Dr Kenneth J. Pienta – Are Poly-aneuploid Cancer Cells the Keystone Cure for Cancer?
‘Our audacious idea is to cure cancer.’ Dr Kenneth Pienta at the Johns Hopkins School of Medicine, USA, speaks with genuine passion about his ground-breaking research. With his team, he has recently discovered that in every type of cancer, a special type of rare cancer cell – a polyaneuploid cancer cell (PACC) – exists and hides within the greater cancer cell population. The team hypothesises that ‘PACCs are a master mediator of therapy tolerance’ and thus, the critical treatment target. Now, in a call to arms, Dr Pienta is asking researchers and scientists across diverse disciplines to unite in developing a cure for cancer.
The Challenge of Cancer
Cancer is a global health concern. There are over 100 types of cancer, which taken together, kill more than 10 million people across the world each year. Although localised cancers can be treated successfully through excision or localised radiotherapy, metastatic cancers spread throughout the body and are incurable, eventually leading to death. Once cancer cells have metastasised, they grow aggressively and are resistant to virtually all treatments. Despite enormous funds and dedicated efforts put into cancer research over the last half-century, half of all people diagnosed with cancer still die from their disease.
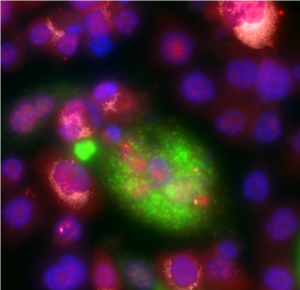
Polyaneuploid Cancer Cell. Credit Laboratory of Kenneth Pienta
Traditional thinking in cancer therapy accepts that cancer is incurable once it spreads. There are billions of cells in a tumour and it only takes one cell to randomly mutate into a form with increased tolerance to survive treatment and then to clone itself. Cancer therapies have focussed on multi-drug chemotherapies to tackle the progression of new cancers as they occur, or to develop drugs that target resistance-associated mutations. However, cancer cells have proven resistant to all high-tech medical innovations to date. Researchers have not understood exactly what cells or mechanisms that cause this remarkable resilience amongst cancer cells.
Dr Kenneth Pienta at the Johns Hopkins School of Medicine, USA, is leading an amazing and bold line of discovery in cancer research. Critically, his team now believes that polyaneuploid cancer cells (PACCs) are the master mediators of this resilience and provide an adaptive way for tumours to survive almost any type of stress.
Cancer as an Ecosystem
Dr Pienta’s work is a fantastic example of thinking outside the box and began with the consideration that cancer research might learn from the study of ecological systems. His team considered the concept of keystone species in known ecosystems. Here, specific species, such as the elephants in the Serengeti or the wolves of Yellowstone Park, are known to be critical to that ecosystem such that if they are removed, the ecosystem will either dramatically change or collapse. The researchers had come to the belief that the connectivity and interdependence of animal ecosystems could be applied to cancer cell populations, encouraging an entirely different observational approach to how these cells interact and respond holistically within the whole body of which they are part. The PACCs are now being explored as keystone species of the cancer cell population.
The Tale of the Grasshopper and the Locust..

Two key questions across all types of cancer research are, why do some cancer cells become immune to treatment and why do they metastasise? Returning to thinking about cancer cells as an ecosystem paradigm, discussions with cancer biologist Dr Sarah Amend and ecologist Dr Joel Brown turned to the example of short-horned grasshoppers. When food is abundant, grasshoppers are typically green and healthy solitary specimens, but in times of nutritional stress, the next generation becomes yellow locusts, growing wings and changing their behaviour to become more gregarious, swarm-forming animals that travel to seek new food sources. Once resources are again abundant, the next generation once again transmogrifies back to the grasshopper morph.
Applying this to a cancer cell ecosystem, in times of induced stress (i.e., therapy), normally stable epithelial cancer cells are known to transition into mobile cells and relocate. To observe this transition, Dr Pienta began a unique project with Professor Robert Austin at Princeton University, USA. In an incredible innovation, Professor Austin was able to create a cancer ecosystem on a (micro)chip. Described as an ‘engineered cancer micro-environment,’ the mini-ecosystem could be visually tracked by a camera and computer to watch the changes in each cancer cell as it occurred in real-time.
Prostate cancer cells were seeded into the micro-environment and treated with docetaxel, an anti-prostate cancer drug at a lethal dose expected to kill 99.9% of the cancer cells. Dr Pienta describes how after 14 days, the cancer cells were largely eliminated, but then, unexpectedly, some grew back. Dr Pienta noted, ‘for the first time – in live time – I watched the birth of drug resistance, it was sobering and totally scary!’ After successfully killing nearly all the cancer cells, the cancer cells returned against all expectations, but now they were resistant to the drug therapy.
Under higher magnification, they observed that large, highly mobile cells began to emerge as the cells died off, surviving despite the seemingly lethal dose, reminiscent of the birth of locusts. The cells were giant and polyploid (containing extra sets of chromosomes). Somehow, these abnormal cells were able to asymmetrically divide, birthing a bloom of new small clone cells that were drug resistant. The team termed the cells poly-aneuploid cancer cells (PACCs); aneuploidy meaning having an abnormal number of chromosomes.Initially, Dr Pienta and his team were sceptical, ‘We sat on this [finding], we just didn’t believe it – if it is this fundamentally important, how did we [the scientific community] miss this, all these years?’ In fact, PACC cells had been reported in the literature for over 100 years (often referred to as polyploid or polymorphous giant cancer cells, multinucleated cancer cells, or blastocyst cancer cells) and had been observed after therapeutic intervention. However, it had long been assumed that these giant polyploid cells where simply senescent dying cells that could not divide. As a result, PACCs were largely ignored and even screened out of automated cellular recognition programmes looking for metastatic cancer cells. Interestingly, across diverse research fields, PACCs were noticed yet nobody had really put together that PACCs could be the central, common actuator of tumour formation, metastasis, and therapeutic resistance across all cancer types – the hypothesis that Dr Pienta and his team are compelled to take forward.
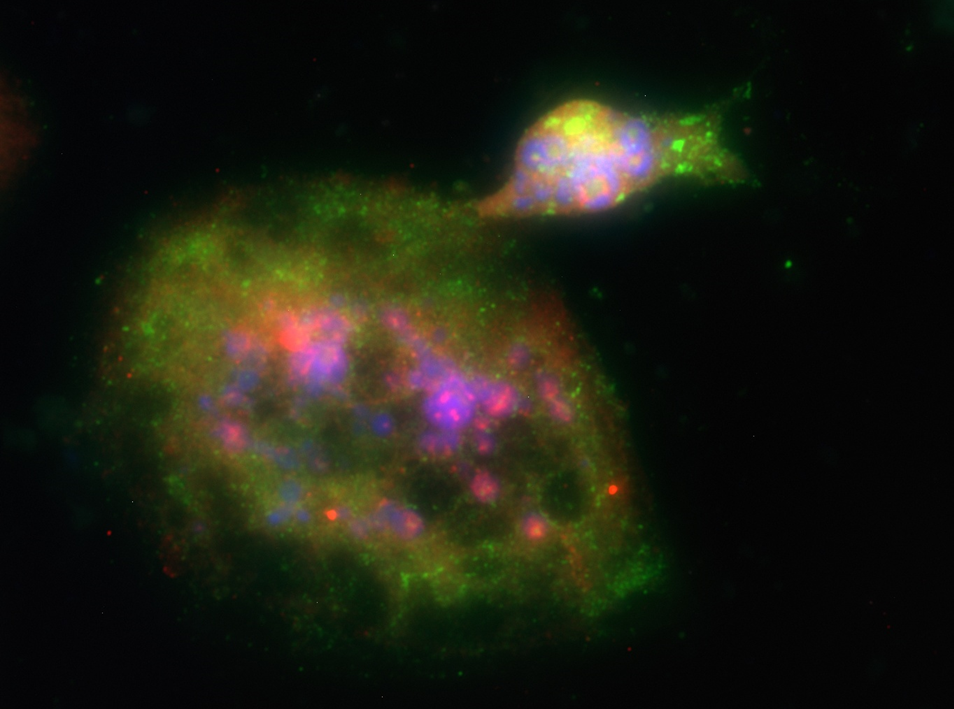
Polyaneuploid Cancer Cell Budding. Credit Laboratory of Kenneth Pienta.
PACCs: Hiding in Plain Sight
Returning to the cancer ecosystem, Dr Pienta believes that PACCs form a keystone species and that they must be targeted to truly eliminate all cancer. With his team, Dr Pienta has since discovered that in every type of cancer, PACCs represent a small percentage of the cancer cell population. When therapeutic stress of any kind is introduced to the ecosystem, the majority of cancer cells die, resulting in a decreased tumour size, but the surviving cells form PACCs, which then repopulate the tumour with resistant cancer cells. The team hypothesise that ‘PACCs are a master mediator of therapy tolerance,’ with the ability to give rise to a new generation of super-resistant cancer cells. PACCs are thus a ‘species’ adapting for survival and not simply a mutated cancer cell. Like the locust, they are formed in response to stress rather than simply existing in a heterogeneous cell population – and critically – may be the true source of cancer resistance and lethality.To further understand these intriguing discoveries, Dr Pienta continues to work with a cross-disciplinary group of investigators. In preliminary data from ongoing research, it seems that PACC cells are formed in two known ways. These are either by failed cytokinesis, in which the cells try to divide, double their DNA content and nuclei but fail to divide, or the fusion of cells which merge together. It seems likely that these processes begin in a primary tumour when it is stressed by low oxygen or low nutrients, as a way to protect itself. Working with geneticist Dr Laura Buttitta at the University of Michigan, the PACC cells were found to enter a ‘hibernating’ state (potentially for years) when exposed to chemotherapy. This is a critical survival mechanism, as cancer therapy requires damaged cancer cells to divide for it to work and to kill the cells. Therefore, the giant PACCs wait until the stress is gone before dividing again.
Dr Pienta has also worked with Dr Max Wicha, a cancer biologist and breast cancer oncologist at the University of Michigan, a founder of solid tumour cancer stem cell theory. This theory suggests that in a cancer cell population, a few cells act as stem cells that reproduce themselves to maintain the cancer and metastasise themselves to disperse around the body. Sounding much like the behaviour of PACCs, Dr Wicha and Dr Pienta were able to demonstrate that PACCs have stem cell-like properties. Dr Wicha noted that while he had commonly observed these cells in cancer, he had not recognised their importance. It has been a typical response from many cancer researchers that have talked to Dr Pienta. In other words, the cells were blatantly hiding in plain sight.
Beginning the Search for the Cure
The next stage for Dr Pienta and colleagues is now to put this new knowledge base together to target the keystone PACCs for destruction, as they believe that by removing the intermediate keystone species, the cancer ecosystem will collapse. Multiple research strands are being planned to provide a deeper understanding of PACCs and to identify ways to eliminate them. One strand is to target how the aneuploid cells with too much DNA reproduce to produce the resistant clones. Such genetically messed-up and damaged cells would normally trigger the body’s natural defence systems to initiate cell death. PACCs have figured out a way around this problem. Early research demonstrates that the PACCs switch on a protein normally only used in the embryonic stage of development to assist the cell to perform mitosis, the normal chromosome division to create two cells. The researchers are searching for ways to target this process in the PACCs.
In a second approach, learning again from ecology, Dr Pienta and his team have noted that hibernating animals survive the winter using fat stores, and strikingly, PACCs are also filled with fats. Thus, the researchers propose that targeting fat storage and metabolism may starve the PACC cells in the hibernation stage and thus prevent the cloning of new cells.
The team recognises that targeting the PACCs is a difficult task since they represent such a tiny fraction of the overall tumour cell burden. Furthermore, they acknowledge that they will likely need to combine multiple strategies that include eliminating a large number of the general cancer cells with a strategy to kill the few in number, but critically important PACCs. Again, drawing from ecology, it may take a pesticide (chemotherapy) to kill the many worker bees (cancer cells) with a specific trap to capture and kill the queen bee (PACC) before it can repopulate the hive (new tumour). Dr Pienta’s call to arms will bring together the expertise of investigators drawn from several different institutions and fields. Excitingly, the targeting of keystone PACC cells may ultimately end all metastatic cancers, and in doing so, solve one of medicine’s greatest challenges.
Reference
https://doi.org/10.33548/SCIENTIA518
Meet the researcher

Dr Kenneth J. Pienta, MD
The Johns Hopkins Hospital
Baltimore, Maryland
USA
Dr Kenneth J. Pienta completed his undergraduate degree in Human Biology in 1983 at the John Hopkins University in Baltimore, USA, followed by his medical doctorate in 1986. Dr Pienta completed further training in internal medicine and oncology before taking faculty positions at Wayne State University from 1991–1993 and the University of Michigan from 1994–2013. In 2013, he returned to the John Hopkins University to take the position of the Donald S. Coffey Professor of Urology and Director of the Brady Urological Institute Research Laboratories. He is also a Professor of Oncology, Professor of Pharmacology and Molecular Sciences, and Professor of Chemical and Biomolecular Engineering. Commensurate with these prestigious appointments, Dr Pienta is recognised as helping pioneer the application of ecological principles to cancer and has international expertise in the development of novel therapeutic programmes for prostate cancer, often accomplished with collaboration with multi-disciplinary teams of scientists and clinicians. He has published extensively, holds a number of consulting positions, and is the well-deserved recipient of numerous honours and awards.
CONTACT
E: kpienta1@jhmi.edu
W: www.kennethjpienta.com
Twitter: @pienta_brady
KEY COLLABORATORS
Sarah Amend, Johns Hopkins School of Medicine
Joel Brown, Moffitt Cancer Center
Bob Axelrod, University of Michigan
Bob Austin, Princeton University
Emma Hammarlund, Lund University
Laura Buttitta, University of Michigan
Max Wicha, University of Michigan
FUNDING
National Institutes of Health
Prostate Cancer Foundation
Multiple additional donors
FURTHER READING
KJ Pienta, EU Hammarlund, R Axelrod, et al, Poly-aneuploid cancer cells promote evolvability, generating lethal cancer, Evolutionary Applications, 2020, 00, 1–9, DOI: 10.1111/eva.12929.
KJ Pienta, EU Hammarlund, R Axelrod R, et al, Convergent evolution, evolving evolvability, and the origins of lethal cancer, Molecular Cancer Research, 2020, DOI: 10.1158/1541-7786.MCR-19-1158.
SR Amend, G Torga, KC Lin, et al, Polyploid giant cancer cells: Unrecognized actuators of tumorigenesis, metastasis, and resistance, Prostate, 2019, 79(13), 1489–1497.
SR Amend, S Roy, JS Brown, KJ Pienta, Ecological paradigms to understand the dynamics of metastasis, Cancer Letters, 2016, 380, 237–-42.
SR Amend, KJ Pienta, Ecology meets cancer biology: the cancer swamp promotes the lethal cancer phenotype, Oncotarget, 2015, 6, 9669–78.
KJ Pienta, N McGregor, R Axelrod, DE Axelrod, Ecological therapy for cancer: defining tumors using an ecosystem paradigm suggests new opportunities for novel cancer treatments, Translational Oncology, 2008, 1(4), 158–64.
Creative Commons Licence
(CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
More articles you may like
Professor Tian Yu Cao | Twistor Theory: A New Framework for Quantum Gravity
At Boston University, Professor Tian Yu Cao is rethinking the foundations of modern physics. His work builds on twistor theory which demonstrates that spacetime is secondarily derived from twistor constructions, but goes further to highlight the most important implication of the Penrose transform in that the primary physical agents can only be mathematically described by elements of cohomology with the defining feature having roots in spin. This view of primary agents combines with Cao’s other major claim that quantum behaviour itself may arise from the physical property of spin leads to a new consistent framework of quantum gravity in which long-standing puzzles in black holes (evaporations) and cosmology (transitions between cycles of cosmos) can be adequately addressed, with the crucial help from the on-going development of operator product expansion formular defined on twistor space.
Professor Mikhail V. Medvedev | Plasma Waves in Extreme Magnetic Fields: Exploring the Quantum Regime
In environments where magnetic fields exceed even the limits of classical physics, such as magnetars and next-generation laser experiments, plasma behaviour is fundamentally altered by quantum effects. Professor Mikhail V. Medvedev and colleagues have developed a framework to understand how these extreme conditions reshape plasma waves, revealing that while familiar wave structures persist, their properties are significantly modified. These insights provide a foundation for interpreting astrophysical observations and advancing high-energy plasma experiments.
Alex Spezowka | Responsible Research Writing in the Age of AI: From Detection to Transparency
Artificial intelligence is often discussed as a future challenge for research, yet it is already shaping how many papers are written. Drawing on emerging evidence and recent analysis, work led by Alex Spezowka highlights a key shift in thinking: rather than trying to detect AI use, the focus is moving towards how it can be used responsibly. This has important implications for how research is produced, evaluated, and trusted.
Laura Daniela Muñoz Duarte – Jo Philips | Rethinking Hydrogen Use in Acetogenic Bacteria: Kinetics, Thresholds, and Biotechnological Potential
Acetogenic bacteria can convert carbon dioxide and hydrogen into useful products, making them promising tools for sustainable biotechnology. Research led by Laura Muñoz and Jo Philips reveals that hydrogen consumption follows a simpler kinetic pattern than previously assumed, while also uncovering major differences between species in how efficiently they operate at low hydrogen levels. Together, these findings provide a clearer framework for selecting and optimising acetogens for industrial applications.




