Life Sciences & Biology

Professor Alan Templeton | When Fire Sparks Ecological Opportunity and Habitat Restoration
How far would you be willing to go to save an endangered species? Would you consider burning part of a forest as a solution? As unconventional as it may sound, conservationists sometimes resort to such measures to restore lost habitats. One remarkable example is the efforts to save eastern collared lizards – and indeed the entire biological community in which they live – in the Ozarks, spearheaded by American geneticist and statistician Professor Alan Templeton of Washington University in St Louis, USA.

Dr Michael Cherney – Professor Daniel Fisher | Unlocking Woolly Mammoth Mysteries: Tusks as Hormone Time Capsules
The impressive tusks found on proboscideans (the order of mammals that includes elephants, woolly mammoths, and mastodons) are like time capsules, preserving detailed records of their bearers’ lives in the form of growth layers and chemical traces. Frozen in time for thousands of years, these layers can unlock secrets about the lives of long-extinct relatives of modern elephants. Dr Michael Cherney and Professor Daniel Fisher from the University of Michigan used innovative techniques to extract and analyse steroid hormones preserved in woolly mammoth tusks. This ground-breaking work opens new avenues for exploring the biology and behaviour of extinct species.

Dr Sebastian Fraune | Microbiota: Fast-tracking Adaptation to Rapidly Changing Environments
As climate change continues at an unprecedented pace, the processes of natural selection and genetic mutation can no longer fully explain how some organisms adapt to their rapidly changing environments. Dr Sebastian Fraune from Heinrich-Heine University and an international team of researchers are the first to demonstrate a causal relationship between changes in the microbiome and changes in thermal tolerance. They propose that microbiota-mediated transgenerational acclimatisation can account for how animals adapt to their environments in much shorter periods of time than classical theory would predict.

Professor Danny Reinberg – Dr Gary LeRoy | Confronting a Rare and Deadly Cancer in Children
A diagnosis of cancer is never welcome but is particularly devastating when a child is found to have diffuse intrinsic pontine gliomas – a rare but particularly aggressive and treatment-resistant malignancy in the brain. In this article, we focus on the contributions of Professor Danny Reinberg and Dr Gary LeRoy at the University of Miami-Miller School of Medicine to understanding what makes these tumours so dangerous and, importantly, to developing much-needed approaches to improve the outcomes for these children.
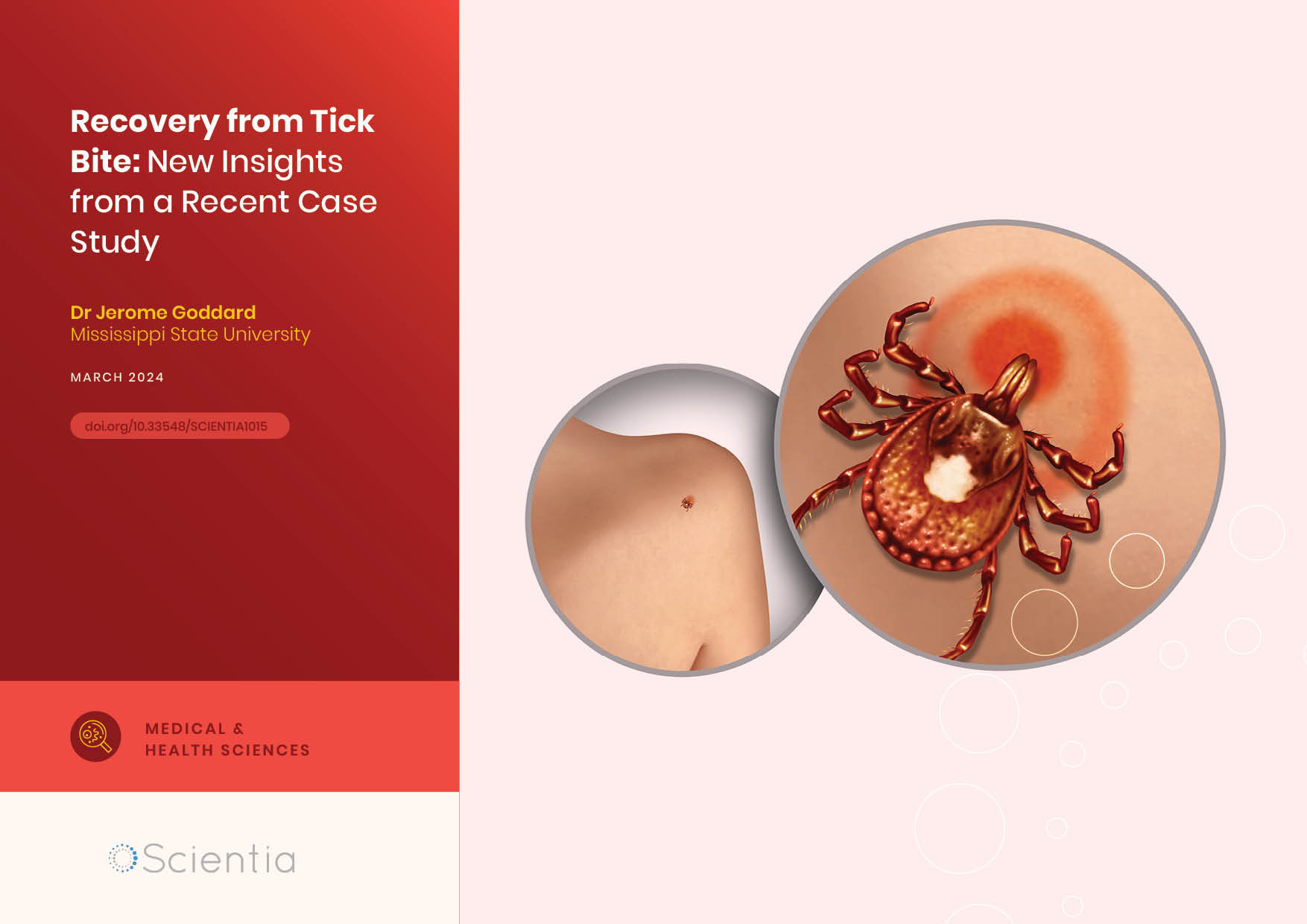
Dr Jerome Goddard | Recovery from Tick Bite: New Insights from a Recent Case Study
Dr Jerome Goddard of Mississippi State University and Dr Julie Wyatt of Wyatt Dermatology Clinic recently presented a case study of a hard tick bite trajectory over 30 days. Their work provides a detailed and novel account of the healing trajectory of an uncomplicated tick bite.
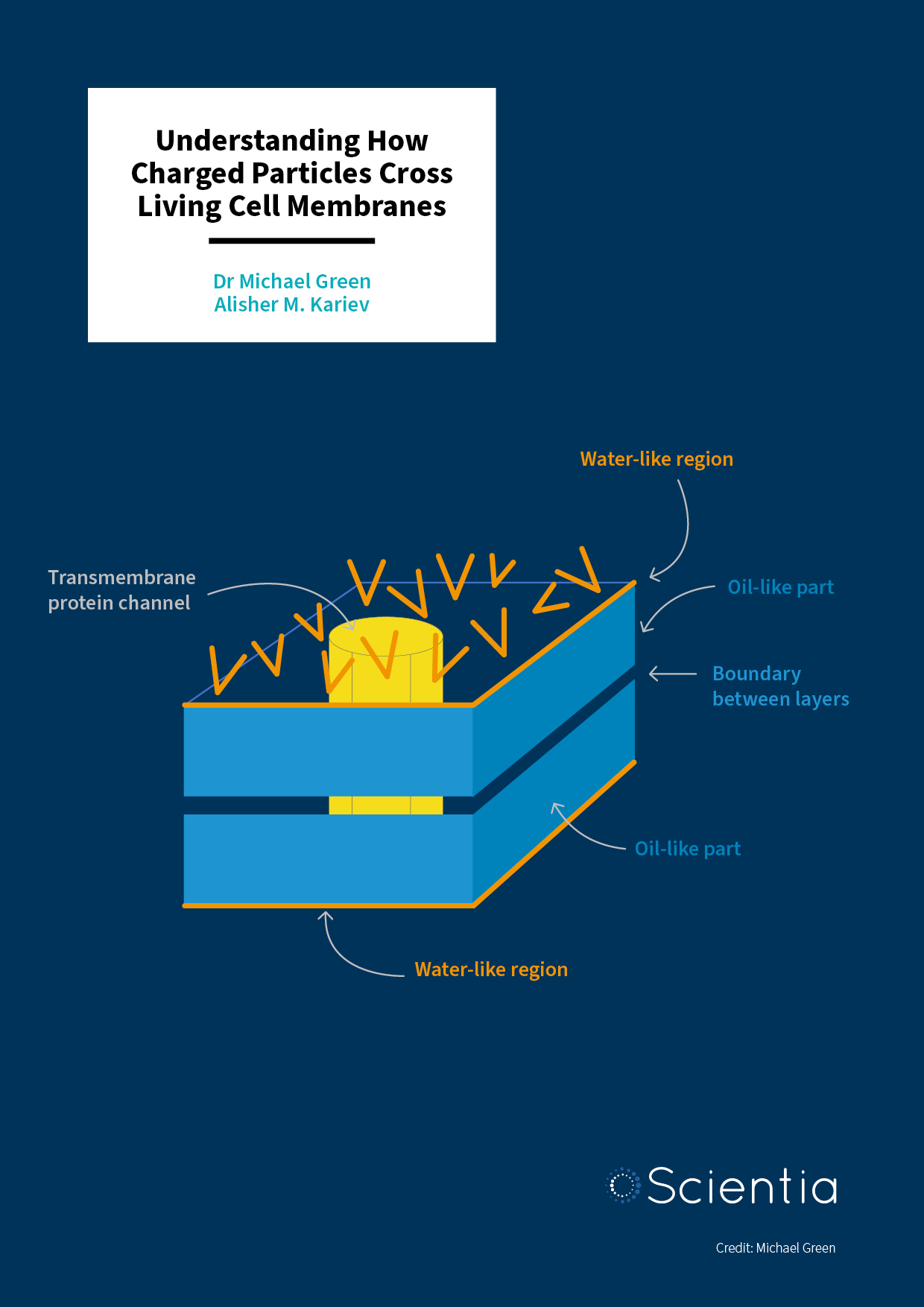
Dr Michael E. Green – Alisher M. Kariev | Understanding How Charged Particles Cross Living Cell Membranes
Living organisms function through a complex interplay of cellular mechanisms. The movement of charged atoms across cell membranes is key to diverse processes like cell growth and electrical signalling in the brain, and channels in these membranes open and close to allow charged atoms to pass through. Dr Michael Green and Alisher M. Kariev at the City College of the City University of New York challenge current models of channel opening and closing to provide a more complete understanding of this crucial and widespread biochemical process.
Professor Zachary Frome Burton | Learning to Code: tRNA and the Origin of Life
The importance of tRNA goes beyond its role in protein translation. tRNA is the molecule that ‘learned to code’ – its primordial emergence led to the genetic code and life itself. Professor Zachary Frome Burton of Michigan State University and Professor Lei Lei of the University of New England have reportedly solved tRNA evolution – proposing a theorem that sheds new light on the origin of life.
Dr Kay Cooksey | Developing Intelligent Packaging Solutions to Prevent Foodborne Illnesses
Many of us avoid foods that contain ingredients we haven’t heard of. However, many of these chemicals are beneficial because they prevent the growth of bacteria that cause foodborne illnesses. Although food quality standards have improved dramatically, outbreaks of foodborne illnesses are still common. Luckily, we have one line of defence to exploit: antibacterial food packaging. Dr Kay Cooksey of Clemson University is leading ground-breaking research to overcome the hurdles associated with antibacterial food packaging, bringing it one step closer to market.

Dale-Ila Riggs | Protecting Berry Crops from Invasive Pests with Exclusion Netting
Berry crops are vulnerable to numerous invasive pests, including spotted wing drosophila. These fruit flies have caused significant losses for farmers growing berries in different parts of the US, Asia, and Europe. A few years ago, Dale-Ila Riggs, the owner and operator of The Berry Patch, developed and implemented a series of exclusion netting systems that can effectively protect berry crops from spotted wing drosophila and other environmental threats, reducing the need for chemical pesticides.

Dr Stephen Love | Cultivating Sustainable Urban Landscapes with Native Plants
Landscapes exist on a spectrum – from those with minimal human impact, to urban sites wherein most natural features have been destroyed. As disturbed urban sites become more geographically prominent, imperatives to create managed urban habitats that provide societal and environmental services are increasingly urgent. Using native plants is a core principle of sustainable urban design, yet we lack standardised protocols to produce enough native plant products to meet demand. Dr Stephen Love at the University of Idaho has employed his expertise in plant breeding to develop high-throughput native plant domestication processes, to enhance offerings of native plants to people who wish to transform personal and public spaces into attractive, resilient ecosystems.

The European Society for Evolutionary Biology
Founded in 1987, the European Society for Evolutionary Biology (ESEB) is an academic society that brings together over 2000 biologists from Europe and beyond. In this exclusive interview, we speak with Professor Astrid Groot, President of ESEB, who discusses the many ways that the society supports scientists and helps to advance the diverse field of evolutionary biology.

Dr Alexandra (Sasha) Pavlova | Professor Paul Sunnucks – Genetic Rescue Saves Species from Extinction
Human impacts on plant and animal populations can be striking. Landscape-level transformation of pristine habitats means less room for plants and animals and the inevitable decline and extinction of many species. However, human impacts can also act on threatened species in less obvious ways, including on their genomes. Because of shrinking populations, many plants and animals are highly inbred, tipping the scales in favour of extinction. Dr Alexandra (Sasha) Pavlova and Professor Paul Sunnucks at Monash University, Australia, are at the forefront of applied research that seeks to increase genetic diversity to help populations rebound.

Complementing Plant Breeding Programs with Biotechnology
There are three components that impact crop harvests: the genetics of the seed sown, the environment in which the crop is grown, and the inputs employed during the growing period. For most species, the genetic component is addressed through breeding programs. Breeding programs require genetic variation within the available crop population from which parental crosses are made. Breeding programs that develop elite cultivars for commodities such as maize, soybean, wheat and sorghum, have wonderful inherent genetic diversity to utilise. However, many desired traits that breeders seek are not present within existing crop populations. It is here where the tools of biotechnology can complement plant breeding programs, by introducing novel pieces of genetic variation that can impart these favourable traits.

Dr Keerti Rathore – Bioengineered Cotton Could Help Solve World Hunger
Humans have relied on cotton’s textile fibre for nearly seven millennia. However, utilising cottonseed as food has been a long and unfulfilled goal of many plant breeders. Along with its abundant, high-quality protein, cottonseed also contains gossypol – a toxic chemical that renders the seed inedible. Cottonseed’s fate as a mostly unusable by-product seemed sealed until Dr Keerti Rathore, a professor at Texas A&M University, announced that he had successfully created gossypol-free cottonseed. Dr Rathore’s tireless devotion has given the world the potential to significantly improve food security worldwide.

Dr Y. Peng Loh – Discovery of Neurotrophic Factor-α1 Reveals New Treatment Strategies for Stress-induced Neurodegenerative Diseases and Depression
Stress produces numerous negative effects on the human body. Lying deep within the brain, one particularly sensitive area is the hippocampus, where chronic exposure to stress hormones can lead to the degeneration and death of neurons. Thankfully, the brain holds defence mechanisms that block some of these negative effects. Deciphering these mechanisms with the aim of better treating neurodegenerative diseases and depression is Dr Y. Peng Loh from the Eunice Kennedy Shriver National Institute of Child Health and Human Development in the USA.
Dr Madeleine Beekman – Unravelling A Biological Mystery: Why We Inherit More Maternal DNA
Around 1.5 billion years ago, our single-celled ancestor had a fateful snack – it engulfed another bacterium. The engulfed organism initially lived within the other, but slowly transformed over generations to give rise to mitochondria – an important organelle found in the cells of Eukaryotes. Interestingly, mitochondria have DNA that is passed exclusively from one parent onto their offspring. In most organisms, including us, that parent is the mother. Why we only inherent maternal mitochondrial DNA is one of the oldest unanswered questions in evolutionary biology. Dr Madeleine Beekman of the University of Sydney has explored this topic deeply, proposing new theories to explain why we are more related to our mother than our father.

Dr Martin Schwarz – Mapping the Brain’s Neuronal Networks to Understand Behaviour
The human brain is wonderfully complex. Billions of neuron cells connect in unique ways to create networks that determine each individual’s brain function and consequent behaviour. Given the expanse and complexity of these networks, it is not surprising that they are not yet fully mapped out or understood. Bringing innovative new and exciting ideas to this field of neurobiology is Dr Martin Schwarz from the Institute for Experimental Epileptology and Cognition Research and the Life & Brain Center at the University of Bonn Medical Center in Germany.

Dr Aliccia Bollig-Fischer – Understanding Why Obesity is a Risk Factor for Cancer
Cancer can be caused by genetic mutations or epigenetic alterations, which are changes to the way DNA is processed, rather than to the DNA itself. These changes can be brought about by obesity, and more specifically, oxidative stress and consequent reactive oxygen species. However, the molecular mechanisms by which this occurs are not well understood. Dr Aliccia Bollig-Fischer from Wayne State University School of Medicine in Michigan is studying these processes and paving the way for the development of novel cancer therapeutics.

Dr Mikail Abbasov – Discovering Unmapped Molecular Targets for Novel Covalent Drugs
Covalent drugs are molecules that irreversibly bind to specific, targeted sites in the body. They work to inhibit the disease-causing functions of certain proteins by preventing them from interacting with other substances. This is a highly promising field of drug development and the focus of Dr Mikail Abbasov from Cornell University, New York, USA. By creating and utilising new technologies and through collaborative research, Dr Abbasov has mapped novel molecular targets for potential covalent drugs to treat ailments ranging from cancer to autoimmune diseases.

Laura Arneson Horn – Planting Hedgerows to Boost Biodiversity and Protect Crops
From a bird’s eye view, farmlands are conspicuous. Unlike wildlands, swaths of agricultural fields form neat squares, fitting together in a landscape-sized jigsaw puzzle. The edges of the puzzle pieces stand out most: dark emerald lines contrasting against the light green interior crisscross across the land. These narrow, vegetated strips – or hedgerows – have been planted for centuries but are receiving renewed interest. Often referred to as ‘living fences’, hedgerows are buzzing communities that provide various ecosystem services. Laura Arneson Horn, the owner of Wild Bee Project in Salt Lake City, works to establish hedgerows and promote their positive impacts on native pollinators and other beneficial insects.

Juan Lavista Ferres | Dr Jan-Marino Ramirez | Dr Tatiana Anderson | Professor Edwin Mitchell – Understanding Sudden Unexpected Infant Death: A Unique Collaboration
When a supposedly healthy infant passes away, it can be hard to understand why. Juan Lavista Ferres (Microsoft), Dr Jan-Marino Ramirez and Dr Tatiana Anderson (both from Seattle Children’s Research Institute), and Professor Edwin Mitchell (University of Auckland), form the core of a novel collaboration to conduct vital and extensive research into the risk factors and mechanisms behind sudden unexpected infant death. This unique collaboration spanning across disciplines, industries and continents, is providing the deeper understanding that is needed to prevent unnecessary infant deaths.

Dr Marta Erlandson – Improving the Outlook for Children with Congenital Heart Disease
Congenital heart disease (CHD) is one of the most common birth defects across the globe. Although prospects and survival rates are improving, there is scant understanding or help available to get children with CHD active. Many believe physical activity is risky or outright dangerous, and as a result, children with CHD are at risk of obesity and other chronic conditions later in life. Dr Marta Erlandson from the University of Saskatchewan has aided the creation of CHAMPS, an innovative program for children with CHD, where researchers and children are learning how to manage the disorder from each other.

Prof Leon WMM Terstappen – A Novel Approach to Single Cell Identification, Isolation and Characterisation
The establishment of cell lines capable of producing high-quality monoclonal antibodies is imperative for the development of therapeutic agents and the advancement of biomedical research. Prof Leon WMM Terstappen and his team from the University of Twente in the Netherlands have developed a highly efficient method of identifying and isolating cells that produce high concentrations of specific antibodies. With his team, Prof Terstappen has further honed this method to aid in the refinement of molecular cloning techniques to increase the yield of superior quality monoclonal cell lines.

Dr. Eric Buckland – Translational Imaging Innovations: Accelerating Ophthalmic Research Through an Integrated Online Platform
Led by Dr. Eric Buckland, Translational Imaging Innovations, Inc. (TII) provides purpose-driven software systems that drive such ophthalmic research forward. The TII image management platform provides researchers with the tools to manage multifaceted imaging workflows and efficiently organize and analyse complex sets of images and data to accelerate the development of new diagnoses and treatments for eye diseases. By unleashing the power of the eye, TII aims to transform medicine.

Dr. David Westaway – Misfolding of Brain Proteins Triggering Neurodegenerative Diseases
Our DNA codes for proteins that are essential for the normal structure and function of our cells, tissues and organs. These proteins are folded in specific ways to facilitate these functions, but in disease states, this folding can go wrong. Dr. David Westaway from the University of Alberta in Canada investigates how and why protein misfolding occurs and how strains of misfolded proteins result in neurodegenerative diseases like dementia. His research is paving the way for novel therapies for these currently incurable and devasting conditions.

Dr W. Wyatt Hoback – NATIVES Program Trains Native Americans in Entomology
Insects are the most diverse lifeform on Earth. However, entomology, the scientific study of insects, is a field with few graduates. To address rising issues in agricultural biosecurity, pest management and biodiversity preservation, we need more trained entomologists. Moreover, because insects impact the health and well-being of almost everyone, there is also a need to increase diversity and to train underrepresented groups in entomology. One such group is Native Americans. To address the lack of entomologists in this key demographic, Dr W. Wyatt Hoback and a team from Oklahoma State University pioneered a program to engage Native American undergraduate students in entomology.

Dr Joanne Maki – Preventing Rabies: A Deadly but Neglected Disease
Rabies is transmitted from animals to humans through the bite of an infected animal, all too often with fatal consequences, particularly in the developing world. Dr Joanne Maki, Technical Director for the Veterinary Public Health Centre at Boehringer-Ingelheim Animal Health, has worked in rabies prevention for 30 years. With extensive background and experience gained in the rabies vaccine industry, Dr Maki shares her perspectives on the call to action to eliminate this deadly zoonotic disease.
Dr Chao Sun – The Complex Relationships Behind Tumour Progression in Cancer
The processes and metabolic pathways in our cells are complex and extensive, but essential for normal life functions, including cell respiration and energy production via the mitochondria. However, sometimes these processes go wrong resulting in disease, including cancer. Dr Chao Sun from the Institute of Modern Physics, Chinese Academy of Sciences, dedicates his research to unravelling the mechanisms behind tumour progression in relation to mitochondrial dysfunction.

Dr Ying-Xian Pan – The Holy Grail of Safer Opioids: Targeting Mu Opioid Receptor Splice Variants
Despite their numerous side effects, opioid drugs and morphine-like agents have remained a pillar in the medical management of pain. Most clinically used opioid drugs act through mu opioid receptors. Dr Ying-Xian Pan and his team from the Rutgers New Jersey Medical School, USA, studies the molecular and cellular mechanisms of mu opioid receptors and aim to develop novel strategies and opioid analgesics for better treating pain without side effects associated with traditional opiates. Efforts to find substitutes for traditional opioid drugs are helping address the opiate abuse crisis that affects many countries around the globe.
Prof Ferruh Artunc – Uncovering the Mechanisms Behind Nephrotic Syndrome to Develop Novel Therapeutics
Kidney disease impacts many people throughout the world and it can sometimes take the severe form of nephrotic syndrome. This condition results in many difficult symptoms but it is best represented by the presence of oedema, which is the severe swelling of the legs and eyelids. Currently, the exact mechanisms underlying oedema formation are poorly understood. Prof Ferruh Artunc from the Tübingen University Hospital in Germany is using his expertise in nephrology to uncover these mechanisms. His work has strengthened our comprehension of oedema formation in nephrotic syndrome and it may even lead to novel therapeutics in the future.

Dr Michael Wangler – Using Genetics to Diagnose Rare Metabolic Diseases
Identifying the cause of an illness in a sick baby or child is not always easy, particularly if the disease is rare. Throughout his career, Dr Michael Wangler, at the Baylor College of Medicine and Jan and Dan Duncan Neurological Research Institute, has investigated rare childhood diseases. Combining his expertise in paediatrics and genetics, Dr Wangler utilises genomics, metabolomics and the humble fruit fly to identify the genes responsible for rare and undiagnosed diseases to improve both diagnosis and treatment.
Dr Stephen Kry – Improving the Quality and Accuracy of Radiotherapy Through Research
Radiation therapy is an effective and widely used method of treating cancer, and as with any treatment, it is essential to get the right dose. However, Dr Stephen Kry from The University of Texas MD Anderson Cancer Center has found widespread errors in the systems that calculate the doses patients receive. Through his research, he has helped to identify where these errors occur, how common they are, and provide possible solutions. He hopes that his work will go on to improve the quality and efficacy of radiotherapy for many cancer patients.
