ensure timely treatment. This is especially critical during the early stages of CKD among high-risk groups, such as those with type 2 diabetes, when the disease is often asymptomatic.
To address this need, researchers Drs. Stephen Carrithers and Aaron Carrithers at PrognostX Health in the USA have developed a new, simple and specific assay that detects early CKD with just one single test in high-risk patients.
Current Diagnostic Standard of Care and Associated Challenges
The current diagnostic standard of care is complicated, not least because it requires at least 90 to 150 days of follow-up visits to the clinic and the repeat laboratory testing of a patient’s samples. Patients who attend follow-up appointments for confirmation of diagnosis are at an unprecedented low, meaning that many are remaining undiagnosed.
The current laboratory tests are not suitably specific in identifying patients with CKD, and so many patients are missed in the first round of testing. For example, the laboratory reporting of eGFR alone does not result in optimal clinical diagnoses. Another type of test can be used to assess glomerular integrity, but is only helpful for picking up the very early stages of CKD in some patients but must be re-evaluated on a routine basis for confirmation of diagnosis.
Current tests are also unable to accurately discern acute kidney disease patients from those with chronic kidney conditions. This means that patients are only being diagnosed at a much later stage after multiple rounds of testing when the disease has progressed further and has reached a more advanced stage. This ultimately results in a larger number of patients who have progressed to dialysis and end-stage renal disease. It is also currently difficult to identify high-risk patients, such as those with diabetes or hypertension who might have developed early-stage CKD but are unaware of their condition.
A New Approach: ConfirmCKD!
The new test – ‘ConfirmCKD!’ – was developed by Drs. Carrithers and Carrithers. It is a simple type of immunoassay test which can be performed quickly and easily in the laboratory using a blood sample obtained from a patient. The test itself takes advantage of specific biomarkers that circulate in the bloodstream in the very early stages of CKD – before a patient or high-risk individual has any symptoms and is still in a stage in which the disease is most likely reversible. Biomarkers are molecules that indicate normal or abnormal processes taking place in your body and elevated levels of a particular biomarker can be a sign of an underlying condition or disease. In this case, the test looks for the presence of a particular biomarker, the gene product(s) expressed by renal Guca2b.
These gene products, specifically the C-terminal region of the expressed and secreted prohormone from Guca2b, have been shown to be involved in the physiological regulation of salt levels in the body. The mature Guca2b gene product levels in the blood are normally low in humans but when certain kidney-related diseases or mechanisms of chronic kidney dysfunction occur in the body, the level of this biomarker can increase significantly, prior to losing significant function of the kidney. This rise can therefore be detected and used to identify patients who have CKD.
Drs. Aaron and Stephen Carrithers have undertaken validation of the test and determined the cut-off value for diagnosing CKD in a patient. In this way, a rise in the serum or blood can be used as a simple and effective way to diagnose and provide treatment in early CKD, and also to monitor the disease progression of kidney dysfunction in a particular patient. Furthermore, the test can be used to predict the outcome of a treatment program for kidney disease and/or dysfunction without interference from other common diseases or cancers that are generally associated with CKD patients.
This new test has been demonstrated to be convenient, quick and easy to use, and can be readily incorporated into any hospital or testing laboratory facility using the equipment that is typically available. Another advantage of this immunoassay is that it can be converted easily into other test formats, such as a lateral flow device, which could potentially be used by patients in their homes.

Aaron, Stephen and Brennan (from left to right)
Gathering Vital Clinical Evidence
The initial and adjudicated validation studies conducted by Drs. Carrithers and Carrithers in 384 patients demonstrated a remarkable accuracy of the new assay in correctly identifying those who had CKD from just one test. Stages 3 to 5 of CKD were diagnosed and staged with a higher than 97% accuracy from patients whose renal function was within normal limits.
Testing produced very few false positives (in which a test incorrectly indicates the presence of a disease) and even fewer false negatives (in which a test result incorrectly indicates the absence of a disease when it is present). The assay has several significant advantages over the current standard-of-care protocols, in particular, the removal of the need for patients to undergo months of repeat testing. Furthermore, the researchers demonstrated that the results of their new assay were not affected by other concurrent pathologies or medications, nor by confounding patient variables such as age, gender or race.
ConfirmCKD! has undergone further development, and now functions as a simple and effective qualitative ‘positive/negative’, ‘treat/no treat’ result, with an accuracy of over 97% for single-assessment identification of eGFR-defined CKD (i.e., patients with stages 3a, 3b, and 4). Drs. Aaron and Stephen Carrithers have adapted their testing approach to work most effectively in tandem with the recently approved pharmaceutical agents that are indicated for the treatment of stages 3a, 3b, and 4 CKD. The combination of novel pharmaceutical agents along with the application of better testing methods promises to lead to a substantial improvement in the detection and treatment of CKD.
Implications and Important Benefits
This simple-to-use and effective test offers the potential to reduce the worldwide burden of CKD and overcome the significant difficulties associated with the current standard of care approach. By helping to identify patients who are at risk of developing ESRD, the individual cost to patients and their families should be considerably reduced, as well as the financial burdens on health systems worldwide.
SHARE
DOWNLOAD E-BOOK
REFERENCE
https://doi.org/10.33548/SCIENTIA949
MEET THE RESEARCHERS
 PrognostX Health
PrognostX Health
Kentucky
USA
PrognostX Health is an in vitro clinical diagnostics development biotechnology company that develops innovative tests for the early identification and treatment of chronic diseases, with an emphasis on addressing the unmet clinical need and gap in testing modalities for chronic kidney disease (CKD), so that high-risk patients such as those with diabetes, hypertension and acute kidney injury can be treated before the disease becomes irreversible. Initially formed as AmDx PrognostX Inc, the unique family dynamic of The Carrithers Group – consisting of Dr. Stephen Carrithers, Dr. Aaron Carrithers and Dr. Brennan Carrithers – assembled their individual skill sets and experiences to form a new venture in which promising but under-developed early-stage technologies in renal care and diabetes health could be developed, validated, and brought to those unaware of their underlying conditions to address the rising incidence of CKD, dialysis and end-stage renal disease. Now doing business as PrognostX Health, the Carrithers are leveraging their experience and global partnerships to help provide better personalised care for those at high risk of (or with) CKD.

Dr. Aaron L. Carrithers, MD
Vice President and Chief Medical Officer, Principal Investigator
Dr. Aaron Carrithers is a physician-scientist and clinical translational entrepreneur who has leveraged his expertise and experience to evaluate, develop and commercialise clinical diagnostics. He graduated from the University of Kentucky College of Medicine in 2018 with his MD followed by a postdoctoral fellowship in Translational Medicine and Biotechnology whilst working at Sequela, a biotechnology company based in Kentucky, where he later became Chief Scientific Officer (CSO) prior to co-founding PrognostX Health. While undertaking his postdoctoral research he also became director of Clinical Operations at Lagrange Scientific, a company addressing kidney cancer through multi-omics technologies and big data algorithm development, a position he still holds. He is currently Vice President, Chief Medical Officer, and Principal Investigator at PrognostX Health.

Dr. Stephen L. Carrithers, PhD
President and Chief Executive Officer
Director of Innovation
Dr. Stephen Carrithers is currently the President and Chief Executive Officer as well as Director of Innovation of PrognostX Health. He received his PhD in Biochemistry and Molecular Biology from the University of Louisville School of Medicine in Kentucky. During his National Institutes of Health Postdoctoral Fellowship in Clinical Pharmacology at Thomas Jefferson University in Philadelphia, he co-founded his first biotechnology company which resulted in licensed products within the clinical diagnostics and therapeutics space for colon cancer and IBS-C. Dr. Carrithers joined the Division of Infectious Disease as Assistant Professor at the University of Kentucky Chandler Medical Center with a joint academic position in the Department of Physiology, Renal Division. During his academic tenure, he co-founded two more biotechnology companies. Over his extensive career, which has spanned more than 30 years, he has researched, explored, developed and commercialised various clinical diagnostics specifically looking at diabetes, diseases and cancers of the kidney. Recent work is also exploring the neurodegenerative sequelae of diabetes and CKD.
CONTACT
E: Aaron@PrognostXHealth.com
Contact@PrognostXHealth.com
W: http://www.PrognostXHealth.com/
COLLABORATORS AND PARTNERS
Dr. Brennan Carrithers, MD, MBA, MSc – New York University, Department of Psychiatry, and Board Member and Advisor for Health Insurance Implementation & Reimbursement at PrognostX Health
Ms. Tien Bui, BA – Chief Business Development Officer and Director of Strategic Partnerships at PrognostX Health
Dr. Eugene Krentsel, PhD – XLerateHealth, CSO and Vice President for Strategic Partnerships and Alliance, and President/Founder of innoVEK, LLC.
Research Use Only (RUO) ELISA kits available only through Ethos Biosciences (Newtown Square, PA)
FUNDING
Kentucky Cabinet of Economic Development, KY Innovation Grant Agreement CED 2021-002-001
Department of Health and Human Services, National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases, Grant 1R43DK129132
KY Innovation, Office of Entrepreneurship, Kentucky Cabinet for Economic Development and Kentucky Science and Technology Corporation
REPUBLISH OUR ARTICLES
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence (CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
SUBSCRIBE NOW
Follow Us
MORE ARTICLES YOU MAY LIKE
Prof Candis M. Morello – Prof Jan D. Hirsch | Recent innovations in pharmacy education
A pioneering research team from the Skaggs School of Pharmacy and Pharmaceutical Sciences, University of California, San Diego, United States, has been instrumental in developing innovative techniques for teaching pharmacy students. The Next Generation of Pharmacist Educators (NextGen-RxEd) programme is a new method of training the next generation of pharmacist educators and academics. To help pharmacists and pharmacy students visualise the complex issues experienced by their patients, the team led by Professors Candis Morello and Jan Hirsch developed an innovative educational tool, called the Medication Therapy Management (MTM) Spider Web.
Identifying Nutritional Risk in Early Childhood: Insights from NutriSTEP®
Early childhood is a critical period for growth and development, yet many young children face nutritionrelated risks that can go unnoticed. Professor Janis Randall Simpson and colleagues have developed NutriSTEP®, validated and reliable screening tools that help identify potential nutritional concerns in toddlers and preschoolers. Their large-scale analysis of Canadian data reveals patterns in diet, behaviour, and food access that could help guide early interventions and support healthy development.
Dr Lynn George | A New Link Between Elongator Dysfunction and TDP-43 Loss in ALS
At Montana State University Billings, Dr Lynn George and her team are investigating the cellular mechanisms underlying amyotrophic lateral sclerosis (ALS). Using a new mouse model, their research reveals that disruption of the Elongator complex leads to nucleolar stress and the loss of the protein TDP-43 from motor neurons. These findings connect two major hallmarks of ALS and offer new insight into how the disease develops.
Jella van de Laak | Could Tumour- Targeting Bacteria Enhance Cancer Immunotherapy?
Cancer immunotherapy has transformed treatment outcomes, yet many patients still fail to respond. At Maastricht University, Jella van de Laak, working with Professor Ludwig Dubois and Professor Jan Theys, is exploring how tumour-targeting bacteria could improve immune responses and expand treatment success.





