Dr Mikail Abbasov – Discovering Unmapped Molecular Targets for Novel Covalent Drugs
Covalent drugs are molecules that irreversibly bind to specific, targeted sites in the body. They work to inhibit the disease-causing functions of certain proteins by preventing them from interacting with other substances. This is a highly promising field of drug development and the focus of Dr Mikail Abbasov from Cornell University, New York, USA. By creating and utilising new technologies and through collaborative research, Dr Abbasov has mapped novel molecular targets for potential covalent drugs to treat ailments ranging from cancer to autoimmune diseases.
Covalent Drugs: Creating Irreversible Bonds
Therapeutic drugs come in numerous forms for an endless number of diseases. Depending on the ailment in question, a drug will target certain biochemical reactions in the body to reduce its negative impacts. This involves the drug in question binding to a target protein in cells. Whereas some therapeutics form bonds that are reversible, others form permanent bonds to create a long-lasting effect. For example, covalent drugs form strong, irreversible covalent bonds with proteins. Well-known examples of this type of drug include the anti-inflammatory, Aspirin, the antibiotic, Penicillin, and the cancer drug, Ibrutinib.
In the past, there have been safety concerns surrounding covalent drugs, with many scientists viewing their irreversible binding actions as dangerous. Some studies showed that these drugs could be metabolised to create highly reactive intermediates that, if accidentally targeted the wrong protein, could become toxic to a patient. More recently, a resurgence in interest in covalent drugs has exploited their compelling benefits whilst fine-tuning how they react in the body to ensure their safety.
These new drugs are called targeted covalent inhibitors and they work by first being guided towards and weakly bonding to the desired protein through a small pocket. This initial interaction facilitates the subsequent formation of the irreversible covalent bond with a nearby amino acid. Consequently, the function of that protein is inhibited.

Covalent inhibitors target nucleophilic amino acids – molecules on proteins that form bonds with an electrophile by donating an electron pair. They can be designed to target very specific nucleophiles on a protein so that other proteins within the same family are not impacted when the drug is administered. This clever technique is being utilised in a number of drugs that are currently in late-stage development. Many of them are targeting different types of cancer and some are even being tested for COVID-19 treatments, such as Paxlovid.
Although the field of covalent drugs is highly promising, there is a distinct lack of research as of yet and much more work is needed to make full use of its potential. Dr Mikail Abbasov from the Department of Chemistry and Chemical Biology at Cornell University in New York, USA dedicates his research to improving our knowledge and understanding of the targets of covalent drugs and how they could be used to create ground-breaking therapies.
Working alongside his colleagues, Dr Abbasov explains, ‘Our highly interdisciplinary research program spans the fields of chemistry, biology and physics with an emphasis on the development of innovative chemical proteomic tools and technologies that modulate protein function and interrogate pathophysiological signalling pathways associated with human diseases, such as cancer, viral infections, autoimmune and neurodegenerative disorders.’
‘Findings from our laboratory have enriched our understanding of the mechanistic underpinnings of pathological processes and provide valuable leads for the design and development of novel therapeutics.’
Discovering Simple Molecules with Drug Potential
Historically, to understand the relationships between the structure and activity of a protein, total synthesis followed by structural derivatisation has been performed. Total synthesis is the process of using chemistry to form a complete, complex molecule using commercially-available ingredients. These complex molecules are usually natural products, which are substances that are created by living organisms and plants, meaning they are already found in nature. Structural derivatisation refers to chemically transforming a chemical compound into a different, yet structurally similar one, known as a derivative.
However, Dr Abbasov saw that these synthetic techniques to create natural products were not usually guided by hypotheses that took into consideration the structural features of the compounds that are needed for bioactivity. Therefore, Dr Abbasov, in collaboration with Dr Daniel Romo at Baylor University in Texas, developed a new strategy for natural product synthesis which he called ‘pharmacophore-directed retrosynthesis’. A pharmacophore is the section of a molecule that is necessary for its biological and pharmacological activity.
Retrosynthesis is a method used to understand molecule synthesis by breaking down the final product into more simple precursors. They are continually simplified until the resulting molecules are commercially available or easy to use in a synthetic reaction for the target molecule. Dr Abbasov performed pharmacophore-directed retrosynthesis with a pre-determined or a hypothetical pharmacophore of a natural product in mind. As a result, the intermediates that were synthesised in the process could each be tested for their bioactivity during the total synthesis of the natural product.
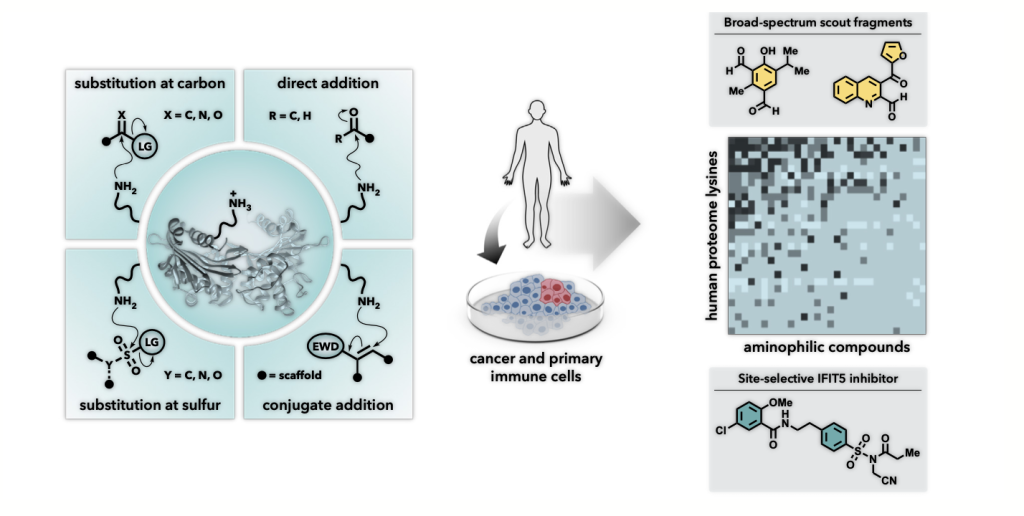
Discovering druggable lysines. Credit Mikail Abbasov
Putting this innovative new method into practice, Dr Abbasov investigated gracilin A, a natural product isolated from the Mediterranean sponge Spongionella gracilis. The biological role of the gracilin A prior to this study was largely unexplored.
For this reason, Dr Abbasov endeavoured to use his pharmacophore-directed retrosynthetic approach to create structurally simplified derivatives of gracillin A that can be accessed in the early stages of the synthesis. He aimed to identify derivatives with selective affinity (attraction) towards cyclophilin A and cyclophilin D, proteins associated with immunosuppressive and neuroprotective activities, respectively. Consequently, Dr Abbasov identified simplified, ultrapotent derivatives that exhibited activity greater than that of gracilin A.
This is an important finding because it demonstrates that structurally simplified intermediates can possess greater biological activity than that of the parent natural product, thus reinforcing the advantages that this approach offers for the rapid discovery of potential therapeutics. In particular, the derivatives that have a high affinity for cyclophilin D over cyclophilin A could be useful as neuroprotective drugs that do not have immunosuppressive side effects.

Discovering and Mapping Druggable Lysines
In a subsequent study, Dr Abbasov and his colleagues conducted new and exciting research on biological targets for covalent drugs. In particular, they focused on covalent targeting of amino acids – the building blocks of proteins. Twenty different amino acids are found in the human body and historically, two of them called cysteine and serine have been the main focus for covalent drugs. However, this limits the possibilities for drug discovery.
Consequently, Dr Abbasov, in collaboration with Dr Benjamin Cravatt at Scripps Research in California, set out to investigate another amino acid called lysine. They also brought in teams from the Massachusetts Institute of Technology, Pfizer and Leiden University for the project. Together, they developed a chemoproteomic technology that allowed them to study how lysines react with small molecules. They aimed to find aminophilic electrophiles – chemical species that form irreversible bonds with nucleophilic amine groups on lysines.
This ground-breaking work led Dr Abbasov to profile and log over 200 of these small molecules. By testing human cancer and immune cells, the team found thousands of reactive lysines. This included functional sites on proteins that were previously seen as too difficult to target. Collating this new data allowed Dr Abbasov and his colleagues to create a comprehensive map of lysines that are now known to be possible drug targets.
By fine-tuning both the recognition portion and the reactive fragment of the small molecules, they were able to identify a wide range of proteins in which these lysines are found. These proteins have a wide range of functions in the human body and are known to be involved in various diseases, including, for example, enzymes and transcription factors (needed for protein synthesis). By inhibiting the function of disease-relevant proteins, these types of small molecules could serve as drugs to prevent the harmful progression of a variety of diseases.
According to Dr Abbasov, ‘The findings from this project provide a blueprint for chemical biologists and medicinal chemists to understand the rules that govern lysine-targeting covalent inhibitor design, eventually leading to new treatments and drugs.’ To aid this development, after mapping the druggable lysines, Dr Abbasov and his team combined this data with human genetic information and cell activation measurements.
This helped to refine their understanding of how different small molecules can impact the activity of the protein and consequently, the cell. They were able to demonstrate that, through lysine binding, aminophilic compounds disrupted a number of biochemical functions within cells, including those involved in the innate immune response. With such real promise for finding and creating new covalent drugs, Dr Abbasov’s work is not stopping there.
‘Findings from our laboratory have enriched our understanding of the mechanistic underpinnings of pathological processes and provide valuable leads for the design and development of novel therapeutics’ notes Dr Abbasov. And this research is ongoing; only one year after starting his position at Cornell University, Dr Abbasov secured a $2M grant from the National Institute of General Medical Sciences to continue discovering the druggable targets of the human body.
SHARE
DOWNLOAD E-BOOK
WATCH THE ANIMATION
LISTEN TO THE AUDIO
REFERENCE
https://doi.org/10.33548/SCIENTIA779
MEET THE RESEARCHER

Dr Mikail E. Abbasov
Department of Chemistry and Chemical Biology
Cornell University
Ithaca, NY
USA
Dr Mikail Abbasov was born in Baku, Azerbaijan, but his family had to flee in 1988 to escape devastating conflict. They moved to a rural community in Rahachow, Belarus, which was part of the Chernobyl exclusion zone. This meant that it was common for research groups to visit and carry out tests at the local hospital near Dr Abbasov’s school. He became fascinated by their work and was keen to help them when he could. Now an American citizen, Dr Abbasov values the importance of quality education and scientific research, and employs this understanding in all of his work.
His mother is a classical pianist and composer and his father is a painter and sculptor, both influencing Dr Abbasov to pursue the Arts in many forms. He graduated from the School of Music and Performing Arts and is classically trained as a baritone and in the piano. As a keen artist, Dr Abbasov has had his illustrations featured on the covers of several scientific journals and as an actor, he has played a supporting role in the award-winning independent film, The Heist.
Dr Abbasov completed his BSc in Biochemistry and his MSc in Optical Physics at West Texas A&M University in Canyon, Texas. He went on to achieve a PhD in Organic Synthetic Chemistry at Texas A&M University in College Station, Texas, and completed postdoctoral research at The Scripps Research Institute in California. Currently, Dr Abbasov serves as an Assistant Professor in the Department of Chemistry and Chemical Biology at Cornell University in New York. Here, he carries out his research into the targets of covalent drugs and how they can be utilised to develop effective, novel therapeutics.
CONTACT
W: https://www.chemikailproteomics.com
Twitter: @Abbasov_Cornell
KEY COLLABORATORS
Scripps Research (La Jolla, California, USA)
- Benjamin F. Cravatt (https://www.scripps.edu/cravatt/)
- Madeline E. Kavanagh
- Taka-Aki Ichu
- Michael R. Lazear
- Yongfeng Tao
- Vincent M. Crowley
- Melissa M. Dix
- Radu Suciu
Massachusetts Institute of Technology (Cambridge, Massachusetts, USA)
- Laura L. Kiessling (http://kiesslinglab.com/)
- Jordan Ho
Leiden University (Leiden, Netherlands)
- Stephan M. Hacker (https://www.universiteitleiden.nl/en/staffmembers/stephan-hacker#tab-1)
Pfizer (Groton, Connecticut, USA)
- Christopher W. am Ende
- Matthew M. Hayward
Baylor University (Waco, Texas, USA)
- Daniel Romo (http://www.danielromogroup.com/)
- Christian M. Chaheine
University of Santiago de Compostela (Lugo, Spain)
- Luis M. Botana (https://investigacion.usc.gal/investigadores/58431/detalle?lang=en)
- Rebeca Alvariño
FUNDING
National Institute of General Medical Sciences: Chemoproteomic mapping of the ligandable ribonucleoproteome using phloroglucinol meroterpenoids (1R35GM143027-01; $2M)
FURTHER READING
ME Abbasov, ME Kavanagh, T-A Ichu, et al., A proteome-wide atlas of lysine-reactive chemistry, Nature Chemistry, 2021, 13(11), 1081–1092.
ME Abbasov, R Alvariño, CM Chaheine, et al., Simplified immunosuppressive and neuroprotective agents based on gracilin A, Nature Chemistry, 2019, 11(4), 342–350.

REPUBLISH OUR ARTICLES
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence (CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
SUBSCRIBE NOW
Follow Us
MORE ARTICLES YOU MAY LIKE
Dr Ralf Adam | New Technologies Shaping the Future of Oral Hygiene
Understanding the efficiency of various toothbrush technologies is essential for achieving optimal oral health. Dr Ralf Adam, who leads a dedicated team at Procter & Gamble in Germany, is keen to investigate the complexities of these technologies. His team have provided new insights into the best toothbrush types for plaque removal and the maintenance of gum health. By highlighting the importance of informed oral care decisions and ongoing investigations, this vital research works towards ensuring everyone can achieve a brighter, healthier smile.
Professor Alan Templeton | When Fire Sparks Ecological Opportunity and Habitat Restoration
How far would you be willing to go to save an endangered species? Would you consider burning part of a forest as a solution? As unconventional as it may sound, conservationists sometimes resort to such measures to restore lost habitats. One remarkable example is the efforts to save eastern collared lizards – and indeed the entire biological community in which they live – in the Ozarks, spearheaded by American geneticist and statistician Professor Alan Templeton of Washington University in St Louis, USA.
Dr Toby Phesse | Revealing the Mysteries of Wnt Signalling: Novel Approaches to Beating Cancer
Cancer remains a leading cause of mortality worldwide, and the need for new, more effective treatments remains an urgent challenge. Dr Toby Phesse from Cardiff University in the UK focuses on the role of the Wnt receptor found on the surface of cells and its involvement with cell communication and cancer growth, bringing fresh hopes for new therapeutic options.
Dr Vijay Reddy | The Virus World Database: An Invaluable Resource for Public Health and Healthcare
Severe viral disease presents an ongoing challenge to the health of humankind. While unparalleled developments in science and technology are improving our understanding of such viruses, this information needs to be readily accessible to researchers to ensure continued progress in public health and healthcare. Dr Vijay Reddy and his colleagues at the Hormel Institute (University of Minnesota) developed the Virus World database, an invaluable resource that details the genome, structure, and host of practically every discovered virus to date.