Dr Joanne Maki – Preventing Rabies: A Deadly but Neglected Disease
Rabies is transmitted from animals to humans through the bite of an infected animal, all too often with fatal consequences, particularly in the developing world. Dr Joanne Maki, Technical Director for the Veterinary Public Health Centre at Boehringer-Ingelheim Animal Health, has worked in rabies prevention for 30 years. With extensive background and experience gained in the rabies vaccine industry, Dr Maki shares her perspectives on the call to action to eliminate this deadly zoonotic disease.
The Origins of Rabies
The rabies virus has plagued mankind since the beginnings of recorded history. Its origins are postulated to be an insect virus that adapted to insectivorous bats and over time evolved to infect other mammalian species – including humans.
Dr Joanne Maki, Technical Director for the Veterinary Public Health Centre at Boehringer-Ingelheim Animal Health notes, ‘The most striking feature of the rabies virus is how it is transmitted. Upon receiving a bite from a rabid animal, the virus replicates locally and invades local nerves. The infection continues with virus replication in neurons eventually invading the central nervous system and travelling to the brain.’
The infected host develops inflammation of the brain, known as encephalitis. This presents either as paralysis and recumbency or agitation and aggression. Dr Maki further explains, ‘It is an evolutionary marvel that the virus modifies the host’s behaviour to seek out and bite objects in the environment.’ As such, the virus is transmitted in saliva from the infected host to another, and the cycle begins again.
As an RNA virus, the rabies virus replicates with low genetic fidelity meaning it can easily modify its genetic code and as a result, has developed different variants capable of infecting all mammalian species. The variants are identified through genetic sequencing. The fingerprints of the variants are unique by both geography and species. They survive by maintaining low levels of transmission in wildlife populations such as raccoons, foxes, skunks, bats, and mongooses. Although originating in wildlife, rabies has adapted readily to circulate in unvaccinated dogs. Dogs, often dubbed ‘man’s best friend’ are the primary cause of human rabies deaths globally.
Can We Eliminate Rabies?
Dr Maki explains that the tools to eliminate human rabies already exist. Human rabies vaccines are routinely given to people bitten by animals in a treatment called post-exposure prophylaxis (PEP). She notes that injectable inactivated canine rabies vaccines have proven time and time again to stop dog rabies outbreaks, and that oral rabies vaccines for wildlife have been used to eliminate fox rabies in Western Europe and the canine variant of rabies from the USA.
So, why are humans still dying from rabies? Dr Maki points to a lack of disease awareness, prioritisation of prevention, and a cultural willingness to vaccinate dogs to protect human lives as being the main obstacles. Despite this, almost all human rabies victims (>99%) have been bitten by a rabid dog. An estimated 59,000 human deaths occur each year due to rabies, and these are primarily in countries lacking adequate health care infrastructure. As many as 40% of these deaths are in children under 15 years of age living in Africa and Asia. Sadly, the Centers for Disease Control in the US estimates one person dies of rabies every 9 minutes worldwide.
For these reasons, the World Health Organization (WHO) has classified human rabies as a neglected tropical disease. Shockingly, the global burden of rabies is estimated to be 8.6 billion USD/year, based primarily on human lives lost, the money spent on PEP, and the loss of economic productivity by those impacted by the disease.
The global rabies community is now challenging all countries to unite and invest in rabies prevention and ultimately, eliminate human rabies by 2030. Known as ‘Zero By 30’, this effort is spearheaded by a group called United Against Rabies (UAR). This effort will require a sustained One Health approach to repeatedly vaccinate dog populations against canine rabies in countries in which rabies is rife to dramatically reduce the risk of exposure to humans.
‘As countries roll out mass COVID-19 vaccination programmes, one can only hope that lessons learned and investments made in health care can have a positive impact on preventing other infectious diseases, such as rabies.’

The Importance of Post-exposure Prophylaxis
Adequate post-bite medical care greatly increases the chance of a person surviving a rabid dog bite. In countries with good health care provision, human deaths from rabies are rare and the disease is routinely prevented through PEP. This consists of a series of injectable rabies vaccines and in high-risk bite cases, infiltration of the wound site with anti-rabies immunoglobulin.
Every year, more than 29 million people across the world receive post-bite medications. However, human deaths due to rabies still occur. Insufficient PEP supply in rural hospitals and clinics (such as in Africa) limits the potential number of dog bite victims treated, directly contributing to the death toll from human rabies. Sadly, in some situations, people cannot afford treatment or there are logistical roadblocks such as not being able to take time away from work or lack of transportation to the hospital for follow-up care.
Whether an individual can receive PEP after a dog bite is often geographically and economically driven. Dr Maki explains that WHO, in collaboration with Gavi, the Vaccine Alliance, has committed to providing PEP vaccine doses to low-income countries based on the country’s gross national income. A 2019 epidemiological and economic modelling study confirmed the value of providing anti-rabies vaccines through the potential to prevent an additional 489,000 deaths between 2020 and 2035.
WHO also investigated the impact of using a one-week course of intradermal vaccinations for PEP instead of the current intramuscular route protocol and approved this for human rabies PEP in 2018. However, as Dr Maki notes, the practice has not been uniformly adopted. This slow transition to intradermal PEP administration is thought to be partially due to a lack of proficiency in staffing and valid concerns about potential contamination of multi-use vaccine vials – both of which can be addressed through training. Benefits of intradermal PEP vaccination include the use of a lower dose per treatment and the need for fewer visits, in addition to an overall reduction in cost by 60–80%.
Integrated bite case management (IBCM) is a decision tool developed for physicians treating dog bite victims. In every case, the attending physician must decide if a patient is truly at risk of contracting rabies. If the dog that inflicts the bite runs away or cannot be identified, then the vaccination status of the biting dog is ‘unknown’. In such instances, PEP doses are routinely administered. IBCM gives physicians an algorithm to not choose to administer PEP if the biting dog is known and currently vaccinated against rabies, given that properly vaccinated dogs pose the lowest risk of rabies transmission. Any dog that has bitten a person should be quarantined and observed. If the dog remains healthy after 10 days of quarantine the PEP series of vaccines can be stopped. Dr Maki notes that incorporating IBCM into routine dog bite patient care can prevent unnecessary administration of rabies vaccines, thus improving cost and resource savings.
Critically, the Hampson/Trotter model hypothesises that if all PEP vaccines were given intradermally and integrated bite case management was implemented on a global scale the current supply of human anti-rabies vaccines would be enough to eliminate human rabies.
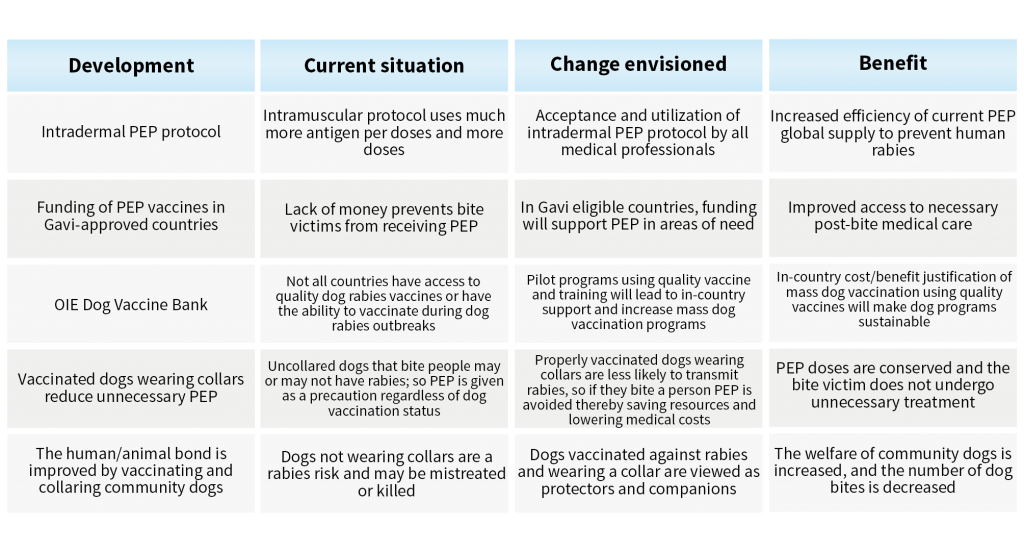
Table 1. Recent developments supporting the elimination of human rabies by 2030.
The Canine Rabies Vaccine Bank
Since vaccinating a sufficient number of dogs each year is critical to eliminating human rabies, the World Organization for Animal Health (OIE) created a canine rabies vaccine bank for countries, non-government organisations and other third parties interested in conducting mass dog vaccination programmes. To date, OIE has delivered 12.5 million vaccine doses to more than 22 countries (principally in Asia and Africa). Since the inception of the vaccine bank, Boehringer-Ingelheim Animal Health has provided doses of their injectable rabies vaccine RABISIN. Dr Maki is particularly proud of this contribution, not least because obtaining quality vaccines for mass dog vaccination programmes by individual countries can be financially very challenging.
Dr Maki hopes that as canine rabies comes under control, governments and policymakers will realise the societal benefits of healthier animals and fewer human deaths due to rabies. As rabies prevention is recognised as being an economically sound practice, mass dog rabies vaccination programmes become routine and canine rabies outbreaks prevented. As recently demonstrated in Mexico, a sustained and well-orchestrated canine rabies prevention programme can eventually eliminate human rabies deaths. As such, Mexico was recognised by WHO for eliminating human deaths due to canine rabies in 2019. RABIFFA, another Boehringer-Ingelheim Animal Health rabies injectable vaccine, was used for many years in this programme.
Recent Developments
Dr Maki recognises five recent advancements that have significantly bolstered the steps to eliminating human rabies. Three of these initiatives impact the delivery of PEP as discussed above: the funding of human rabies vaccines in impoverished countries, the implementation of IBCM, and the transition from intramuscular administration of PEP vaccines to intradermal delivery. The other two actions supporting effective mass dog vaccination are using collars to identify vaccinated dogs and the creation of a canine rabies vaccine bank (see Table 1).
Looking to the Future
Taking a reflective stance, Dr Maki suggests that only time will tell if the challenges caused by the COVID-19 global pandemic will bring a suitable investment in public health infrastructure and disease prevention to the world’s stage. Can a positive outcome of our current suffering be the realisation that brings equality to improved access to vaccines and medical care? Dr Maki asserts, ‘As countries roll out mass COVID-19 vaccination programmes, one can only hope that lessons learned and investments made in health care can have a positive impact on preventing other infectious diseases, such as rabies.’
Dr Maki further reflects that ‘While we wait for better times, we can continue to raise awareness about rabies and do our best to vaccinate dogs. We can commit to building a global network that shares the benefits of intradermal PEP and IBCM to improve medical post-bite care.’ Clearly, such changes are not complex and can greatly improve a person’s chance of not dying of rabies. By working together in a One Health mindset, Dr Maki believes we can implement these changes and move closer to the global elimination of human rabies arising from the canine variant.
SHARE
DOWNLOAD E-BOOK
LISTEN TO THE AUDIO
REFERENCE
https://doi.org/10.33548/SCIENTIA757
MEET THE RESEARCHER

Dr Joanne Laila Maki
Veterinary Public Health Center
Boehringer-Ingelheim Animal Health
North America
Dr Joanne Maki is the Technical Director for the North America, Veterinary Public Health Center at Boehringer-Ingelheim Animal Health. She is a recognised global technical expert in rabies and rabies prevention. Her areas of expertise include supporting programs focused on the elimination of human rabies transmitted by dogs and wildlife rabies prevention. Dr Maki received her Doctor of Veterinary Medicine from Louisiana State University and her PhD from the University of Georgia. She was a National Institutes of Health principal investigator at the University of Georgia, Athens from 1996 to 2002 where she developed a model to study mucosal immunity. As a pharmaceutical technical director, she led efforts to license three commercial veterinary rabies vaccines including a non-adjuvanted injectable recombinant vaccine for cats, an oral vaccine for wildlife and a combination vaccine for horses.
CONTACT
E: joanne.maki@boehringer-ingelheim.com
W: https://acc-d.org/about-us/leadership-staff/joanne-maki-bio
KEY COLLABORATORS
Dr Anne‑Laure Guiot
Dr Michel Aubert
FUNDING
Boehringer-Ingelheim Animal Health
FURTHER READING
J Maki, A Guiot, M Aubert, et al., Oral vaccination of wildlife using a vaccinia-rabies-glycoprotein recombinant virus vaccine (RABORAL V-RG®): a global review, Veterinary Research, 2017, 48(1), 57.
JL Maki, HW Dickerson, Systemic and cutaneous mucus antibody responses of channel catfish immunized against the protozoan parasite Ichthyophthirius multifiliis, Clinical and Vaccine Immunology, 2003, 10(5), 876–81.
A Fischer, VA Benk, JR Briggs, et al., Effectiveness of GonaCon as an immunocontraceptive in colony-housed cats, Journal of Feline Medicine and Surgery, 2018, 20(8), 786–792.
A Wohlers, EW Lankau, EH Oertli, J Maki, Challenges to controlling rabies in skunk populations using oral rabies vaccines, Zoonoses Public Health, 2018, 65(4), 373–385.

REPUBLISH OUR ARTICLES
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence (CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
SUBSCRIBE NOW
Follow Us
MORE ARTICLES YOU MAY LIKE
Dr Ralf Adam | New Technologies Shaping the Future of Oral Hygiene
Understanding the efficiency of various toothbrush technologies is essential for achieving optimal oral health. Dr Ralf Adam, who leads a dedicated team at Procter & Gamble in Germany, is keen to investigate the complexities of these technologies. His team have provided new insights into the best toothbrush types for plaque removal and the maintenance of gum health. By highlighting the importance of informed oral care decisions and ongoing investigations, this vital research works towards ensuring everyone can achieve a brighter, healthier smile.
Professor Alan Templeton | When Fire Sparks Ecological Opportunity and Habitat Restoration
How far would you be willing to go to save an endangered species? Would you consider burning part of a forest as a solution? As unconventional as it may sound, conservationists sometimes resort to such measures to restore lost habitats. One remarkable example is the efforts to save eastern collared lizards – and indeed the entire biological community in which they live – in the Ozarks, spearheaded by American geneticist and statistician Professor Alan Templeton of Washington University in St Louis, USA.
Dr Toby Phesse | Revealing the Mysteries of Wnt Signalling: Novel Approaches to Beating Cancer
Cancer remains a leading cause of mortality worldwide, and the need for new, more effective treatments remains an urgent challenge. Dr Toby Phesse from Cardiff University in the UK focuses on the role of the Wnt receptor found on the surface of cells and its involvement with cell communication and cancer growth, bringing fresh hopes for new therapeutic options.
Dr Vijay Reddy | The Virus World Database: An Invaluable Resource for Public Health and Healthcare
Severe viral disease presents an ongoing challenge to the health of humankind. While unparalleled developments in science and technology are improving our understanding of such viruses, this information needs to be readily accessible to researchers to ensure continued progress in public health and healthcare. Dr Vijay Reddy and his colleagues at the Hormel Institute (University of Minnesota) developed the Virus World database, an invaluable resource that details the genome, structure, and host of practically every discovered virus to date.