Dr Diana P. Bratu – Real-Time Visualisation Of Mrna Regulation And Transport
Dr Diana Bratu’s laboratory utilises biophotonics to image the movement of mRNA and the proteins it interacts with during the development of the fruit fly egg, informing research ranging from the basics of mRNA transport to therapeutic development.
The Importance of mRNA Regulation
Since graduate school, Dr Diana Bratu has been developing technologies to visualise the movement of RNA and proteins within the cell. When asked what attracted her to this particular field of research, she replies ‘simply the curiosity of understanding the underlying mechanisms that drive development’. However, research on intracellular nucleic acid transport informs a much wider range of topics than growth and development – from the progression of Alzheimer’s to the development of pharmaceuticals targeting proteins to treat HIV infection.
Eukaryotic messenger RNA (mRNA) is the critical intermediary molecule between DNA and protein as it is translated in the ribosome. The regulation of such mRNA by co-factor proteins is critical to controlling gene expression, having implications for research in embryonic development, neurodegenerative disorders, long-term memory, and learning processes.
As such, research efforts to better understand the regulation of mRNA are afoot worldwide, with one of the key challenges being the visualisation of mRNA and how it interfaces with these proteins to influence the expression of important genes. Dr Bratu and colleagues are pioneering this field through a gamut of unique biophotonic methods, from probe design to advanced imaging approaches, thus improving upon the detection and accuracy of mRNA visualisation and its co-localisation with trans-acting proteins important for the normal function of processes within a cell.
‘It was only natural for me to follow the field of RNA biology and develop my own research group exploring the intricate processes that make up the life-cycle of an RNA molecule. Visualising these processes is the most attractive aspect of this field!’
Using Drosophila melanogaster (the fruit fly) as a model organism, Dr Bratu’s research group employs fluorescent probes and spinning disc confocal microscopy to track the movement of mRNA and proteins throughout the egg chamber. They employ genetically encoded fluorescent proteins and short molecular probes (i.e. molecular beacons) allowing for the detection of various endogenous proteins and mRNAs, thus enabling real-time tracking of these molecules as they are transported within the cell. While this has obvious implications for the research of oogenesis, these studies act as a biologic proof of principle to guide other researchers’ studies examining mRNA transport in other systems. By combining molecular, genetic, biochemical, and advanced imaging approaches, Dr Bratu seeks to inform the scientific community on RNA transport machinery and metabolism while maintaining a strong mentorship program for undergraduate students interested in pursuing research in the biological sciences especially in biophotonics.
Real-time Visualisation of mRNA-Protein Complex Movement
The egg chamber of D. melanogaster is an ideal system to study mRNA transport, as it is a complex tissue yet easy to manipulate in the lab and stable enough to withstand longterm imaging. While there are countless types of mRNA whose localisation is important for the proper growth of the oocyte, Dr Bratu and colleagues initially selected oskar mRNA as the subject of their research. This mRNA is transcribed within the nurse cell nuclei in the egg chamber and then transported to the oocyte’s posterior on a railway of microtubule tracks. It is not translationally active until it is successfully transported to the posterior. Upon translation, the Oskar protein is anchored at the posterior pole and plays a critical role in establishing the body axis of the developing embryo, as well as in determining the future germline.
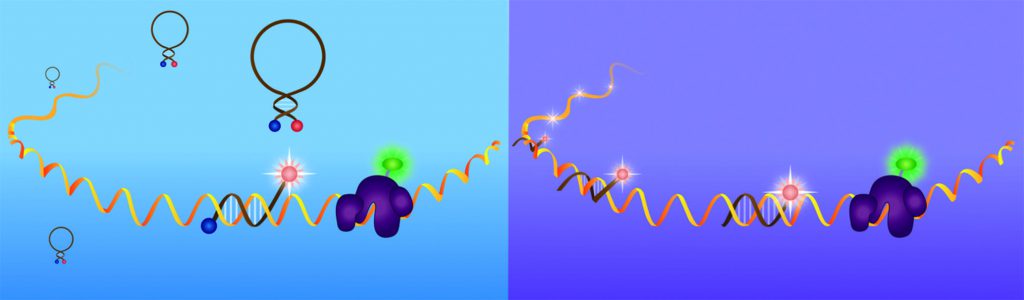
During oskar mRNA transport to the posterior pole, it associates with several proteins, among which are Bruno and Cup. These two proteins work together to suppress translation of oskar mRNA throughout this process. Livia Bayer, a PhD student in Dr Bratu’s lab is able to track these mRNA protein (mRNP) complexes within the nurse cells and the oocyte, by employing a combination of techniques to visualise these proteins with fluorescent tags and the transcript when hybridised by fluorescent probes.
For visualising mRNAs, the team designs, synthesises and delivers into the egg chambers, fluorescently labelled, small, hairpin-shaped oligonucleotides called molecular beacons, which hybridise with high specificity to endogenously expressed mRNAs and emit a fluorescent signal. The team selects target regions within an mRNA utilising two computer algorithms, mfold and OligoWalk, which assist them in narrowing down sequence stretches for the highest affinity and accessibility results in vivo. The probes are then synthesised from modified phosphoramidites, which render the probes stable within the cellular environment, and labelled with bright fluorophores that provide a great signal for detection of the dynamic processes involved during an mRNA’s lifecycle. This allows for the visualisation of oskar mRNA transport to the posterior pole of the fruit fly oocyte and movement throughout the egg chamber over extended periods of time.
This technology will provide unique advancements in this field, as they are ideal for detecting very small targets, but also sensitive enough to identify them when they are in small quantities. Shorter probes have also been developed in Dr Bratu’s lab that can detect the smallest of targets, such as microRNAs.
The Role of microRNAs
MicroRNAs have received much attention in recent years, as their roles in both development and disease have become more evident. Because of their ability to downregulate, or silence, gene expression, they hold great potential for future drug development. MicroRNAs belong to the largest family of noncoding RNAs; they are not translated into protein, but rather directly influence the expression of other genes by binding to the mRNA and either repressing translation or destroying it.
In the team’s most recent investigations into the role of microRNAs, via a study led by PhD student John McLaughlin, they have identified a conserved binding site for a particular microRNA, miR-305, on the maternal mRNA bicoid. They are currently investigating the in vivo function of miR305 in the translational repression of this maternal mRNA during D. melanogaster oogenesis. Interestingly, the team’s cell culture experiments have so far shown that miR-305 can inhibit the translation of a bicoid mRNA reporter gene.
mRNA post-transcriptional regulation can also occur in organelles known as Processing Bodies (P-bodies), which have known roles in microRNA function, mRNA storage, decay, and translational repression. With this in mind, the team hypothesise that recruitment to P-bodies helps to stabilise bicoid mRNA during oogenesis, and this process may involve the microRNA machinery in mediating its translational repression.
Untangling the Intracellular Vesicular Trafficking of RNA Viruses
A project that Dr Bratu and her Senior Research Associate, Dr Irina Catrina, have undertaken as of late is elucidating the role of certain host proteins in viral RNA transport. The life cycles of RNA viruses, including HIV and Influenza A, rely heavily upon the transportation of genomic RNAs, from the nucleus into the cytoplasm, where translation or packaging occurs. Proteins involved in viral RNA transport during replication are of great interest as pharmacological intervention targets.
The host protein AGFG1 is necessary for HIV-1 and Influenza A replication, but not for cell viability, making it a promising candidate as an antiviral drug target. While it is understood that AGFG1 is involved in the movement of viral RNAs from the perinuclear region to the plasma membrane, elucidation of the cellular role(s) of AGFG1 and how it interacts with other proteins within the cell will allow for more efficient and accurate drug design. It has been shown that AGFG1 participates in the uptake of select cargo via endocytosis, however this does not fully account for the defects observed in viral RNA transport and localisation. Drs Bratu and Catrina set out to identify the role(s) this protein plays in cytoplasmic transport.
Once again using D. melanogaster egg chambers, they study the fruit fly homolog of AGFG1 known as Drongo, in particular the effect of reduced Drongo protein levels on development of the egg. The results may then be extrapolated to highlight the actions of AGFG1 in human cells. Using fluorescence and real-time tracking during oogenesis, they determined that Drongo co-localises at the oocyte cortex with Clathrin, a major player in vesicle formation, and F-actin, a critical cystoskeletal component during vesicular transport. This association suggests that Drongo plays a role in endocytosis, much like its mammalian homolog, AGFG1. Dr Catrina determined that Drongo also co-localises with endosome markers Rab5 and Rab11, both of which play roles in endosome trafficking, and that proper localisation of Drongo requires functional Rab11 protein. By confirming the importance of Drongo/ AGFG1 in intracellular vesicular trafficking, the team has provided valuable information that may guide the development of novel antiviral drugs that target conserved interactions between viruses and host co-factors.
Creating the Next Generation of Scientists
Dr Bratu has harnessed her knowledge in the use of molecular beacons and advanced live cell imaging to excite many cohorts of young scientists about visualising these processes that drive development and have such wide implications for the understanding of disease progression and pharmaceutical development. She pioneered a laboratory course for upper level undergraduates which has since been expanded to multiple affiliated institutions and is now offered to graduate students entitled, ‘Laboratory in Fine Cell Structure: Fluorescence Imaging’ as well as a seminar course entitled ‘MicroRNAs and Development’, both of which are always fully enrolled. The microscopy course introduces students directly to the many uses of light to monitor cellular events, ranging from confocal microscopy to high-resolution imaging (i.e. structural illumination microscopy). Students have the opportunity to spend time at the Bio-Imaging Facility at Hunter College which houses several advanced imaging set ups (fluorescence microscopes, laser scanning and spinning disc confocal microscopes, SIM/TIRF microscope). Under Dr Bratu’s leadership as Scientific Director of this facility, several new microscopy set-ups have been acquired, thus improving the imaging capabilities available for her students and colleagues.
![]()
Using D. melanogaster and various fixation techniques, students learn the basics of fluorescent probe design and fly husbandry while generating images in which they are able to visualise cellular structures. The students then present their findings at local symposia, and many submit their images to various microscopy competitions, such as the annual ‘Nikon’s Small World: Photomicrography Competition’. Furthermore, these courses teach students to think critically as they analyse peer-reviewed journal articles, to learn scientific writing as they write reports, and to speak publicly as they prepare and give oral presentations. Many students in these classes reported them as being pivotal moments in their undergraduate career. One student’s microscopy course feedback stated, ‘This is the class that made me apply to graduate school’.
The Next Steps for the Bratu Lab
Dr Bratu and her students have many projects planned that will build upon recent findings. Now that they have successfully visualised realtime nucleic acid and protein transport within the oocyte, they hope to study this movement within other cell types – cell types more difficult to work with in a laboratory setting, such as neurons. As mRNA regulation is likely a key player in the development of neurodegenerative diseases, results of these studies could have vast implications for diseases like Alzheimer’s, Parkinson’s and Huntington’s.
They will be expanding on their results on oskar and its association with Bruno and Cup by determining whether these translational repression mechanisms are coupled to the repression mechanisms of microRNA via recruitment into the P-bodies. By knocking down the expression of each translation repression factor in combination of other protein factors in a systematic fashion, Dr Bratu and colleagues will elucidate the translation control process of oskar mRNA and the many proteins it involves during oogenesis.
Building upon the discoveries of Drongo’s co-localisation with Clathrin light chain and F-actin at the oocyte cortex, the team aims to dig even deeper, determining if Drongo participates in the endocytosis of VAMP7 (Vesicle Associated Membrane Protein 7) in fruit fly egg chambers or if it acts as a GAP effector for Arf6, a protein involved in biological membrane trafficking, using in vitro assays.
By deciphering these intricate biological processes, and gaining further insight into how mRNA-protein complexes influence gene expression and protein translation, Dr Bratu and others in her field pave the way for colleagues to unravel the complexities of developmental or neurologic disorders. This work may lead to the development novel therapeutics for deadly viruses like HIV or Influenza A.
Meet The Researcher

Dr Diana P. Bratu
Department of Biological Sciences
Hunter College, CUNY
New York
USA
Dr Diana P. Bratu is an Associate Professor in the Department of Biological Sciences and the Scientific Director of the Bio-imaging Facility at Hunter College, CUNY, US. She began her career at New York University, where she obtained her B.A. in Chemistry and Mathematics, and pursued a M.S. and Ph.D. in Molecular and Cell Biology. She then completed two postdoctoral fellowships, one in Molecular Genetics at the Public Health Research Institute, NJ and another in Cell Dynamics at the University of Massachusetts Medical School, MA. She became involved in cellular imaging during her doctoral training and has since been an avid contributor to the field of biophotonics.
CONTACT
E: bratu@genectr.hunter.cuny.edu
T: (+1) 212 772 5235
W: http://bratu.bioweb.hunter.cuny.edu/
W: http://www.bratulab.com
FUNDING
NSF
NIH
PSC-CUNY
REFERENCES
IE Catrina, LV Bayer, G Yanez, JM McLaughlin, K Malaczek, E Bagaeva, SAE Marras and DP Bratu, The temporally controlled expression of Drongo, the fruit fly homolog of AGFG1, is achieved in female germline cells via P-bodies and its localization requires functional Rab11, RNA Biology, 2016, RNA Biology, 2016, 11, 1117–1132.
LV Bayer, M Batish, SK Formel and DP Bratu, Single molecule RNA in situ hybridization (smFISH) and immunofluorescence (IF) in the Drosophila egg chamber, Drosophila Oogenesis: Methods and Protocols, Methods in Molecular Biology, Springer Protocols, Humana Press, 2015, 1328, 125–36.
IE Catrina, SAE Marras and DP Bratu, Tiny molecular beacons: LNA/2’Omethyl RNA chimeric probes for imaging dynamic mRNA processes in living cells, ACS Chemical Biology, 2012, 7, 1586–1595.
MM Mhlanga, DP Bratu, A Genovesio, A Rybarska, N Chenouard, U Nehrbass and JC Olivo-Marin, In vivo colocalisation of oskar mRNA and trans-acting proteins revealed by quantitative imaging of the Drosophila oocyte, PLoS One, 2009, 4, e6241.
DP Bratu, BJ Cha, MM Mhlanga, FR Kramer and S Tyagi, Visualizing the distribution and transport of mRNAs in living cells, Proceedings of the National Academy of Sciences USA, 2003, 100, 13308–13.



