Professor Etienne Sibille | Professor James Cook – Lifting Brain Fog
Effective treatments for cognitive dysfunction, such as declines in memory and other mental faculties often associated with depression or old age, may be within reach, according to Professor Etienne Sibille at the Centre for Addiction and Mental Health (CAMH) and the University of Toronto, Canada. Professor Sibille has shown for the first time that newly synthesised compounds targeting GABA receptors improve specific types of memory in mice, opening the door to the development of effective new pharmacological options.
Cognitive dysfunction occurs alongside many psychiatric disorders, and as we advance in years a large proportion of us will experience some degree of age-associated cognitive impairment. Symptoms such as confusion, inability to focus attention, memory lapses, or even poor judgement, can make day to day living far more challenging than it needs to be. For many, the experience creates additional stress while decreasing quality of life.
Even if we haven’t been touched by the effects of cognitive dysfunction directly, it is likely that each of us knows someone who has struggled at some point in their life with these kinds of symptoms. For instance, while the focus is mostly on mood, the majority of people with depression also suffer from cognitive dysfunction even when they are in remission. The number of people suffering from cognitive impairment of some form is estimated to be over 600 million worldwide, but effective treatment has been elusive. The impact of cognitive dysfunction isn’t just a personal one, it can be wide-reaching, affecting relationships, ability to work, or even effectively manage self-care.
Despite this wide-reaching impact, scientists have yet to uncover a truly effective, well-tolerated treatment. The stated goals of Professor Sibille and his team at CAMH and the University of Toronto, Canada, are undoubtedly ambitious. As a collective, they aim to conduct cutting edge research into the neurobiology of aging and depression, and harness their discoveries to create truly effective treatments. In order to reach these goals, they employ large-scale genome, molecular, and bioinformatic approaches, as well as several animal models to test their hypotheses.
In recent years, this arsenal of methods has been turned to investigations of cognitive dysfunction across disorders and aging, to internationally recognised success. They’ve recently made promising steps towards an effective, well-tolerated pharmacological treatment for cognitive dysfunction, using a new approach which targets specific receptors in the brain of the neurotransmitter gamma-aminobutyric acid, also known as GABA.

Brain microcircuits: Targeting α5-GABAA-Rs for antidepressant, anxiolytic and pro-cognitive therapeutic efficacies. Glutamatergic pyramidal neuron (PN) excitatory information is controlled by various interneurons, including somatostatin- (SST), parvalbumin- (PV), and vasopressin- (VIP) expressing cells. Reproduced with permission from ACS Chemical Neuroscience
Target Practice
In the adult brain, GABA is typically associated with neuronal inhibition. More specifically, its action decreases the likelihood that a neuron will fire, and can, therefore, decrease the amount of activity in a given area. Inhibition is a vital process in the brain, offering a balance to neuronal excitation, and keeping everything in working order.
When inhibitive processes function atypically, our brain starts to run into problems. In neuropsychiatric and aging research, a reduced inhibitory activity is often identified as playing a significant role in cognitive dysfunction.
As Professor Sibille explains in a recent paper, at a cellular level, this inhibitive action is created by different types of GABA-ergic interneurons – cells responsible for GABA-release and communications between neurons. These influence the activity of other neurons, such as excitatory pyramidal neurons, which are central to coding information in the brain and to overall cognitive processing.
Not only that, but GABA-ergic interneurons come in different types, express different proteins, and are involved in various activities, each of which with differing therapeutic potential. Professor Sibille’s group has now identified one of these neuron subtypes as a weak link in the brain, showing they are specifically vulnerable in brain disorders and aging, and that these neurons are involved in cognitive processing.
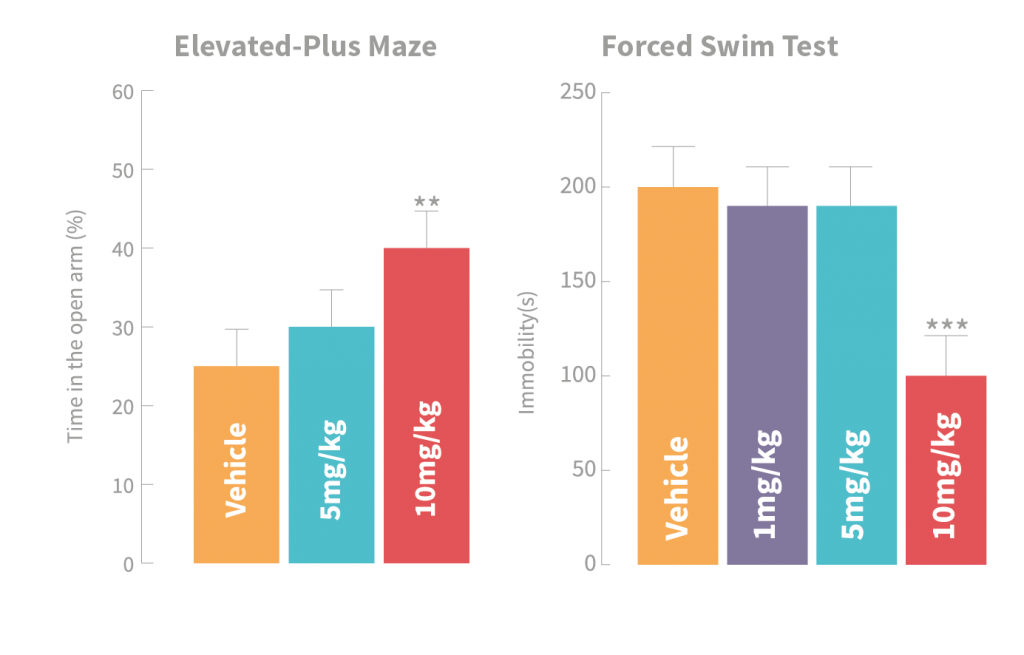
Acute treatment of GL-II-73 induces anxiolytic effect in the elevated plus maze (left panel),and antidepressant effects in the forced swim test (right panel) in mice and show significant differences compared to Vehicle group. Reproduced with permission from Karger Publishing
Current therapeutics that aim to improve inhibitory processes leave a lot to be desired. Benzodiazepines interact with many types of GABA-ergic neurons and GABA receptors in a non-specific manner, meaning that the drugs have wide-ranging anxiolytic, sedative, anticonvulsant, and amnesic properties. While some of these effects are desirable, the side effects created by this kind of non-specific binding have limited their therapeutic ability. Many find the drowsiness created by treatment with benzodiazepines to be too intrusive to justify continuing these types of medication.
However, Professor Sibille and medicinal chemist colleague, Professor James Cook, from the University of Wisconsin in Milwaukee, USA, assert that by narrowing the action of a drug to specific types of GABA receptors, they can target the function that is specifically affected by the weak cells and also minimise the side effects. To this end, they have now identified which type of GABA receptor can be targeted to directly treat cognitive dysfunction, and have demonstrated the effectiveness of this approach.

Of Mice and Mazes
Professor Sibille and Dr Thomas Prevot, a behavioural pharmacologist working in his group, hypothesised that by activating α5-GABA receptors, they could rescue the identified weak interneuron link by normalising their inhibiting activity, and alleviate the associated cognitive dysfunction using their rodent models.
In order to test this, they designed and created three new ligands – small molecules that attach to receptors and facilitate their function – that would target these receptors. These took the form of imidazobenzodiazepines based on a hybridisation of two pre-existing drugs: diazepam and flumazenil. Both of these drugs are recognised for their clinical safety and effectiveness, and are known to target GABA receptors, making them ideal candidates for this kind of work. Professor Sibille’s and Professor Cook’s teams then worked on refining these molecules so that they target more specifically the α5 subtype.
The team administered the newly synthesised ligands to mice that had been exposed to chronic stress conditions, as well as older mice, for one or 10 consecutive days. Both kinds of mice are regularly used as models for human mood disorders and cognitive dysfunction in this type of research. Any evidence that markers of cognitive functioning, such as working memory (information currently held in mind for immediate use), had been positively affected by these new ligands would provide solid reasoning to pursue further research into therapeutics produced in this manner.
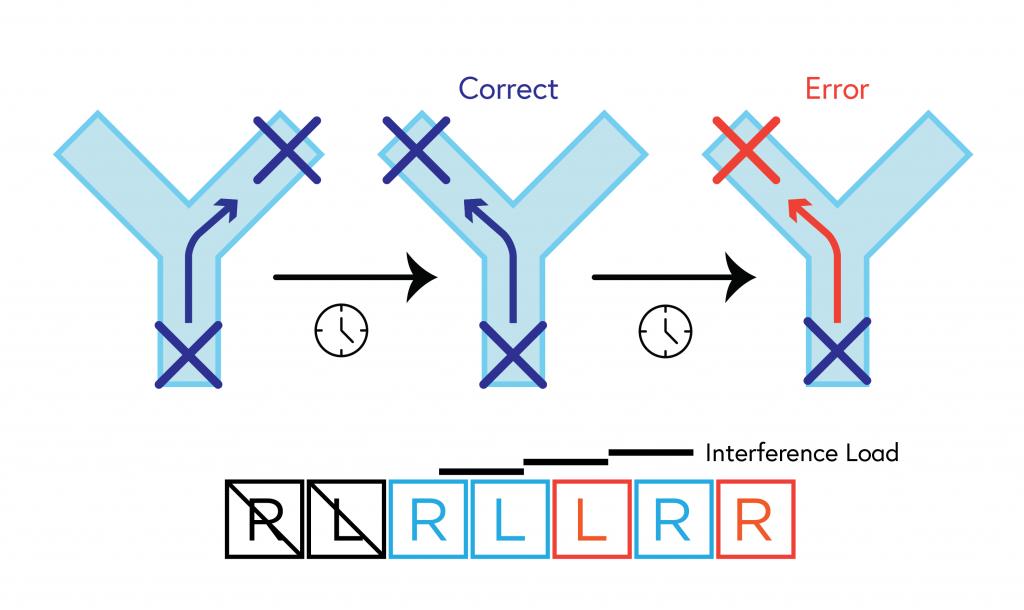
he Y Maze assesses spatial working memory. The mouse is placed in the start box and is free to choose between the left or right arm of the Y Maze. When an arm is chosen, the mouse stays in that arm for 30 sec and is then returned to the start box. After a delay, another trial starts in which the animal is supposed to alternate. Alternation is considered a correct choice, visiting the same arm as before is considered an error. With an increase in the number of trials, interference occurs making it difficult to remember the last trial because of the multiple trials previously performed. Lack of alternation is considered a marker for cognitive deficits. Credit Thomas Prevot.
To begin with, the mice were tested against controls who had been injected with either a placebo solution or diazepam for evidence of anxiolytic and anti-depressant effects. The researchers employed two tasks: the Elevated Plus Maze and the Forced Swim Test.
The Elevated Plus Maze consists of two intersecting planks which create a ‘plus’ shape. Two of the arms of this plus are illuminated with bright lighting, and two arms are surrounded by an opaque wall that keeps the level of light low, and allows rodents to feel more secure. In animals that are anxious, more time will be spent in the less lit, covered areas, whereas those feeling comfortable will venture into brightly lit spaces for longer periods of time. Following ligand administration, rodents in this test spent significantly more time in the bright areas, suggesting lower levels of anxiety, and therefore, anxiolytic effects of the newly created ligands.
Similar positive effects were found in the Forced Swim test, where high amounts of time spent still in the water are interpreted as depression-like behaviours. The rodents that had been given the novel ligands spent significantly less time stationary, demonstrating the potential anti-depressant effects of the new ligands.
While these findings alone are promising, the results of tests probing cognition were even more dramatic. When testing cognitive dysfunction in the mice, researchers employed a classic Y-maze task. As the name suggests, mice are placed alone in a ‘Y’ shaped maze comprising three arms, each placed at 120 degrees to the others. This task is used to test spatial working memory, that is, memory about one’s environment. As the maze is very simple, rodents with typically functioning working memory easily alternate between arms on successive explorations. However, rodents with impaired working memory are more prone to forgetting where they’ve already explored. This results in a marked increase in random wandering between arms, indicating the extent to which working memory may be impaired. This is what is observed in old mice or in mice exposed to chronic stress.
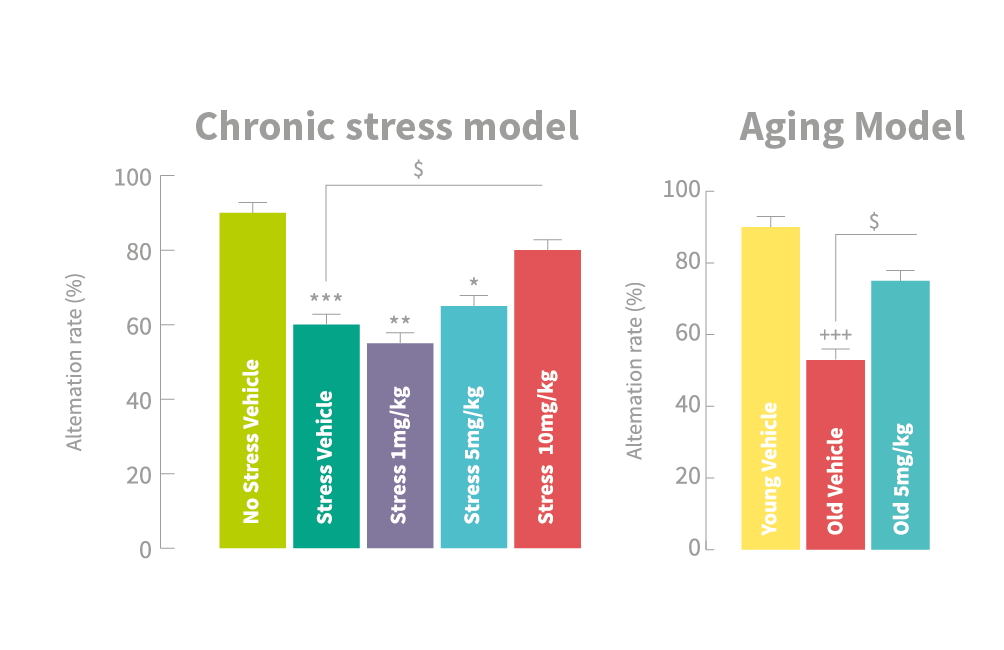
Stress (left panel) or aging (right panel) alter the cognitive performances in a working memory task, but such impairment is reversed by the use of the appropriate dose of GL-II-73 and show significant difference compared to Vehicle groups (no stress or young); $ show significant difference compared to Stress-Vehicle or Old-Vehicle group.
In their study, both the stressed and aged rodents dosed with two of the novel ligands had a significantly higher alternation rate than stressed and aged rodents that had received control non-active substances. Dr Prevot notes that in these tests, old mice treated with the novel ligands were performing near the level of younger, non-impaired mice. This seeming lack of disorientation indicates improvements in the way they were able to store and process information about the maze. By employing ligands that specifically targeted α5-GABA receptors, the researchers were able to significantly improve upon cognition in their rodent models of cognitive dysfunction.
Further inspection of the mouse tissues revealed that pyramidal neurons supporting information processing in the brain and that are associated with cognitive function, responded to the ligand treatment with new growth, effectively reversing the shrinking of brain cells that occurs during normal aging, and improving the connections and possibly the function of these cells on a physical level. This is particularly promising, as it suggests that the compound may be suitable for adaptation into a medication that could be taken long-term to ensure efficacy over time.
Into the Future
Professor Sibille and his colleagues believe that these findings present a strong argument for the development of novel therapeutics which target α5-GABAA receptors as a potential treatment for cognitive dysfunction across brain disorders and during aging. With this information in hand, his studies are likely to have a far-reaching effect on how we approach the treatment of cognitive dysfunction. Given the novelty of their approach, the findings of the lab may represent a coming paradigm shift in psychiatry.
Cognitive dysfunctions are associated with depression, suicidality and other neuropsychiatric conditions and are prevalent during aging and age-related brain disorders. These continue to rise, already playing a negative role in the lives of many. With this research, Professor Sibille and colleagues aim to actively pursue the creation of α5-GABAA receptor targeting drugs to tackle cognitive impairment. They hope to create more palpable medications with reduced side-effects, and in doing so, improve the lives of millions of people with various memory and other cognitive problems.
Reference
https://doi.org/10.33548/SCIENTIA384
Meet the researcher

Professor Etienne Sibille
Centre for Addiction and Mental Health
Toronto and Department of Psychiatry
University of Toronto
Toronto, ON
Canada
Professor Etienne Sibille received his PhD in Pharmacology in 1999 from Cornell University Medical College, New York, then proceeded to rise rapidly through the academic ranks at the University of Pittsburgh, Pennsylvania. In 2014, he accepted the appointment of Professor at the University of Toronto and that of Senior Scientist at the Centre for Addiction and Mental Health, Toronto. From 2017, Professor Sibille has also been the Deputy Director of the Campbell Family Mental Health Research Institute, Toronto. Professor Sibille investigates the neural mechanisms of depression and aging, and develops novel treatments for associated cognitive dysfunction. He is a highly esteemed neuroscience researcher, having published more than 100 peer-reviewed papers across a range of high-impact journals, and is the recipient of many prestigious grants and awards, commensurate with his ongoing contribution to science.
CONTACT
W: https://www.sibillelab.com/; etienne.sibille@camh.ca
T: @esibille1000
FUNDING
Campbell Family Mental Health Research Institute
NARSAD
Farcombe Institute
Canadian Institutes of Health Research
National Institutes of Health
Centre for Addiction and Mental Health
American Foundation for Suicide Prevention

Professor James Cook
University Distinguished Professor
University of Wisconsin–Milwaukee
Milwaukee, WI
USA
Professor James Cook received his PhD in organic chemistry in 1971 from the University of Michigan, USA. After completing a postdoctoral fellowship at the University of British Columbia, Vancouver, he took up a position at University of Wisconsin–Milwaukee, USA, and remains there to this day, currently in the role of University Distinguished Professor. Thus, Professor Cook has directed a university research laboratory for more than 30 years, focusing his research on synthetic organic and medicinal chemistry, and confirming his specific interest in compounds that modulate central nervous system neurotransmission. Over his illustrious career, he has published more than 500 peer-review journal articles and has filed over 60 patents, many of which have been issued. He has maintained an active collaboration with Professor Sibille since 2012.
CONTACT
W: https://uwm.edu/chemistry/people/cook-james/
FURTHER READING
TD Prevot, G Li, JM Cook, E Sibille, Insight into Novel Treatment for Cognitive Dysfunctions across Disorders, ACS Chemical Neuroscience, 2019, doi: 10.1021/acschemneuro.9b00148/
TD Prevot, G Li, Novel benzodiazepine-like ligands with various anxiolytic, antidepressant, or pro-cognitive profiles, Molecular Neuropsychiatry, 2019, 5, 84–97.
![]()

Creative Commons Licence
(CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
More articles you may like
Grandmothers: Innovation Through Tradition
Grandmother Project – Change through Culture (GMP) is an organisation dedicated to documenting the role of grandmothers and demonstrating the effectiveness of grandmother-inclusive strategies in improving the health and well-being of women, children, and adolescents. GMP’s groundbreaking work challenges conventional wisdom to transform community-based interventions in Africa and beyond, harnessing a powerful but often overlooked resource: the wisdom and influence of grandmothers.
Dr Robert Larkin | Cultivating Change to Improve Soil Health and Increase Potato Yield
Environmental quality and food production are facing the pressing challenges of climate change and global population growth. Dr Robert Larkin from the United States Department of Agriculture-Agricultural Research Service (USDA-ARS) and a team of plant scientists developed and tested a range of crop management systems to help overcome these compounding challenges. Their work is improving soil health and increasing the yield of potato crops, contributing to the future food security of nations.
Professor Giorgio Buttazzo | Artificial Intelligence and a Crossroads for Humanity
Where do we stand with artificial intelligence? Might machines take over our jobs? Can machines become conscious? Might we be harmed by robots? What is the future of humanity? Professor Giorgio Buttazzo of Scuola Superiore Sant’Anna is an expert in artificial intelligence and neural networks. In a recent publication, he provides considered insights into some of the most pressing questions surrounding artificial intelligence and humanity.
Dr Ralf Adam | New Technologies Shaping the Future of Oral Hygiene
Understanding the efficiency of various toothbrush technologies is essential for achieving optimal oral health. Dr Ralf Adam, who leads a dedicated team at Procter & Gamble in Germany, is keen to investigate the complexities of these technologies. His team have provided new insights into the best toothbrush types for plaque removal and the maintenance of gum health. By highlighting the importance of informed oral care decisions and ongoing investigations, this vital research works towards ensuring everyone can achieve a brighter, healthier smile.




