Dr Samantha Meenach – A Three-Dimensional Model of Lung Cancer
New and successful drug development for the treatment of lung cancer requires imaginative and creative thinking by scientists and doctors alike. Dr Samantha Meenach and her colleagues at the University of Rhode Island have developed an innovative approach for testing new anti-cancer drugs before they reach the stage of human trials.
Lung Cancer and Drug Development
Cancer is the second most common cause of death in the United States, with lung cancer resulting in the highest mortality rate. Cigarette smoking is the greatest cause of lung cancer, with exposure to radon gas and some industrial chemicals also contributing to the development of the disease.
Surgery is a very effective form of treatment for lung cancer; however, it is usually only helpful in about a quarter of patients at their time of diagnosis. The current recommended treatments for people who cannot undergo surgery are radiotherapy and chemotherapy. Radiotherapy involves directing beams of ionising radiation (X-rays) into the lung, which kill cancer cells where the beams intersect. Chemotherapy, on the other hand, requires the administration of drugs that also kill cancer cells, but these drugs often have many unwelcome side effects on other parts of the body. To allow the patient to recover from the treatment, these drugs are given at regular intervals with gaps in between.
There are several distinct stages involved in the development of any new drug. Firstly, researchers will discover a chemical that they believe will influence a disease. Secondly, this discovery is followed by laboratory studies, where the drug is given to cells in plastic dishes (in vitro), after which it can be tested on animals (in vivo) to gain a better understanding of how the drug works and determine potential safety problems. Finally, the drug is tested on small groups of humans followed by much larger clinical trials to test its effectiveness, safety and side effects.
‘Our mini tumours act more like tumours in the lungs and thus, when they are treated with newly developed drugs, we will have a better idea as to the efficacy of the drug before animal or human studies’
Thus, developing new, potentially better drugs for treating lung cancer is costly. It takes a long time and may involve the suffering of many animals to check various aspects of a drug’s behaviour before it can be tested on humans. And despite all this, many drugs fail to work when they finally make it to human clinical trials, or are found to be unsafe.
‘This failure is often due to poor evaluation of drugs at the laboratory stage,’ says Dr Samantha Meenach of the University of Rhode Island. ‘Therefore, we need better ways to assess lung cancer treatments before moving further in the drug development process.’

Scientists are trying to reduce the need for animal testing in drug development for both ethical and scientific reasons. Many drugs, including anti-cancer treatments, may behave very differently in laboratory animals compared to humans. More realistic drug testing in the laboratory would reduce the need for animals and produce more accurate results. Such improvements would also speed up the drug development process and reduce the costs of testing.
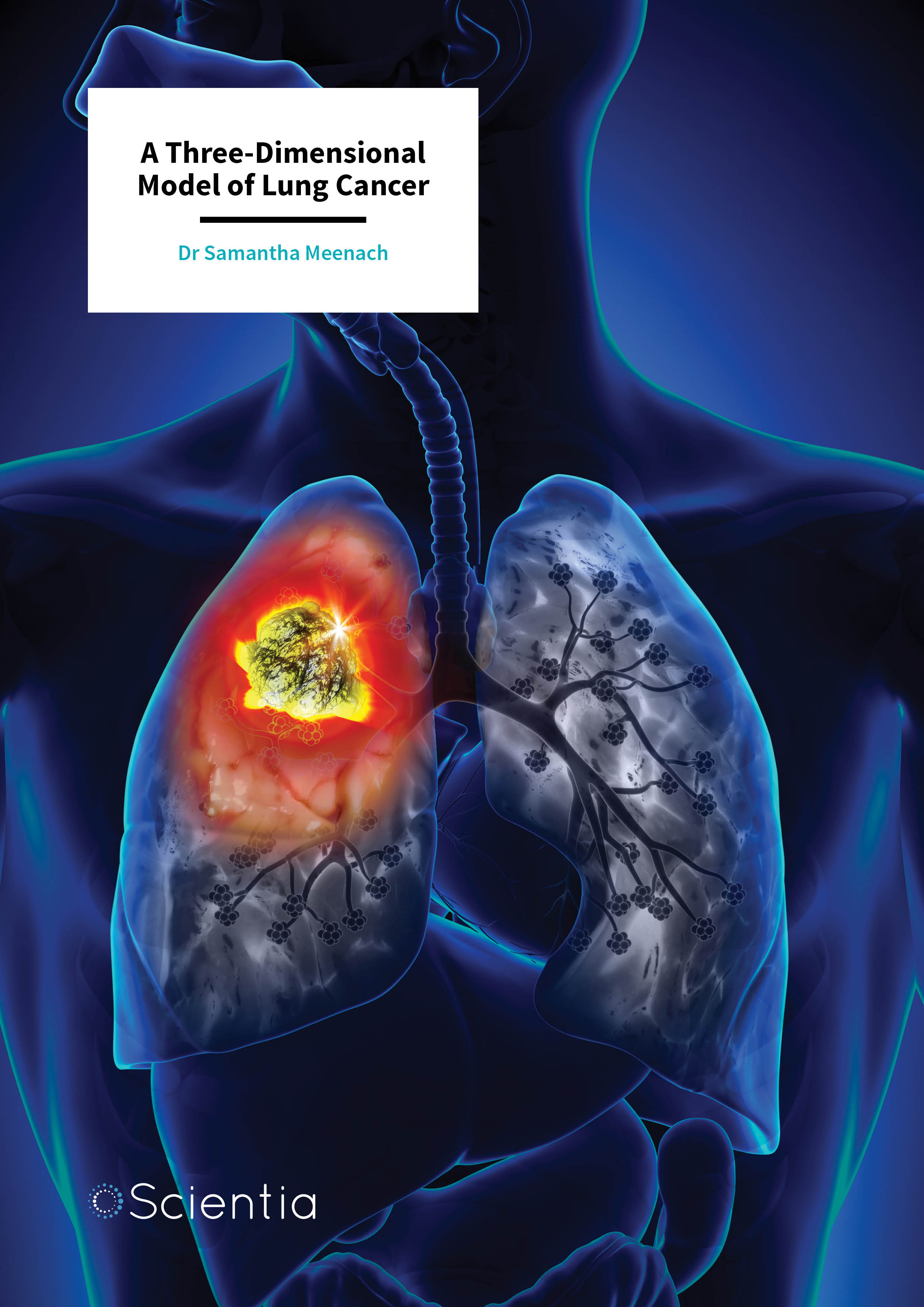
Individual tumour spheroid
3D Multicellular Spheroids
Typically, when scientists wish to test a potential anti-cancer drug, they first grow up cancer cells in a plastic dish filled with a liquid in a ‘2D culture environment’. They then add the drug to the dish to see what effect it has on the cells.
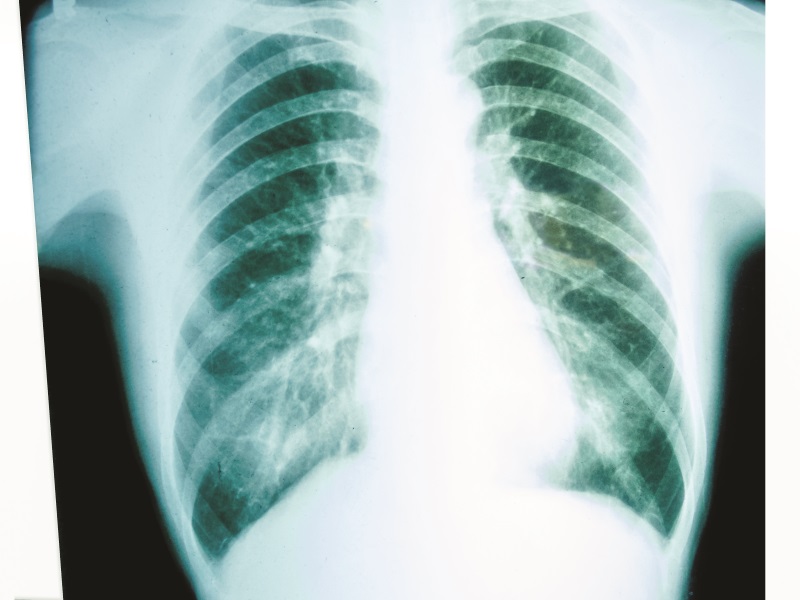
Rather than using this 2D arrangement of cells as a model for cancer, Dr Meenach’s group has developed a new in vitro model where lung cancer cells grow into 3D spheres, which they call ‘multicellular spheroids’. These 3D spheroids, which represent ‘mini-tumours’, have diameters that range between 100 microns (one hundredth of a millimetre) and a millimetre. It is important to note that the team’s mini-tumours begin as a small collection of cells (typically around a few thousand), just like in the early stages of lung disease.
‘Our mini tumours act more like tumours in the lungs and thus, when they are treated with newly developed drugs, we will have a better idea as to the efficacy of the drug before animal or human studies,’ explains Dr Meenach.
Lung cancer tumours grow quite differently from normal tissue within the body. After growing for between 21 to 30 days, tumours develop into a well-recognised three-layered structure. The outer layer of the tumour consists of living and actively-dividing cells, the middle layer contains cells that are alive but not very active and the inner core is made up of dead cells that have little or no access to oxygen or nutrients. In general, tumours have a poor blood supply as they have a smaller number of blood vessels compared to normal tissue, especially in the centre of the tumour.
Drug Penetration: Getting to the Heart of the Matter
After developing their 3D spheroids, Dr Meenach and her team decided to investigate how they would respond to common lung cancer drugs and how to improve their effectiveness. Remarkably, they discovered that their mini-tumours have the same layered structure as real lung tumours, and that commonly-used anti-cancer drugs acted mainly on the outer layer of cells. Just like in a real tumour, the internal cells did not respond very well to drugs. Dr Meenach believes that this variable response needs to be better addressed when probing new anti-cancer drugs in vitro, and further development of her team’s 3D tumour model could be the answer.
To improve the effectiveness of a particular anti-cancer drug, especially on the cells deep within a tumour, Dr Meenach and her colleagues co-administered a peptide with the drug molecule. They used the peptide called internalising arginylglycylaspartic acid (iRGD), which has a very special property in that it can increase the penetration of drugs inside a tumour.
The team added both the anti-cancer drug and iRGD to a culture containing their 3D spheroids at the same time. They found that the mini-tumours did not grow as well in the presence of iRGD and there was an increase in cancer cell death compared to spheroids that were exposed to the drug alone. This exciting step forward has enabled scientists, in a laboratory setting and without the use of animals, to deliver an anti-cancer drug that not only acts on the developing cells on the outside layer of a tumour, but also penetrates the tumour to kill the elusive internal cells.
Growth of tumour spheroids
Improved Drug Delivery
Typically, drug development experiments for lung cancer are conducted within a liquid culture; however, this does not accurately reflect how tumours grow within the airways of the lungs. To make their 3D spheroid tumour model even more realistic, Dr Meenach’s group developed a system where the spheroids are grown in a very shallow culture. Using the team’s approach, the cancer cells are not only exposed to the culture liquid, but they are also exposed to air, just like tumours in the lungs.
When drugs are delivered orally or through the blood stream, they become naturally diluted throughout the body. Therefore, a greater dose may be needed to have the desired effect on a disease. New drugs developed for the treatment of lung cancer may be more effective if they are delivered using an inhaler, just like those used by people with asthma.
By using an inhaler system, the drug is delivered directly to the lungs, which means that a smaller dose of the drug may be used and the side effects in other areas of the body could be reduced. In addition to this, since the drug is targeted to where it’s needed, it can begin to act more quickly. The actual administration of the drug would not require an injection by a doctor or nurse, and could easily be taken by the patient themselves.
Using their 3D spheroid mini-tumours, Dr Meenach and her group are currently developing lung cancer drugs that can be delivered as a spray and breathed in by a patient. The team’s preliminary results have shown that cancer cells can be initially treated much more effectively when using the aerosol, compared to administration of the drug through traditional means.
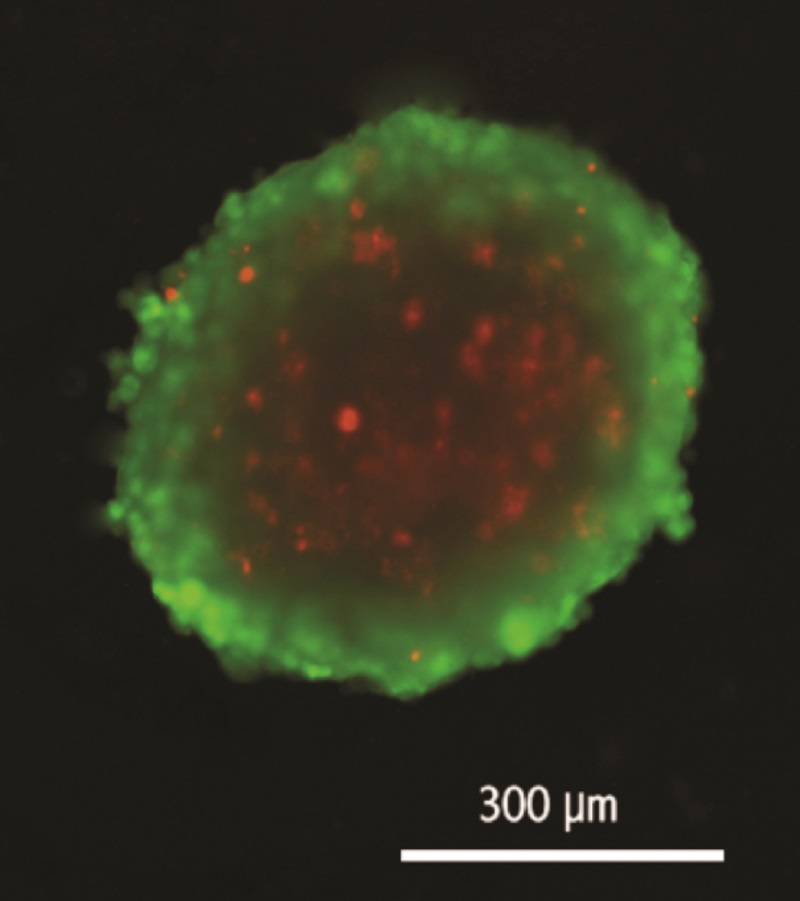
Stained tumour spheroid: green = live outer later, red = central non-growing cells
Future Directions
‘The next step in our work is to expand the types of lung cancer cell types we can develop as air-grown tumour spheroids,’ said Dr Meenach, when asked about her future plans. ‘In addition, we will be evaluating the efficacy of aerosol anti-cancer therapeutics on lung cancer tumour spheroids and comparing these results against conventional 2D cell culture, animal, and human studies.’
Dr Meenach and her group also wish to develop similar models for different types of tumours, not just within the lung but in other areas of the body too. By offering scientists a much more accurate alternative to traditional 2D cell culture models, the team’s techniques will provide a huge benefit to society, reducing costs, animal suffering and the time it takes to develop drugs, ultimately improving the survival rate of cancer patients worldwide.
Meet the researcher

Dr Samantha Meenach
Department of Chemical Engineering
Department of Biomedical & Pharmaceutical Sciences
University of Rhode Island
Kingston, RI
USA
Dr Samantha Meenach received her PhD in Chemical Engineering from the University of Kentucky in 2010. She currently holds the joint position of Assistant Professor at the University of Rhode Island, USA, in the Departments of Chemical Engineering and Biomedical and Pharmaceutical Sciences. She has published widely in her field and has been invited to present her research work at many international and national conferences. Dr Meenach is a member of a range of professional research organisations including the American Institute of Chemical Engineers, American Associations of Aerosol Research and Association of Cancer Research. In 2011, Dr Meenach received the prestigious National Cancer Institute-Cancer Nanotechnology Center Training Fellowship. In her spare time, she loves to travel, kayak and camp.
CONTACT
E: smeenach@uri.edu
W: https://web.uri.edu/cheme/smeenach/
FUNDING
National Science Foundation
Alternatives Research and Development Foundation
Rhode Island IDeA Network for Excellence in Biomedical Research
The Champlin Foundations
REFERENCES
SA Meenach, AN Tsoras, RC McGarry, HM Mansour, JZ Hilt, KW Anderson, Development of three-dimensional lung multicellular spheroids in air- and liquid-interface culture for the evaluation of anticancer therapeutics, International Journal of Oncology, 2016, 48, 1701–1709.
SK Gupta, EA Torrico Guzman, SA Meenach, Coadministration of a tumour-penetrating peptide improves the therapeutic efficacy of paclitaxel in a novel air-grown lung cancer 3D spheroid model, International Journal of Cancer, 2017, 141, 2143–2153.