Professor Irva Hertz-Picciotto – The Conception Of Autism Spectrum Disorders
Professor Irva Hertz-Picciotto has been fascinated by early development since childhood. Her interest brought on a life long journey, studying the dangers lurking around the corner for the not yet born – now focusing on risks for autism spectrum disorders.

Among your multiple other epidemiological interests, the focus on environmental effects on pregnancy outcomes has followed you from the very start of your career. What spurred this interest in the first place?
When I was 11, my mom announced that she was pregnant. I was incredibly excited and curious about what was happening inside her! I went to the library to find books but what I found did not provide enough to satisfy me. My brother was born, and I loved being the big sister of a tiny baby. Shortly thereafter, the newspapers and television were showing pictures of deformed babies, who had been exposed to thalidomide – a medication given to pregnant women to treat nausea. It was shocking, and drove home for me how vulnerable the foetus is, and that some are luckier than others.
What inspires you in your work?
A few things energise me. One is my deep concern for children, who need so much from the rest of us – being fully dependent on, and intensely affected by the people who take care of them. A second motivator has been the sense of urgency felt by many families dealing with a child who has an autism spectrum disorder. Parents are justifiably frustrated at the lack of answers about what caused the disorder, and what they can do to make their child’s life the best possible. Third is the energy and creative thinking of the students and post-docs that I have been privileged to mentor and work with. Fourth is the sheer pleasure of delving into the process of scientific discovery and challenging my brain to tackle big problems that concern real people by searching for a new perspective.
You have shown that there is an increased risk of autism spectrum disorders following exposure to substances commonly used in our everyday lives. Are law and policymakers regulating use of these substances in a timely manner?
Overall, there is a bias in our society that favours rapidly moving new products onto the market. Once on the market, it is a completely different story when questions arise regarding adverse effects on health from industrial or consumer products: action is usually very slow. There is a high bar for eliminating or restricting use or production of the chemical agents under scrutiny. In fact, we have a dual problem. First, there are thousands of chemicals that were grandfathered in when regulation began to address potential health effects by requiring testing, but mainly for new products. Second, the testing that is required is inadequate. Under certain laws in the U.S., testing of new chemicals covers cancer, gross malformations, and acute toxic effects, but does not examine long-term adverse developmental effects stemming from prenatal or early life exposures. Additionally, the problems of multiple exposures and their interactions remain a major gap. We have little or no information about the impact of a complex environment in which we are exposed, simultaneously, to a multitude of compounds, and whether the combined effects exceed what one might expect from simply adding up the individual influences on health or development. Thus, tens of thousands of chemicals are on the market for which we cannot say that they are safe for foetuses and small children, either individually or in combination. Moreover, our regulatory apparatus is vulnerable. It is subject to serious pressures from stakeholders representing financial and political pressures who often succeed in delaying action to restrict or eliminate chemicals for years or even decades – even when abundant evidence of toxicity has accrued. In short, regulation of toxins that influence brain development has been slow, some say glacial, leaving many children unprotected.
Your studies also focus on gene – environment interactions. You have shown that in children with a variant COMT gene, associated with reduced efficiency for the repair of our DNA and the neurotransmitters necessary for brain function, folic acid supplementation reduced autism risk by 82 per cent. How can this data be used to target at risk children?
Yes, there appear to be several genetically defined subsets of the population in which we found folic acid supplementation to have quite a huge protective effect against the development of autism spectrum disorder, or ASD. I would hasten to add the caveat that this particular genetic variant of the COMT gene that you mention was fairly rare in the children. The other gene with a pronounced protective effect in combination with folic acid supplementation was found in the mother: her MTHFR677a. For public health prevention, we might do better targeting the mom before she has even conceived her child, since the benefits of supplementation with prenatal vitamins are primarily for the three months before and the first month of pregnancy. Before embarking on such a targeted direction, however, replication of the roles of those genes is needed. At this stage, the first step would be to replicate these results. I have not yet seen an attempted replication: either some researchers have tried but decided not to publish their results, or no one has yet examined these gene-byenvironment interactions.
If another team does succeed in replicating the gene-by-environment interaction, then your question about how to move from scientific evidence to a real intervention on those at elevated risk would become a high priority. A possible recommendation to consider is for all girls, when they reach reproductive age, to be tested for these genetic variants. Those with the high risk genetic profile could be put on prenatal vitamins for their entire reproductive years. Alternatively, this could be suggested only for those planning a pregnancy, but about half of pregnancies are not planned.
You have received the Goldsmith Lifetime Achievement Award from the International Society for Environmental Epidemiology. What did this mean to you privately and professionally?
I am a firm believer that the legacy of someone in my position lies at least as much in those we train and mentor, as it does in the specific scientific discoveries that we might make. After all, in 20 years, our papers may hardly be noticed, being supplanted by newer and better research methods. Our students, postdocs, and other faculty that we may influence are the ones who carry the torch, including the passion for doing rigorous science, and hopefully they pass the inspiration to the next generation.
ENVIRONMENTAL-METABOLICGENE INTERACTIONS IN AUTISM
Professor Irva Hertz-Picciotto’s concern for children has led her to launching the MARBLES and CHARGE studies, two large-scale projects that may trigger a paradigm shift in the way scientists view environmental risks and early markers of autism spectrum disorders.

Our current understanding of what causes autism
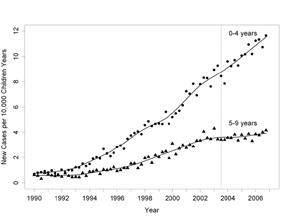
In 2013, it was estimated that almost 22 million people live with autism, a neurodevelopmental disorder characterised by a set of core symptoms that include impairments in communication ability and in social interaction, and presenting in the first three years of life. However, individuals with this disorder manifest varying degrees of severity, comorbidities and idiosyncratic behaviours. Despite this variation, however, all autism types are encompassed by the umbrella term: autism spectrum disorders (ASD). Autism rates have increased during recent decades, partly due to greater awareness and improved diagnosis and reporting of ASD. However, Professor Hertz- Picciotto found that a substantial proportion – potentially more than half – of the increase cannot be attributed to younger age at diagnosis, changes in clinical practice and/or broadened diagnostic criteria.
After 15 years of research and over a billion dollars invested in studies of genetic risk factors for ASD, the scientific community has identified a large number of rare and very rare variants, adding substantially to the body of knowledge about etiology of this condition. Using databases documenting the functions of genes and linking them to molecular mechanisms, the field has moved from specific genes to biologic pathways. For instance, in the case of ASD, scientists are confident that aberrant synaptogenesis – the formation of junctions where one neuron transmits its message to the next one – plays a role in the brains of children with ASD. Yet heritability studies have increasingly shown that, as compared with genes, environmental factors play an equal or greater causal role in ASD. Based on the complexity of ASD and decades of research on other complex conditions such as heart disease and cancer that repeatedly established links with both genetic and environmental factors, Professor Hertz-Picciotto reasoned that early life exposures likely contributed to ASD. Moreover, she realised that a search for modifiable environmental factors was necessary in order to lay the foundation for intervention that could ultimately halt the steady rise in autism and potentially decrease severity of impairments in those affected. From this line of thinking emerged the complementary CHARGE and MARBLES studies, seeking to explore the impact of early life exposures, both maternal and postnatal, on the emergence of autism. To date, these studies have focused on several areas such as maternal nutrition, metabolism, and exposure to certain substances during pregnancy.
A possibly disturbed metabolic pathway
The CHARGE Study takes a retrospective approach. Specifically, children with either ASD or typical development are identified at the same ages, and then compared, with data collected to learn about differences in their early life exposures. The MARBLES study follows children from long before birth to their third birthday, an age when a firm diagnosis can usually be made. MARBLES recruits pregnant mothers who already have had an autistic child and therefore have an increased risk of a second affected child. The study also aims to identify markers of autism in infants, because, first, an early diagnosis facilitates severity-decreasing interventions that can improve the skills and autonomy of the children, and second, a connection of such markers to environmental exposures may hold the key to understanding both environmental causes and their mechanisms. The MARBLES study team therefore collects blood, urine, hair, placental tissue, and breast milk to both measure the exposures and explore biomarkers on potential pathways through which they may influence the development of the foetal and infant brain.
The data in the CHARGE study demonstrated that mothers of children with ASD had a different metabolic profile during pregnancy than those of typically developing children, even after adjusting for a wide array of potential confounders, encompassing maternal demographic and socioeconomic characteristics, as well as child’s sex and age. The metabolic conditions included type 2 diabetes, gestational diabetes, and obesity, and the CHARGE team also validated these from prenatal medical records. Four independent groups corroborated the findings of CHARGE, including several very large studies, providing a robust body of evidence and thus reinforcing the idea that a disrupted metabolism can influence infant brain development through direct or indirect pathways.
Inflammatory processes produced by several types of immune cells, fat cells, and other types of tissue produce inflammatory proteins. One of Professor Hertz-Picciotto’s working hypotheses was that, besides dysregulation of insulin and resulting impacts on cell energy metabolism, the maternal production of inflammatory proteins can alter immune signalling in the child. Because immune cells are abundant and in tight communication with neurons in the brain while it is undergoing exceedingly rapid growth, neuro-inflammation is a potential and plausible pathway linking metabolic conditions to ASD. Further research by Professor Hertz-Picciotto’s team and others detected antibodies against the foetal brain that are found in blood of over 15% of mothers of ASD affected children but less that 2% of those with typical children. Despite mounting evidence regarding the link between maternal immune activation and autism, scientists are not yet able to draw a definitive conclusion as to whether the maternal immune alterations found in humans are causal of ASD, or her immune disturbance is a consequence of the disorder, or the child’s ASD and the maternal immune changes have no direct connection and are simply connected through a common upstream factor.
To further pursue this question, Professor Hertz-Picciotto obtained new-born blood from CHARGE children and analysed cytokine levels – proteins that act to either promote or dampen inflammatory responses. Her team was able to confirm the association between elevated levels of the pro-inflammatory cytokines IL-1ß and IL-4 (interleukins) at birth and subsequent increased risk of ASD, nailing down one link in the chain she hypothesised. Additionally, maternal metabolic conditions clearly precede both the presence in the new-borns of these proteins, and the development of autism, thus hinting at the possibility to demonstrate causation in future work.
 Maternal nutrition may reduce risk
Maternal nutrition may reduce risk
A second set of studies by Professor Hertz-Picciotto’s team has focused on maternal nutritional status. Mothers in CHARGE reported on their use of vitamin supplements and their intake of grains and cereals that are rich in the B-vitamins. Interestingly, those mothers who took the recommended prenatal supplements before or during the first month after conception had a 40% lower likelihood of having a child with ASD! These prenatal vitamin supplements are formulated with folic acid, and the recommendations to take them before and early in pregnancy, are aimed at reducing the occurrence of neural tube defects, a type of birth defect in which the brain does not form properly in the embryonic period. Although retrospective maternal report can sometimes be unreliable, these CHARGE study results have now been replicated in a larger study that collected information about use of prenatal supplements prospectively, i.e., before the mother knew about her child’s diagnosis. Not only did women from another population experience lower risk if they took the prenatal supplements, but the window of protection was consistent with what was observed in CHARGE.
However, the CHARGE study went further: mothers and children were genotyped for targeted genes involved in metabolism of folic acid to folate and in the role of folate as a donor of methyl groups. Methyl groups, consisting of a carbon linked to three hydrogen atoms, are epigenetic marks that sit outside the DNA code and regulate gene expression. They are also essential for production of neurotransmitters, chemicals such as dopamine and serotonin, that carry signals across synapses. In the CHARGE Study, gene variants that confer a less efficient conversion of folic acid to folate (e.g., MTHFR677A), and other gene variants that impart a less efficient transfer of methyl groups to DNA (e.g., COMT), were associated with the greatest reduction in risk from folic acid intake. This finding is significant for several reasons. First, it represents the first gene-by-environment interaction to be discovered in the field of ASD etiology. Second, it identifies a genetically vulnerable subset of the population that may gain virtually all of the benefit from an environmental – in this case nutritional – intervention. Third, it is actionable and extends the positive impacts of nutritional supplements that are already strongly recommended. Fourth, it identifies a critical window around the time of conception for which nutritional supplementation may reduce risks for autism. Fifth, it supports sitespecific epigenetic mechanisms (i.e., DNA methylation for particular genes) in ASD etiology.
Environmental hazards in CHARGE and MARBLES
Humans are exposed to environmental chemicals in air, water, food and soil, as well as from a wide variety of household and personal care products. For example, pesticides are present as residues in foods, including fruit, vegetables, dairy, meat and fish. Other sources are commercial applications in agriculture, golf courses and around highways that result in drift at substantial distances (e.g., 5-10 km); indoor applications at malls, and household uses. Pesticides are notably different from most other environmental pollutants such as metals, plasticizers, and flame retardants, in that their primary purpose is to kill animals of other species, usually by acting on nerve cells to disrupt ion channels that regulate transmission of messages across the synapse. This type of action is also relevant to human beings and hence it should not be surprising that most pesticides are neurotoxic. However, until recently, most information on pesticide neurotoxicity pertained to its short-term acute effects, rather than long-term impact from prenatal exposures, a period when half a million cells in the foetal brain are dividing every minute.
To address this developmental window, Professor Hertz-Picciotto used the CHARGE Study to examine the impact of living close to agricultural applications by mapping the residences before and during pregnancy. These addresses were then linked with a database of recorded use on crops by commercial applicators. Professor Hertz-Picciotto then showed that the children in families living near applications of certain pesticide classes during gestation were more likely to develop autism than children from families living further away. These classes included organophosphates, which in the U.S., are now banned for residential uses, and pyrethroids, a group of synthetic mimics of the naturally occurring pyrethrins; the pyrethroids are far more potent than the pyrethrins.
With these results, along with suggestive reports from other populations, Professor Hertz-Picciotto is now turning to the MARBLES Study to measure biomarkers of exposure to the pregnant mother. Multiple urine specimens have been collected in each trimester for determining both organophosphate and pyrethroid metabolites. Because these compounds have short half-lives before being excreted, a single urine sample measurement can be misleading as to the level of exposures throughout the gestational period.
While some people might argue that short-lived compounds pose little or no threat to health, Professor Hertz-Picciotto points to the role of these substances during critical development periods: ‘Biologic activity does not require a long-term continuous exposure. For embryonic and foetal development, or even early childhood, a short-term exposure can have a profound effect if a critical process is taking place during that time window’, she notes. ‘Moreover, many short-lived compounds are used repeatedly. This is especially true of certain household and personal care products, for example, phthalates, which are present in vinyl flooring and in most scented products such as air fresheners, hand and body lotions, and shampoos.’
 Why it matters
Why it matters
Taking a step back from her large program of research on pre- and post-natal neurodevelopment, Professor Hertz-Picciotto comments that ‘many of the chemicals of concern appear to affect multiple organ systems. In other words, the same chemicals can interfere with early child brain development, influence metabolism to increase risk for obesity or diabetes, disrupt thyroid or sex hormones, and affect immune responses in the lungs. This multiplicity of effect underscores the need for a more pro-active approach to regulation of environmental chemicals.’ Unlike drug safety, pre-market testing for effects on the foetal brain has been minimal at best, and non-existent for the vast majority of the thousands of chemicals in commerce. Meanwhile, studies have repeatedly shown that new-borns already carry detectable levels of dozens of chemicals considered to have adverse health or developmental consequences.
By understanding the consequences of environmental exposure, finding the exact pathways by which these factors impact brain development, and detecting early signs of autism through the MARBLES study, Professor Hertz-Picciotto’s work aims to identify risk factors amenable to reduction, protective substances that can be promoted, and the broad field of co-factors that can be modulated. As she explains: ‘I am confident that we can substantially reduce the incidence of this disorder, and decrease the severity of symptoms in those who are genetically predisposed to develop autism spectrum disorders.’ Tackling the environmental exposures, from nutrition to chemical pollutants, and the epigenetic, immune, and metabolic mechanisms that can alter brain development, she hopes to one day see the rates of ASD finally decline to their pre-1990 levels.
Meet the researcher

Professor Irva Hertz-Picciotto
Professor, UC Davis MIND Institute
Director, UC Davis Environmental Health Sciences Center
University of California, USA
Starting out as a high school math teacher, Professor Irva Hertz- Picciotto found her true passion in Environmental Epidemiology later in life. She now directs the Program in Environmental Epidemiology of Autism, at UC Davis MIND Institute. She is the founder and director of CHARGE, the first large, population-based study to identify risk factors, amenable to intervention, for autism spectrum disorders, and MARBLES, a study searching for early biomarkers of autism. She has over 300 publications investigating effects of environmental factors on pregnancy outcomes and early child development. In 2011, she received the Goldsmith Lifetime Achievement Award from the International Society for Environmental Epidemiology.
CONTACT
T: (+1) 530 752 3025
W: https://www.ucdmc.ucdavis.edu/mindinstitute/ourteam/faculty/ picciotto.html
KEY COLLABORATORS
Rebecca Schmidt, UC Davis
Deborah Bennett, UC Davis
Robin Hansen, UC Davis
Cheryl Walker, UC Davis
Judy Van de Water, UC Davis
Daniel Tancredi, UC Davis
Lora Delwiche, UC Davis
Paula Krakowiak, UC Davis
Janie Shelton, UC Davis
Isaac Pessah, UC Davis
Paul Ashwood, UC Davis
Sally Ozonoff, UC Davis
Flora Tassone, UC Davis
Janine Lasalle, UC Davis
Kathleen Angkustsiri, UC Davis
Heather Volk, University of Southern California
Rob McConnell, University of Southern California
Li-Ching Lee, Johns Hopkins University
Rebecca Harrington, Johns Hopkins University
Dan Campbell, University of Southern California
Aake Bergman, Stockholm University
Santhosh Girirajan, Penn State University
Scott Selleck, Penn State University
Evan Eichler, University of Washington
FUNDING
National Institute of Environmental Health Sciences (NIEHS)
Environmental Protection Agency (EPA)
Science to Achieve Results (STAR) program
The M.I.N.D. Institute
Autism Speaks
Cure Autism Now
The Allen Foundation
National Institute for Occupational Safety and Health
National Institutes of Health (NIH)

