Professor Costantino Vetriani | Professor Frank T. Robb – Uncovering the Relationship Between Aquatic Thermophiles and Human Bacterial Pathogens
Thermophiles thriving on hydrothermal vents are opening new avenues towards understanding the chemical mechanisms of human diseases. Here we discuss the work of Professors Costantino Vetriani and Frank Robb, who isolate these extreme microorganisms and study their metabolism, towards the development of novel biotechnological and therapeutic applications.
Hydrothermal Vents: Teeming with Life
Hydrothermal vents arise as a result of volcanic activity on the seafloor. In volcanically active areas, water enters cracks in the Earth’s crust, dissolves minerals, and becomes super-heated by the magma below. This hot water can then erupt as a geyser from a hydrothermal vent, where water temperatures can change from near freezing to 400°C over short distances. The dissolved minerals crash out of the water upon contact with the cold seafloor, gradually leading to the formation of a chimney around the vent.
In hydrothermal vent communities, the first step in the food chain are chemosynthetic bacteria – bacteria that extract energy from reactive inorganic compounds such as hydrogen sulphide. These bacteria can exist as either free-living, or in symbiotic relationships with creatures such as vesicomyd clams, vestimentiferan tubeworms, and bathymodiolid mussels. Shrimps that host sulphur-dependent bacteria can also be found near hydrothermal vents.
The microbial communities in these active, hot, venting chimneys have been increasingly studied since the late 1970s. They have attracted many groups of scientists from around the world as research opportunities grow and potential commercial applications are realised. Here, we discuss the work of two scientists – Professor Costantino Vetriani at Rutgers University and Professor Frank Robb at the University of Maryland – and their contributions to expanding our understanding of the biochemistry of these important marine environments.
This pair of scientists first started working together back in 1995, when Professor Vetriani joined Professor Robb’s research group as a postdoc, and their collaboration continues to this day.

Active deep-sea hydrothermal vents releasing hot fluids at the bottom of the ocean
The Importance of Thermophiles
Hydrothermal systems support an enormous diversity of microbial species that is, in many places, comparable to that of a rainforest. One of the most abundant groups of bacteria are known as Epsilonproteobacteria. This group of bacteria include the families Campylobacteraceae and Helicobacteraceae, represented by the pathogenic Campylobacter that can cause gastrointestinal infection in humans and Helicobacter pylori, a well-known cause of ulcers in the stomach and duodenum in humans. Disentangling the evolutionary relationships between deep-sea vent Epsilonproteobacteria and human pathogens can therefore provide insight into the origins of virulence in pathogens.
Professor Costantino Vetriani is a microbiologist immersed in the ecology and evolution of deep-sea bacteria and the adaptations of these bacteria to extreme environmental conditions. In 2003, Professor Vetriani and colleagues conducted experiments to study the structure and composition of microbial communities in mixtures of cold seawater and fluids heated by magma at hydrothermal vents on the East Pacific Rise – a mid-ocean ridge that runs from the Gulf of California to South of Easter Island in the Pacific Ocean. The aim of their research was to describe the different stages of colonisation of vents by bacterial populations and to uncover any associations between the microbial colonisers and the chemical regimes in the vents. The results were quite interesting – in vents with fluids flowing down the chimneys, the microbial communities were dominated by Epsilonproteobacteria, while in parts of the seafloor with no flow, the microbial communities were dominated by Gammaproteobacteria related to the family Thiotrichales. Fundamental studies like these have at least two direct applications – they help to improve mathematical models describing the transfer of energy and carbon on the seafloor and to explain the role of microbial communities in transforming the carbon into biomass, which becomes available for higher life-forms. In fact, several studies have found that Epsilonproteobacteria not only dominate communities on the surface of vent chimneys, but they are also common on other animal surfaces. Because of their high biomass, fast growth rates and adaptable metabolism, these bacteria are possibly the first to colonise the dynamic diffuse flow vent environments.

Frank Robb and postdoc Lydia Kreuter operate a specialised high temperature 200L fermenter. CREDIT: Lisa Tossey, University of Delaware
Since then, many other studies have shown that these bacteria diverged in the tree of life, as antecedents of their human pathogenic relatives and developed novel systems for respiration, sensing and environmental responses to changes in habitat conditions. Professor Vetriani is also hoping to increase knowledge of bacterial communities in shallow-water (<200 m depth) hydrothermal vents. For many years, these ecosystems were much less studied than deep-sea vents. An important characteristic of the shallow-water vents is that they are largely influenced by photosynthesis. This project took Professor Vetriani and colleagues to Paleochori Bay, on the coast of the Island of Milos, in Greece. Here, the venting area is surrounded by patches of the seagrass Posidonia oceanica, where temperatures range from 25 to 119°C. Epsilonproteobacteria were again remarkably abundant, representing 60% of the total microbial diversity. Molecular studies revealed the presence of species that had only been previously found at deep-sea hydrothermal vents, such as the chemolithoautotrophic bacterium Sulfurovum spp.
In addition to thriving at high temperatures, certain Epsilonproteobacteria from deep-sea vents grow in the absence of oxygen. By culturing them in the laboratory, scientists have found that these bacteria use a diverse spectrum of electron donors and acceptors such as hydrogen, sulphur compounds, nitrate and oxygen for cellular respiration and even share the ability to use the same electron donors and acceptors. For example, Cetia pacifica, a new thermophilic bacterium isolated from samples collected from a black smoker chimney on the East Pacific Rise by Professor Vetriani’s research team, grew when nitrate or sulphur were available in the growth media. Because of these versatile metabolic characteristics, it is believed that Epsilonproteobacteria play a significant role in the ecological and biogeochemical processes of diverse environments on our planet, from marine geothermal habitats to the gastrointestinal tract of mammals.

Costantino Vetriani collecting rock colonising microbial biofilms from the gas vents of Tor Caldara, Italy
Linking Chemosynthetic Bacteria to Human Pathogens
Quorum sensing (QS) is a communication mechanism between cells that depends on their density and the production of signalling molecules. QS is used by bacteria to control gene expression, including virulence genes and functions associated with biofilm formation and host colonisation. One QS system that appears to be widespread across the bacterial domain and has been proposed to function as the universal language for communication between species, is based on a furanone derivative known as autoinducer-2 (AI-2) which is synthesised by the LuxS enzyme.
In 2007–2008, Professor Vetriani and colleagues took part in a scientific mission to the East Pacific Rise aboard R/V Atlantis. Using the Deep-Submergence Vehicle Alvin, the researchers used experimental microbial colonisation devices made of stainless steel mesh to sample microbial biofilms. The samples were used in genomic and phylogenetic studies to understand the ancestry of the LuxS lineage and LuxS gene flow in Epsilonproteobacteria. The results indicated that LuxS is conserved in all Epsilonproteobacteria and that the mesophilic strains, including pathogens, shared a common LuxS ancestor nested within the thermophilic lineage, suggesting that the epsilonproteobacterial LuxS lineage originated in geothermal environments. Further, these analyses indicated two episodes of horizontal gene transfer: Helicobacter pylori acquired the gene from Enterococcus faecium, a Gram-positive bacterium that also inhabits the human gastrointestinal tract. Moreover, marine Gammaproteobacteria of the genera Photobacterium and Vibrio, along with Escherichia coli and other human associated bacteria, share a common LuxS ancestor with Arcobacter nitrofigilis and Arcobacter butzleri, a marine organism and a human pathogen, respectively. Together, these results indicated that the LuxS gene represents an evolutionary link between thermophiles and human pathogens.
Chaperones
If early life on earth existed near hydrothermal vents, then heat shock would have been prevalent and life-threatening, as vents frequently ejected hot fluids. In environments with very sharp temperature gradients between hot emissions and ambient water, organisms that could survive the dangerous thermal and chemical conditions close to a vent would be at a huge advantage. There is an important family of proteins produced by cells in response to these stressful conditions – called heat shock proteins (HSPs). Many of them are chaperones, proteins that nurse damaged client proteins back to activity.
These proteins are turned on in response to heat shock to protect the other cellular proteins against denaturation. They work by binding to other proteins during heat shock to prevent them from becoming irreparably damaged by unfolding, and then they help to restore and refold these proteins afterwards.
HSPs are found in all three domains of life – Archaea, Bacteria and Eukarya, and are involved in many vital biochemical processes such as photosynthesis, mitochondrion function, DNA replication, spermatogenesis and membrane function in eukaryotes.
Professor Frank Robb, a microbiologist at the University of Maryland, has been studying these HSPs for many years. His main interest is to learn specifically how thermophiles can stabilise their proteins when exposed to extreme temperatures. The search for thermophiles has taken Professor Robb to some exotic places, including terrestrial and marine volcanic springs.

A thermophilic microbial mat reveals several layers of anaerobic thermophiles beneath the photosynthetic biofilm covering the consortium
His working hypothesis is that organisms with well-developed HSPs would have been able to survive closer to vents, where they could grow and reproduce rapidly in the warm water. In contrast, organisms that were safe from sudden heating, far from the vent, would run the opposite risk of finding themselves in cold, barren conditions where they were unable to reproduce.
A specific group of protein aggregates called amyloid fibrils has been linked to more than 15 neurological human diseases, including Alzheimer’s and Parkinson’s Syndromes. These malfunctions are often called ‘conformational disorders’, many of which result from the conversion of a normal protein into a specific polymeric amyloid form. In an interesting study conducted by Professor Robb and his colleagues, the mechanism of deconstruction of amyloid fibrils was examined by looking at the disassembly of neurotoxic fibrils by chaperones both in the presence and absence of ATP – the vital molecule that transports chemical energy within cells.
This study indicated that HSPs adsorbed on the fibril surface formed clusters with no ATP present, at specific intervals on the fibril surface where these chaperones could unravel the fibril to release short fragments. Their motivation for using HSPs to disintegrate amyloid fibrils is based on the potential of these proteins to restore the form of native ones and normal biological activity. From these studies, Professor Robb and his team demonstrated a way to disperse fibrils with minimal increase in cytotoxicity.
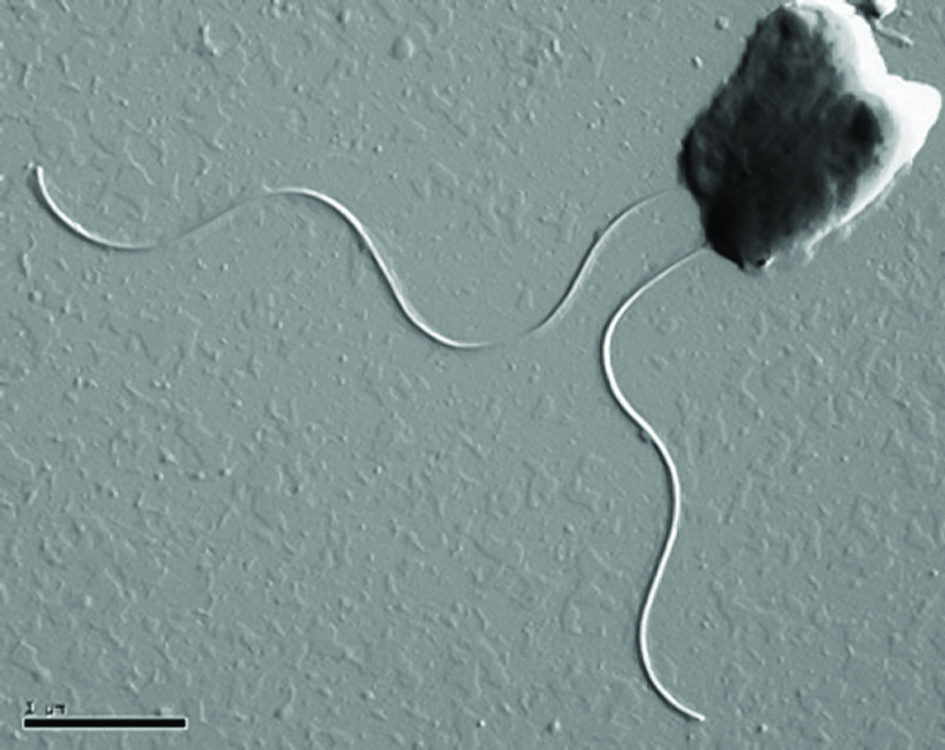
Electron micrograph of Nautilia nitratireducens, a bacterium isolated from a deep-sea hydrothermal vent
In a previous study, the team had constructed a cold-adapted mutant form of HSP60 from the hyperthermophile Pyrococcus furiosus, a bacterium that grows at temperatures of around 100°C. This organism had been isolated from the shallow marine volcanic hydrothermal vent systems. The results presented in this study were considered a potential advance towards the development of a treatment for protein conformation diseases in humans.
Other applications of Professor Robb’s current studies are methods to improve the durability and immunogenicity of vaccines through the expression of HSPs from thermophiles. He and his team are hoping to identify molecular processes that could enable the long-term storage of vaccines. This would be an important step forward for the supply of vaccines to remote locations that lack refrigeration, such as developing countries.
There is growing excitement in the field of thermophile molecular biology. Hydrothermal vents offer ideal conditions for the evolution of microbial metabolic processes. The evolutionary links between human and animal pathogens and their non-pathogenic deep-sea relatives studied by Professor Vetriani have revealed the role of genomic plasticity in the diversification of microbial life. Thermophiles are also model systems to study complex cellular functions relevant to human disease, such as protein folding and stress responses. Studies by Professor Robb on HSPs in thermophiles provide a better understanding of the progressive neurodegenerative stages in Alzheimer’s and Parkinson’s, making these proteins potential therapeutic candidates.
Meet the researchers

CREDIT: Nerissa Escanlar
Professor Costantino Vetriani
Department of Biochemistry and Microbiology
Institute of Earth, Ocean and Atmospheric Sciences
Rutgers University
New Brunswick USA
Professor Costantino Vetriani completed a MSc in Microbiology at the University of Rome La Sapienza and a PhD in Molecular Biology in 1994 at the University of Rome Tor Vergata. Before joining Rutgers University in 1997, Dr Vetriani was a guest investigator at Woods Hole Oceanographic Institution and Research Associate at the Center of Marine Biotechnology in Baltimore in Professor Robb’s laboratory. Research in his laboratory focuses on the ecology and evolutionary relationships of deep-sea prokaryotes and the microbial adaptations to extreme environmental conditions. He has published more than 60 peer reviewed papers and has participated in 20 oceanographic expeditions.
CONTACT
E: vetriani@marine.rutgers.edu
T: (+1) 848 932 3379
W: http://dbm.rutgers.edu/pages/Costantino_Vetriani.html
FUNDING
Vertex Pharmaceuticals
National Science Foundation
NASA Exobiology
NSF Center for Deep Energy Biosphere Investigations


Professor Frank T. Robb
Institute of Marine and Environmental
Technology, and Department of Microbiology and Immunology, School of Medicine
University of Maryland
Baltimore
USA
Professor Frank Robb received a BSc (Hons) from the University of Cape Town (UCT) in 1969 and a PhD from the University of California, Riverside in 1973. He completed postdoctoral studies at the University of California, San Diego and the University of Chicago. Before coming to the University of Maryland in 1988, he was Associate Professor at the University of Cape Town, South Africa. Research projects in his laboratory include protein folding mechanisms in hyperthermophiles and genomic studies on extremophilic Bacteria and Archaea. He has published in several high-impact journals including Nature and PNAS.
CONTACT
E: frobb@som.umaryland.edu
T: (+1) 410 234 8870
W: http://www.imet.usmd.edu/people/robb.html
FUNDING
NIST-IMET Program in Environmental and Marine Science
NASA Exobiology
Bill and Melinda Gates Foundation
PATH Foundation NSF Center for Deep Energy Biosphere Investigations (see:http://www.darkenergybiosphere.org/http://www.darkenergybiosphere.org/)


