Dr Annarita Di Lorenzo – Sphingolipids: Fats that Protect the Heart
Despite current therapies, cardiovascular disease is a leading cause of death worldwide, suggesting that alternative drug targets are urgently needed to preserve cardiovascular health. Sphingolipids – a class of biologically active molecules and components of cell membranes – are a potential target, but how their synthesis is regulated in cardiac disease remains an elusive mystery. Dr Annarita Di Lorenzo and her group at Weill Cornell Medicine have begun to elucidate the molecular mechanisms regulating this pathway, which may lead to novel therapeutic treatment of cardiovascular diseases.
The Need for Alternative Targets in Cardiovascular Disease
Cardiovascular diseases represent a huge global disease burden, and are a leading cause of death worldwide – approximately 63% of global deaths are due to non-transmissible diseases, with half of those attributed to cardiovascular diseases (30%). One of the major risks factors for cardiovascular disease is high blood pressure or hypertension. Hypertension causes approximately 7.5 million global deaths annually and nearly 1 billion people are estimated to be affected. In addition, it has been linked to a higher risk of coronary heart disease and stroke. Despite the use of drugs to control hypertension, it still remains one of the most common causes of heart failure, kidney impairment and stroke, suggesting alternative drug targets are needed for therapeutic treatment.
Physiologically, the dysfunction of the endothelium, which is made up of endothelial cells that line the inner surface of blood and lymphatic vessels, has been linked to hypertension. In healthy individuals, the endothelium is a complex system that is crucial for the maintenance of vascular homeostasis, functioning through cell signalling in response to various physical, biological and chemical stimuli. Moreover, endothelial cell dysfunction is an early event contributing to the disruption of vascular tone, or the partial contraction of the arteries to maintain blood flow, and thus, regulate blood pressure.
Sphingolipids – Bioactive Molecules with a Role in Cardiovascular Dysfunction
In terms of biological stimuli, endothelium-released nitric oxide is known to be crucial for the regulation of blood pressure. However, sphingolipids are also emerging as biologically active molecules with cardiovascular effects, including blood pressure control. Sphingolipids are a class of lipids with a characteristic 18-carbon amino-alcohol backbone referred to as the sphingoid base or sphingosine. First discovered from brain extracts in the 1870s, they were named after the mythological Sphinx because of their enigmatic nature. Their complexity arises from the various chemical groups that can be attached to sphingosines. The most well-characterised sphingolipid is sphingosine-1-phosphate, created by adding a phosphate chemical group to sphingosine.
Functionally, sphingolipids are essential structural components of the cell membrane, as well as being bioactive signalling molecules. They are involved in many cellular processes such as cell stress responses, cell survival, differentiation, migration and inflammation. They are synthesised through the sphingolipid de novo (from scratch) biosynthesis pathway in the endoplasmic reticulum – the largest organelle in eukaryotic cells. Although the synthetic pathway is well-defined, little is known about how this pathway is regulated during physiological or hypertensive conditions. Hence, Dr Annarita Di Lorenzo and her research group sought to uncover this mystery.
Dr Di Lorenzo’s research on endothelial cell dysfunction first began during her PhD at the University of Naples, Italy. Building on her PhD training, she joined the lab of Dr William Sessa at Yale Medical School for her postdoctoral training in 2006, where she continued to examine the mechanisms of endothelial cell dysfunction. One line of research led her to study Nogo-B – a membrane protein highly expressed in the walls of blood vessels. This study unveiled an important role of Nogo-B in blood vessels in the development of inflammation. However, how Nogo-B influenced the onset of inflammation was still unknown.
The research in her laboratory at Weill Cornell eventually led to the major discovery that Nogo-B is a negative regulator of sphingolipid biosynthesis, which has implications for cardiovascular functions and treatments.
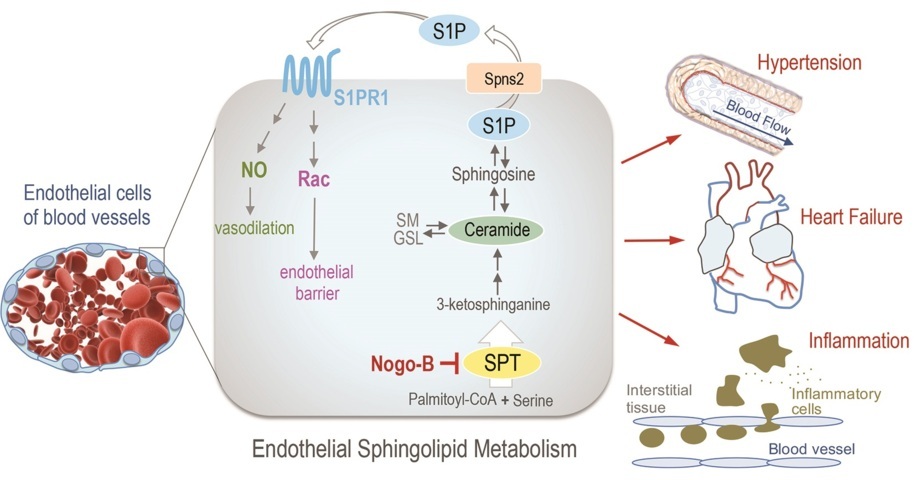
Nogo-B-mediated inhibition of sphingolipid biosynthesis plays an important role in the pathogenesis of hypertension, heart failure and inflammation.
Nogo-B as a Therapeutic Target to Control Hypertension
Through a series of experiments in vitro and in vivo, Dr Di Lorenzo and her research team at Weill Cornell Medicine demonstrated that Nogo-B negatively regulates sphingolipid de novo synthesis by inhibiting serine palmitoyltransferase (SPT) – the first and rate limiting enzyme of the sphingolipid synthesis pathway. The Nogo-B protein has two functionally similar variants, called Nogo-A and Nogo-C, which are abundantly expressed in the central nervous system, and Nogo-C is also found in skeletal muscle. On the other hand, Nogo-B is highly (but not exclusively) expressed in endothelial cells.
Specifically, Dr Di Lorenzo and her colleagues found that mice without Nogo-A and Nogo-B variants (also known as Nogo-A/B-deficient mice), had markedly lower blood pressure compared to control (or wild-type) mice. Since nitric oxide lowers blood pressure, not surprisingly, Dr Di Lorenzo also found that nitric oxide production was notably increased in the blood and in mesenteric arteries (arteries that drain from the aorta) of Nogo-A/B-deficient mice compared to controls. Therefore, Nogo-B may be responsible for activating endothelial nitric oxide synthase (NOS) – the enzyme that catalyses the formation of nitric oxide – to increase nitric oxide production and alter blood pressure.
Since the locations of Nogo-B and endothelial NOS in the cells were different, Dr Di Lorenzo and her group hypothesised that these two proteins did not interact directly. Rather, they believed that Nogo-B was likely to regulate sphingolipid synthesis, as one of the sphingolipids, S1P, is known to be vastly important in controlling blood vessel function. Indeed, Nogo-B was found to interact with SPTLC1 – a subunit of the SPT enzyme – in cells from mouse lungs and lung lysates (disintegrated lung cells), suggesting an interaction between these proteins. In addition, sphingosine-1-phosphate (S1P) was suppressed in both Nogo-A/B-deficient and control cells isolated from mouse lung when SPT – the rate-limiting enzyme of sphingosine synthesis – was inhibited by the compound myriocin. These findings indicate that Nogo-B also exerts its effects through inhibiting SPT. Thus, in the absence of Nogo-B, the inhibition of SPT is relieved, and sphingolipid and nitric oxide production in the cell is increased, thereby lowering blood pressure.
Most importantly, Dr Di Lorenzo and her group found that Nogo-B mediated vascular dysfunction in a mammalian model of hypertension. When Nogo-A/B-deficient mice were continuously infused with the peptide hormone angiotensin II, wild-type mice showed sustained levels of hypertension. In contrast, it only induced a mild increase in blood pressure in Nogo-A/B-deficient mice. However, if Nogo-A/B-deficient mice were treated with myriocin, their blood pressure returned to the levels observed in wild-type angiotensin II treated mice. Not only do these findings indicate that Nogo-B inhibits SPT, leading to higher blood pressure, it also demonstrates that Nogo-B, SPT and the sphingolipid synthesis pathway can be used as novel pharmacological targets to regulate blood pressure.
Can Inhibiting Nogo-B Protect the Heart Against Cardiac Dysfunction?
Apart from hypertension, cardiac hypertrophy – an increase in the mass of the heart due to pathological stimuli – can also lead to cardiac dysfunction. Cardiac hypertrophy can be induced by pressure overload, but not all hypertrophy is considered detrimental. The mechanism behind adaptive and pathological hypertrophy remains poorly understood. To this end, Dr Di Lorenzo and her group found that Nogo-B regulated sphingolipid synthesis within the endothelium to protect the heart in a mouse model of cardiac failure. The team established this mouse model by surgically tightening the aortas in the mice – a procedure called transverse aortic constriction (TAC). In sham-operated mice, who underwent ‘placebo surgery’ so they could be used as an accurate control, nylon string was only tied loosely around their aortas.
Following TAC, the control mice developed cardiac hypertrophy after 2 weeks, with a 2-fold increase in heart size reached at 3 months. In contrast, the heart size of Nogo-A/B-deficient mice was reduced compared to sham-operated controls and in mice at 2 weeks and 3 months post-TAC. The absence of Nogo-A/B did not greatly alter the magnitude of hypertrophy. However, the cross-sectional areas of cardiac muscle cells in Nogo-A/B-deficient mice were significantly smaller than those of the control TAC-operated mice. Moreover, ultrasound results revealed that Nogo-A/B deficiency protected the heart from failure 3 months post-TAC.
Cardiac inflammation is intimately linked to hypertension and scarring, which can compromise the heart’s pumping capacity and oxygen diffusion to hypertrophied cardiac tissue. Notably, Dr Di Lorenzo’s team found that the Nogo-A/B-deficient mice were resistant to pressure-induced inflammation and scarring at 2 weeks and 3 months following TAC. Mice lacking Nogo-B specifically in endothelial cells were also similarly resistant to pressure-induced vascular permeability and inflammation compared to control mice. Cardiac function was also preserved in these mice 3 months after TAC. These findings suggest that the loss of Nogo-A/B may sustain cardiac muscle function in response to long-term pressure overload by maintaining cardiac cell function and reducing vascular permeability and inflammation.
Next, Dr Di Lorenzo and her group investigated whether SPT activity was altered following TAC. As expected, in the absence of Nogo-A/B, SPT activity was notably increased in sham-operated hearts and in hearts 3 days post-TAC. Critically, when SPT activity was inhibited with myriocin 1 day before, on the day of and 2 days following TAC, vascular permeability and inflammation from pressure overload in Nogo-A/B-deficient mice were elevated and returned to control levels compared to wild-type mice. Further ultrasound tests revealed that the heart in these Nogo-A/B-deficient mice were no longer protected – in fact, cardiac dysfunction had been re-established to wild-type levels. Taken together, the inhibition of SPT mediated by Nogo-B plays a crucial role in pressure overload-induced cardiac dysfunction. Therefore, Nogo-B and the sphingolipid synthetic pathway may also be targeted to protect the heart from long-term cardiac dysfunction.
Future Perspectives
Although the sphingolipid synthesis pathway had previously been well-defined, limited was known about its effects on blood pressure and the mechanisms underlying its regulation in physiological and hypertensive conditions. In view of this, the innovative work of Dr Di Lorenzo and her team has shed light on the crucial regulatory role of Nogo-B-mediated sphingolipid synthesis in hypertension and pressure overload-induced cardiac hypertrophy. Specifically, her team found that Nogo-B is a negative regulator of SPT and subsequent decrease in sphingolipid synthesis leads to an increase in blood pressure. Nogo-B also regulates cardiac cell function and its absence protects the heart from dysfunction in response to long-term pressure overload. These observations also suggest that sphingolipid synthesis may play a causative role in development of cardiovascular diseases.
Nevertheless, some open questions about the regulation of sphingolipid synthesis remain. For example, our understanding of the molecular mechanisms relieving the inhibitory actions of Nogo-B on SPT and the structure of SPT is currently incomplete. And what about the role of SPT and sphingolipid synthesis in other diseases? The research of Dr Di Lorenzo and her group forms a solid framework to investigate the role of sphingolipid synthesis in other pathological conditions associated with lack of Nogo-B, such as asthma, liver scarring and inflammation. The team’s seminal findings also provide compelling evidence that Nogo-B and the sphingolipid synthesis pathway are promising therapeutic targets for the treatment of hypertension and cardiac hypertrophy. Given the involvement of sphingolipids in numerous cellular processes, it would not be surprising for Dr Di Lorenzo’s team to discover that modulating this pathway may impact therapeutic treatment of many human diseases in the near future.
Meet the researcher

Dr Annarita Di Lorenzo
Department of Pathology and Laboratory Medicine
Weill Cornell Medicine
Cornell University
New York, NY
USA
Dr Annarita Di Lorenzo received her PhD in pharmacology and vascular biology in 2004 under the supervision of Drs Maria Rosaria Bucci and Giuseppe Cirino at the University of Naples ‘Federico II’ Italy. She went on to complete her postdoctoral training at Yale Medical School in the United States between 2006 and 2011. Subsequently, she established her own laboratory in the Center of Vascular Biology directed by Dr Timothy Hla, in Department of Pathology and Laboratory Medicine at Weill Cornell Medical College, where she studies the role of sphingolipid signalling in cardiovascular function and disease. Her research has been published in high profile journals including Science Signalling, PNAS, Hypertension and Nature Medicine. She is also a member of the Editorial Advisory Board of the Journal of Pharmacology and Experimental Therapeutics.
CONTACT
E: and2039@med.cornell.edu
T: (+1) 212 746 6476
W: http://vivo.med.cornell.edu/display/cwid-and2039
KEY COLLABORATORS
Dr Timothy Hla, Harvard Medical School, Boston, USA
Dr William Holland, UT Southwestern Medical Center, Dallas, USA
Dr Bodo Levkau, University Hospital Essen, Germany
Dr Giuseppe Faraco, Weill Cornell Medicine, New York, USA
Dr Teresa Sanchez, Weill Cornell Medicine, New York, USA
Dr Costantino Iadecola, Weill Cornell Medicine, New York, USA
Dr Maria Rosaria Bucci, University of Naples ‘Federico II’, Italy
Dr Luigi Gnudi, King’s College London, London, UK
Dr Carl Blobel, HSS Research Institute, New York, USA
Dr Xian-Cheng Jiang, SUNY Downstate Medical Center, Brooklyn, NY, USA
Dr Mario Gaudino, Weill Cornell Medicine, New York, USA
Dr Alessio Accardi, Weill Cornell Medicine, New York, USA
Dr Matthew E. Fink, Weill Cornell Medicine, New York, USA
Dr Richard L. Proia, NIDDK, Bethesda, MD USA
FUNDING
NIH/NHLBI
REFERENCES
L Sasset, Y Zhang, TM Dunn and A Di Lorenzo, Sphingolipid De Novo Biosynthesis: A Rheostat of Cardiovascular Homeostasis, Trends in Endocrinology & Metabolism, 2016, 27, 807–819.
Y Zhang, Y Huang, A Cantalupo, PS Azevedo, M Siragusa, J Bielawski, FJ Giordano and A Di Lorenzo, Endothelial Nogo-B regulates sphingolipid biosynthesis to promote pathological cardiac hypertrophy during chronic pressure overload, JCI Insight, 2016, 1, e85484.
A Cantalupo, Y Zhang, M Kothiya, S Galvani, H Obinata, M Bucci, FJ Giordano, X-C Jiang, T Hla and A Di Lorenzo, Nogo-B regulates endothelial sphingolipid homeostasis to control vascular function and blood pressure, Nature Medicine, 2015, 21, 1028–1037.