Professor Arthur Devries – Cold As Ice: Antifreeze Proteins In Polar Fishes
Polar environments: extreme temperatures and unique adaptations
Organisms in the Polar Regions are highly adapted for survival right at the lower end of the thermal range of our planet. Survival in such conditions requires a variety of specialised biological attributes, in both the land and marine creatures that inhabit and thrive in these areas. Endothermic animals, which can generate their own body heat through metabolic processes, tend to develop layers of insulatory tissue to maximise the efficiency of their metabolism. Examples include the blubber layers of whales and Polar Bears. However, ectothermic animals, such as fishes, which rely on the external environment to regulate their body temperature, face a significant challenge to generate enough energy for growth and for routine activity. Some fishes can avoid freezing by living in deeper waters in a supercooled state, thereby avoiding contact with ice crystals that could enter their body and grow, causing them to freeze. However, for those that live in shallower waters and in particular those that exploit ice structures as specialised habitats that offer shelter from predators, exposure to and ingestion of ice crystals is inevitable. Therefore, for such fishes, some biochemical cunning is required to prevent their bodies from freezing in the frigid polar oceans.
Polar oceans are cold all year-round, but unlike the air over polar landmasses that can fall to incredibly low temperatures, they rarely fall below their freezing point of –1.9°C, as a consequence the high heat capacity of water and the insulating ice cover. Nevertheless, polar water temperatures are sufficiently low to freeze poorly adapted organisms. Despite the challenges to survival and success posed by the polar marine environment, a plethora of species thrive and reproduce here. The area most studied by Professor DeVries is called the Ross Sea, which is located in Antarctica. This area harbours a wide variety of species including approximately 100 species of fish. This includes the aptly named Antarctic Toothfish, which can grow up to a fearsome two metres long, demonstrating that cold water is no obstacle to the growth of ectotherms, provided they are suitably adapted. Other animals that call this area home include penguins, seals and whales. However, ecologically speaking, one of the most important families of animals in this area is the notothenioids. The notothenioids represent the dominant family of Antarctic fish. They have evolved to fill a huge variety of niches and are found at a range of depths within this aquatic environment, despite lacking swim bladders, which enable fish to control their buoyancy. They provide an essential source of food for a significant number of other species and are fundamental to the biodiversity of this area.

‘How the antifreeze glycoproteins, as well as other antifreeze proteins, evolved into functional antifreeze molecules is of much interest to evolutionary biologists’
A significant key to the success and versatility of the notothenioids is their ability to resist becoming frozen at very low temperatures. This is in large part due to their production of antifreeze proteins, which inhibit the growth of ice crystals in their bodily fluids. Professor DeVries tells Scientia of the importance of this adaptation for biodiversity in the region: ‘If it were not for the capability of polar fishes to avoid freezing – as a result of the presence of blood antifreeze proteins – the polar ecosystems would be quite different. Many polar birds, seals and whales prey upon the polar fishes and they make up a large portion of their diet. If the fishes were absent then there would only be invertebrates as a source of prey and the diversity of the bird and seal fauna would be much reduced.’
Professor DeVries has devoted his career to studying how these proteins function to permit survival at extremely low temperatures. In this article, we take a look at his fascinating research career and some of his key results and motivations. His work has involved challenging research in remote Polar Regions to catch fish samples and determine how their evolutionary adaptations in antifreeze proteins make them uniquely suited for life in the freezer.

Professor DeVries’ discovery of antifreeze proteins in the late 1960s
Professor DeVries discovered antifreeze proteins while working as a young researcher in the 1960s. He was based on the shore of McMurdo Sound on Ross Island at McMurdo station, a major Antarctic research base run by the United States Antarctic program. Here, he tells Scientia how he came to be involved in this type of research and what he enjoyed about it: ‘I heard about a research position in Antarctica with a professor from Stanford University and applied and spent 13 months at McMurdo Station, Antarctica in 1961 performing an investigation of the respiratory metabolism of the endemic notothenioid fishes caught in the Sound. I enjoyed the combination of rigorous field work (catching the fish) and laboratory work in this remote location.’ He went on to explain how he made his fortuitous discovery of antifreeze proteins: ‘During these experiments I noticed that a deep water notothenioid fish would freeze to death if any ice was present in our refrigerated salt water while those caught in the shallow water survived in the presence of ice. I decided to investigate why there was a difference in these species living in water of the same temperature (-1.9°C) for my PhD thesis research at Stanford. I investigated what compounds were responsible for their capability to avoid freezing in this environment while fishes in temperate waters would freeze to death at -0.8°C. My study culminated in the discovery of the antifreeze glycoproteins, the compounds responsible for their extreme freeze avoidance.’
In his early characterisation of this extreme freeze avoidance, Professor DeVries showed that the freezing point of the serum of such fish was -2°C, which was well below the freezing point of the serum of fish that inhabit warmer waters. Additional salt content in the serum of the cold-water fish could only explain a portion of this difference and the balance was largely because of the presence of antifreeze glycoproteins.
This discovery of antifreeze proteins has opened up a completely new field of research into biological antifreezes, with labs all over the world now investigating these proteins, their occurrence in nature and use in biotechnological processes.
Current knowledge and applications of antifreeze proteins
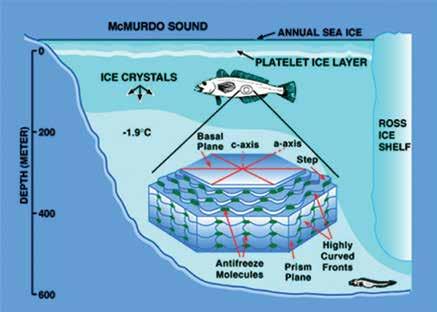
In the years following the initial discovery of antifreeze proteins, significant research has been devoted to identifying new types of these proteins and characterising their mechanism of action, which is not yet completely understood. However, in general terms, the current accepted mechanism of action of antifreeze proteins involves essentially irreversible binding (through adsorption) to the sites of growth of the crystal faces of ice crystals. This results in the antifreeze proteins dividing the ice surface into many small domains whose growth fronts become highly curved. Such fronts would have a higher surface free energy than a straight growth front. For growth to occur, energy must be removed from the system, which is accomplished by lowering the temperature. This is the same thing as lowering the freezing point. Probing the ice/water interface indicates that this is a viable hypothesis. Antifreeze proteins are an order of magnitude more effective (on a molal basis), when compared with automobile antifreezes such as ethylene glycol, which operate via the mass action effect, and hence aren’t as effective in preventing ice formation. Antifreeze proteins have been of immense interest to scientists in the years since their discovery. Professor DeVries tells Scientia about their impact in the fields of biochemistry, evolutionary biology and physical chemistry: ‘In the field of biochemistry it is of great interest how proteins interact with water. The fact that the antifreeze proteins can recognize the solid phase (ice) in the presence of an excess of water molecules is amazing. How the antifreeze proteins recognize the ice and preferentially bind to it is of interest to physical chemists and biologists alike. The details of exactly how the bound antifreeze proteins prevent the growth of ice remains to be elucidated,’ he explains. ‘In the area of evolution, the antifreeze proteins are an excellent example of convergent evolution as the antifreeze glycoproteins in the Antarctic notothenioid fishes are identical to those in the unrelated Arctic cods but they have different evolutionary origins. How the antifreeze glycoproteins, as well as other antifreeze proteins, evolved into functional antifreeze molecules is of much interest to evolutionary biologists.’
Biological antifreezes also have a wide range of potential applications in biotechnological industries. The ability to prevent freezing using biologically compatible agents, has inherent value, and the production of highly potent synthetic antifreezes in the lab, is the goal of modern research. Professor DeVries mentions some of the potential future uses: ‘There is interest in creating synthetic compounds which mimic the antifreeze compounds in fishes but have greater antifreeze activity with the end point being able to prevent freezing of cells and tissues well below their natural freezing points. Such compounds might be useful for storing organs for transplantation as well as preventing deterioration of certain foods during cold storage.’
The latest development in this research has been the somewhat surprising discovery that antifreeze proteins also prevent ice crystals from melting. This more recent research was carried out in collaboration with Dr Paul Cziko of the University of Oregon and Professor Chi-Hing Cheng of the University of Illinois. The team found that ice that had been stabilised through binding with antifreeze proteins resisted melting and would not melt until exposed to much higher temperatures than expected. This was true both in the laboratory and also in living fish specimens. It is thought that the ice crystals present in polar fishes arise from their external environment and are introduced into their bodies through processes such as feeding or water entering their gills. It had been previously observed that the bodies of polar fishes contained ice crystals in locations such as their blood and spleen. It was unclear if the fish somehow eradicated these crystals or if they melted during summer warming periods. The team monitored water temperatures in Antarctica for about ten years and concluded that the water temperatures never got high enough to cause the ice crystals inside the fishes to melt, especially given that they were stabilised with antifreeze proteins. Some Arctic fishes, in contrast, encounter seasonal warming, and so do not require antifreeze proteins year-round. Fascinatingly, these fishes degrade their antifreeze proteins in the summer to conserve energy, to build up their reserves again in the autumn in preparation for the cold winter.
However, for fishes in the colder Antarctic climate, with a year-round complement of antifreeze proteins it is currently unclear if they are able to eradicate the protein-stabilised ice crystals from their bodies. If not, and they continue to accumulate for the entire lifetime of the fish, it is unknown if they result in adverse effects, such as blockage of blood vessels or production of inflammatory reactions. If so, then the unique adaptation that enabled such fishes to thrive in this hostile environment, antifreeze proteins, could also come with a down side, as they appear to contribute to accumulation of the crystals. This question of ice crystal accumulation and eradication is the current focus of Professor DeVries’ research. He believes that there must be a method that the fishes use to eradicate the crystals: ‘The fishes must have a biological mechanism for ridding themselves of these ice crystals because if they continue to accumulate them, one would assume that the ice burden would become so great that it would overwhelm their physiological systems. Thus, how do the fishes avoid accumulating detrimental levels of ice’. How indeed?
Meet the researcher

Department of Animal Biology
University of Illinois,
USA
Professor Arthur DeVries is an Emeritus Professor in Animal Biology and Molecular and Integrative Physiology at the University of Illinois. Professor DeVries has devoted his career to the discovery and characterisation of antifreeze proteins present in the blood and tissues of polar fish. He was the first person to discover these proteins, and their discovery opened up a completely new field of research. Professor DeVries was elected a Fellow of the American Association for the Advancement of Science in 1984, for his distinguished research on biological antifreeze compounds of coldwater fishes. He has been presented with many awards, including the Premio Internazionale ‘Felice Ippolito’ (international prize for biological excellence) by the Italian the Accademia Nazionale dei Lincei and National Antarctic Programme (2005), the Life Time Achievement Award for discovery and research on antifreeze proteins in polar fishes at the 1st International Ice-Binding Protein Conference. Finally, he also caught a previously unknown Antarctic fish, which was described by the famed Russian Icthyologist, Dr A. P. Andriashev and named ‘Paralaparis devriesi’
CONTACT
E: adevries@life.illinois.edu
T: (+1) 217 333 4245
W: https://mcb.illinois.edu/faculty/profile/adevries/
KEY COLLABORATORS
Dr Konrad Meister, AMOLF, The Netherlands
Professor Clive Evans, Auckland University Auckland, New Zealand
Professor C-H Christina Cheng, Animal Biology, University of Illinois, Urbana, IL.
Dr P.A. Cziko, University of Illinois, Urbana, Illinois
Dr Charles Knight, NCAR, Boulder, Colorado
FUNDING
Division of Polar Programs, Geosciences, The National Science Foundation
REFERENCES
JA Raymond and AL DeVries, Adsorption inhibition as a mechanism of freezing resistance in polar Fishes, Proc. Nati. Acad. Sci. USA, 1977, 74, 2589–2593.
PA Cziko, AL DeVries, CW Evans and C-H Christina Cheng, Antifreeze protein-induced superheating of ice inside Antarctic notothenioid fishes inhibits melting during summer warming, PNAS, 2014, 111, 14583–14588.

