Cylerus: An Innovative Approach to Vascular Drug Delivery
Prosthetic vascular grafts for dialysis access have a limited lifespan and usefulness due to inflammation, infection and especially blood vessel narrowing at the site of graft implantation. Consequently, patients need repeated surgeries to revise or replace the vascular conduits, which is an expensive and difficult procedure. Cylerus is an innovative company dedicated to developing new methods of drug delivery to prosthetic vascular grafts that has developed a novel medical device called the Drug-Eluting Cuff (DEC). The DEC significantly increases the lifetime of these critical access sites while decreasing costs and the harmful consequences to patients of repeated interventions to maintain graft function (patency) to support life-sustaining hemodialysis.
Patients with end-stage renal disease (ESRD) must undergo hemodialysis – filtering of the blood – to eliminate toxins and waste products from their circulation two to three times per week. Currently, there are over 400,000 patients requiring multi-weekly dialysis sessions in the United States and over one million worldwide. During dialysis, a dialyser and a special filter, called an artificial kidney, are used to clean the blood, taking over the function of diseased kidneys that no longer function or work effectively. To enable routine dialysis, the patient undergoes a minor surgical procedure in which vascular access is established that can be used to connect the patient’s circulation to the dialyser.
Choices for establishing chronic vascular (blood vessel) access are limited. Currently, the most common access utilised is a fistula – vascular access is made by joining an artery and a vein in the lower or upper arm. A fistula is considered the best choice by many healthcare professionals and patients; however, fistulas cannot be used unless the patient has healthy veins and it can take months for arteriovenous fistulas (AVFs) to mature. Furthermore, about half of AVFs fail to achieve adequate blood flow to sustain effective hemodialysis, requiring further intervention in the patient to establish vascular access.
Another option for ESRD patients to establish vascular access is implantation of a prosthetic (synthetic) graft. This is accomplished by using a soft piece of tubing-like material called ePTFE (expanded polytetrafluoroethylene), known as Teflon® in the unexpanded state, which provides a new blood-carrying conduit between an artery and a vein. The option of utilising a prosthetic graft is attractive as it can be used for dialysis much sooner than an AVF. However, the primary patency (or continued function) of prosthetic grafts is poor as three months after implantation, almost half of these grafts require re-intervention to maintain adequate blood flow to support hemodialysis.
Generally, an obstruction occurs at the outflow tract of the graft due to the abnormal accumulation of smooth muscle cells and connective tissue near the site where the graft is connected to the vein (the anastomosis site). Thus, to re-establish blood flow mechanical (thrombectomy) and pharmacological (thrombolysis) tools are employed to achieve patency and regain the grafts therapeutic function – this is defined by the practitioner as adequate blood flow to perform dialysis.
As vascular access sites fail, either AVF or prosthetic, ESRD patients need to undergo repeated surgical procedures in order to revise, fix or create a new access site. In doing so they face associated risks and complications such as blood clots (thrombosis), central vein stenosis (abnormal narrowing of the blood vessel due to trauma and inflammation) and infection. A number of mechanical approaches, such as the use of hooded ePTFE, and pharmacological, such as heparin-coated grafts, have been used in attempts to prevent or reduce the aggressive proliferation and migration of cells that can occur at the graft site. However, these methods have been mostly unsuccessful. The costs of keeping grafts functioning in dialysis patients are staggering and now exceed $1 billion annually in the US alone.
The Drug-Eluting Cuff Synthetic Graft
The drug delivery Company, Cylerus, aims to improve the utility and functionality of prosthetic vascular grafts with their novel Drug-Eluting Cuff (DEC) delivery device. Cylerus, Inc. was formed in 2007 by Dr Stephen Hanson, a professor and previous chair of the Biomedical Engineering Department at Oregon Health & Science University in Portland Oregon. His goal was to develop new drug delivery methods for increasing the longevity and functionality of prosthetic vascular grafts.
Based in Melbourne, Florida, Cylerus specialises in vascular drug delivery innovations and has developed the DEC that can be combined with an implantable drug pump, and the FDA approved drug, sirolimus. This drug-device combination is intended to prevent cell proliferation at the site where a prosthetic graft is implanted in patients. The DEC is a product of many years of research that originated in Dr Hanson’s laboratory at Emory University in Atlanta, GA and subsequently at Oregon Health & Science University (OHSU) in Portland, OR.
Cylerus is headed by President and CEO, Dr Ronald J. Shebuski, Sr, who leads the research and development effort to develop a formulation of sirolimus that is biocompatible with the DEC, catheter delivery tubing and drug pump. Dr Shebuski oversees the preclinical safety and efficacy studies of the DEC sirolimus combination that are the first steps to be completed before the device can be tested in ESRD patients. The DEC is being developed to prevent narrowing downstream of the prosthetic graft implantation site and may lead to more prosthetic graft usage if failure rates can be diminished significantly by the Cylerus approach. This could potentially swing the balance of vascular access from AVFs to greater use of DECs and prosthetic vascular grafts.
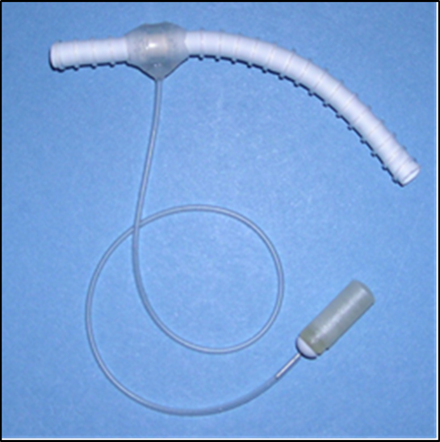
The DEC, manufactured for Cylerus by a contract research organisation, utilises standard wall six mm ringed or non-ringed ePTFE as the starting material and the other components are all medical grade silicone. A standard medical grade catheter connects the DEC to a drug-filled osmotic pump to deliver sirolimus to the interior of the graft in a continuous and circumferential manner. The drug pump delivers sirolimus – a cytostatic drug that interrupts the cell cycle and thus prevents excessive cellular proliferation – in a continuous manner. The drug pump can be refilled or replaced on a monthly or quarterly basis.
Sirolimus is delivered through the porous wall of the graft and to the immediate surrounding area, where it remains highly concentrated, preventing cellular and tissue build-up around the connection (i.e. anastomosis) between the graft and the vein. The specialised design of the DEC allows sirolimus to remain concentrated at the graft site while minimising its concentration in the systemic circulation. This is important as sirolimus is also FDA approved as an immunosuppressant drug (Rapamune®) for kidney transplant patients.
However, local DEC delivery of sirolimus provides pharmacologically effective concentrations at and downstream of the graft anastomotic site where it’s needed, but systemic levels will be well below immunosuppressive levels of the drug (this has already been demonstrated preclinically). The total amount of sirolimus to be administered to the vascular access site by the Cylerus DEC is approximated at 1-2 mg/month. In comparison, Rapamune® is administered orally at a dose of 2-4 mg/day. The other FDA approved product that contained sirolimus (< 1 mg), to effectively prevent smooth muscle cell proliferation in drug-eluting coronary stents, was the CypherTM stent.
The DEC and drug pump can be individually manipulated without the prosthetic graft material and may potentially be used for medical applications aside from hemodialysis. For example, the DEC has the potential to deliver different types (drugs, cells or genes) and concentrations of therapeutic agents singly, in combination or sequentially. The delivery of the drug can also be adjusted or stopped, depending on the needs of the patient.
Promising Results
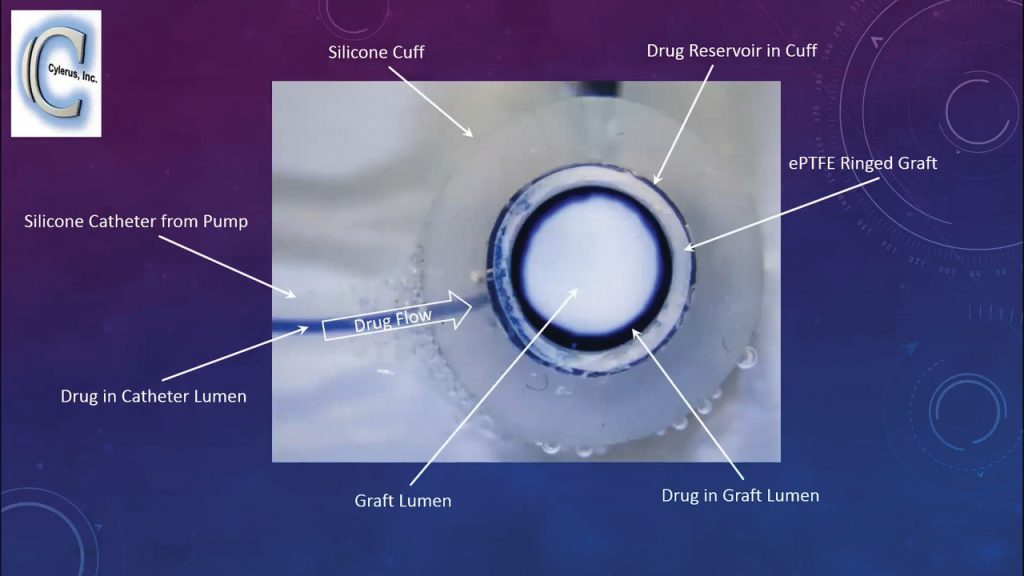
Dr Prabir Roy-Chaudhury, the Chief of Nephrology at the University of Arizona at Tucson, is a co-principal investigator for the project with Dr Shebuski and brings extensive experience in kidney failure, vascular access and hemodialysis to the Company. Dr Roy-Chaudhury has evaluated the DEC and demonstrated efficient drug delivery through the ePTFE graft wall using a blue dye as a drug surrogate (top figure on the previous page). The DEC provides the drug (sirolimus) to the inside wall of the vascular conduit in a continuous and circumferential manner.
Furthermore, the blood flow in a tube or cylinder is fastest in the middle of the bloodstream and slowest near the vascular wall, where sirolimus is entering the conduit. Thus, the flow dynamics are preferential for sirolimus to have intimate contact with the vascular wall where the smooth muscle cell accumulation occurs. Importantly, sirolimus concentrates at the graft anastomotic site and flows in a continuous and circumferential manner to the inner and downstream wall of the prosthetic graft site and so is an extremely effective means of drug delivery. A high local concentration of the drug is provided where you need it – continuously.
In earlier studies, researchers in the laboratory of Dr Hanson tested the DEC prosthetic graft device in a non-human primate model by measuring blood vessel thickening after 30 days. His research team implanted femoral arterial interpositional prosthetic ePTFE grafts on both sides of the groin. Sirolimus was then infused slowly (2 mg/month) to the graft on one side and vehicle saline on the other, and the results were compared. The researchers found an 80% reduction in smooth muscle cell proliferation at 30 days following graft implantation on the graft side that had been slowly infused with a very low dose of sirolimus compared to the graft side exposed to saline only.
Porous ePTFE membrane under an electron microscope.
To further test the sirolimus formulation, Dr Shebuski’s team conducted a study in pigs over a period of 28 days where the drug was delivered continuously through osmotic mini-pumps at doses of 0.5 and 1.5 mg/month. The researchers found that the drug was stable for 28 days, maintaining a high concentration at the infusion site and a low concentration in the systemic circulation. The researchers now plan to carry out longer-term studies up to 90 days in length, in order to demonstrate the safety and effectiveness of the body temperature stable sirolimus formulation on smooth muscle cell accumulation in the gold standard porcine AV prosthetic graft model in Dr Roy-Chaudhury’s laboratory at the University of Arizona with the assistance of Dr Diego Celdran, an expert vascular large animal veterinary surgeon and scientist.
Other drug delivery systems do exist – for example, drug-eluting balloons and drug-polymer reservoirs. However, these therapies can only deliver small doses of the drug at diminishing concentrations over short periods of time. In addition, they are generally delivered during the graft implantation surgery. Cylerus relies upon 1) the porosity of ePTFE, 2) the known action of sirolimus at very low concentrations to act as an anti-proliferative and 3) the ability to continuously deliver drug via a refillable and replaceable drug pump. This provides a system that will be more effective, safer and cheaper compared to other methods attempted thus far to maintain the utility of prosthetic vascular grafts.
The Cylerus DEC device has been shown to deliver sirolimus continuously at stable concentrations and is applicable to any porous prosthetic graft. Synthetic ePTFE grafts are also used in vascular reconstructions in diabetic patients and in those with peripheral arterial disease. Cylerus’s DEC device could become the best choice for these patients, as well as for those with small vessels and diseased or defective blood vessels requiring prosthetic replacements.
The Future of Vascular Drug Delivery
Cylerus has developed a proprietary, long-acting, body temperature stable formulation of sirolimus and will soon complete preclinical safety and further efficacy testing in preparation for human clinical trials. The Company is currently Phase I/II Fast-Track SBIR funded by the National Institute on Aging (NIA), part of the National Institutes of Health (NIH), to complete preclinical studies with plans to file an IND (Investigational New Drug – IND) application with the FDA in 2020 to initiate Phase II clinical studies in the US.
The Company is now in the process of applying for further funding for continued pre-IND studies (Good Laboratory Practice – safety and biocompatibility study), regulatory document preparation and the start of human Phase II clinical trials. Cylerus is seeking a potential commercial partner to support further development activities and is applying for follow-on NIH grants to the currently funded Phase I/II Fast-Track SBIR (NIA) to support the clinical program to a large degree.
The novel Cylerus DEC is projected to reach 50% market share after five years, with roughly 75% of sales being vascular access and maintenance providers, and the remainder of sales to the peripheral vascular disease market. As Cylerus continues to progress in their efforts to bring this novel prosthetic graft saving technology to human trials, patients with ESRD will be one step closer to a better quality of life. The DEC will allow for improved sustainability of prosthetic vascular grafts leading to fewer re-intervention surgeries, markedly decreased healthcare costs, and an increased overall quality of life for ESRD patients.
Meet the Team
 Dr Stephen R. Hanson
Dr Stephen R. Hanson
Dr Stephen R. Hanson is the Founder and Board Chairman of Cylerus and holds a PhD in Chemical Engineering from the University of Washington in Seattle, USA. From 2003 until 2012, he was a Professor of Surgery and a Professor in the Department of Biomedical Engineering at the Oregon Health Science University in Portland, Oregon. There, his research focused on preventing vascular graft failure. He produced the first generation of local drug delivery devices and completed numerous drug studies focusing on the prevention of blood vessel thickening following injury. In 2007, he founded Cylerus in order to bring his novel drug delivery technology and approach to market.
 Dr Prabir Roy-Chaudhury
Dr Prabir Roy-Chaudhury
Dr Prabir Roy-Chaudhury is currently the Division Director for Nephrology and the Director of the Kidney and Vascular Centre at the University of Arizona, where his research focuses on uremic vascular biology and dialysis vascular access. Previously, he was a Medical Director at the University of Cincinnati Health Vascular Access Centre and a Professor in the Division of Nephrology at the University of Cincinnati College of Medicine. He holds a PhD in renal medicine from the University of Aberdeen in Scotland and an MD from the Royal College of Physicians.
E: proychaudhury@deptofmed.arizona.edu
 Dr Diego Celdran
Dr Diego Celdran
Dr Diego Celdran is a veterinary surgeon (DVM) and associate scientist (PhD) at the University of Arizona working in Dr Prabir Roy-Chaudhury’s laboratory. He is an expert in large animal vascular surgery and is responsible for the placement of AV grafts in large animal models of prosthetic vascular graft safety and efficacy. Dr Celdran was trained at the College of Veterinary Medicine, Caceres, Estremadura Spain.
E: dceldran@deptofmed.arizona.edu
 Dr Ronald J. Shebuski, Sr
Dr Ronald J. Shebuski, Sr
Dr Ronald J. Shebuski has been the President and CEO of Cylerus since 2010. He completed his PhD in Pharmacology at the University of Minnesota Medical School in Minneapolis in 1985. He has led successful drug development teams at several large US-based pharmaceutical companies and co-founded a number of start-up companies focusing on vascular biology. Dr Shebuski has extensive drug development experience in the commercial sector and also served as a Special Government Employee to the Center for Drug Evaluation and Research (CDER) at the FDA for two terms.
KEY COLLABORATORS
The University of Arizona
FUNDING
National Institute on Aging (NIA) – Phase I/II Fast Track SBIR Grant
WEBSITE: