Professor Jeff Keelan – Preventing Preterm Birth and Improving Infant Outcomes
Professor Jeff Keelan and his colleagues at the Western Australian Preterm Birth Prevention Initiative are on a mission to lower rates of child morbidity and mortality through research into the prediction, treatment and prevention of premature labour and birth.
Landmark Initiative in Lowering Prematurity
The leading cause of death and disability in children under the age of five isn’t a contagious disease or genetic condition – it’s complications associated with preterm birth (PTB). The rate of PTB, defined as the birth of a baby at less than 37 weeks’ gestation, is around 5–8% in developed regions such as Europe and Australasia, but is much higher (between 16 and 18%) in developing areas such as sub-Saharan Africa or SouthEast Asia. According to the World Health Organisation, nearly one million infants’ lives are lost annually due to PTB. Unfortunately, despite intensive research, the rates of PTB have remained static for decades – and have even risen in some populations.
Much of Professor Jeff Keelan’s research is affiliated with the Western Australian Preterm Birth Prevention Initiative which was launched in 2014 under the leadership of leading Maternal-Fetal Medicine specialist, Professor John Newnham. Made up of a team of clinicians, researchers, public health professionals and patient advocate groups, the aim of the Initiative was to safely reduce the rate of PTB in the state by a third over a five-year period. This comprehensive programme encompasses new clinical guidelines, a cervical length screening program, an outreach programme for healthcare practitioners, a public health scheme for women and families and a new dedicated PTB prevention clinic for referral of high risk pregnancies. The initiative, supported by the Women & Infants Research Foundation, also incorporates ongoing research into the prediction, treatment and prevention of prematurity and PTB. A population study assessing the effects of the Initiative, which was just published in the American Journal of Obstetrics & Gynecology, showed an 8% reduction in the rates of PTB after just 18 months of operation – the lowest rate in the state in six years and a major achievement.
Despite the achievements of the Initiative to date, current strategies do not target key aetiologies such as infection and inflammation, and available pharmacological interventions have had little impact on the preterm birth rate to date. In order to develop effective therapies, Professor Keelan and his team are focussing on two challenges: firstly, they must determine how best to identify the women who are at risk of PTB, and secondly, they must develop effective and safe ways to treat amniotic and fetal infection and inflammation.
Infection and Preterm Birth
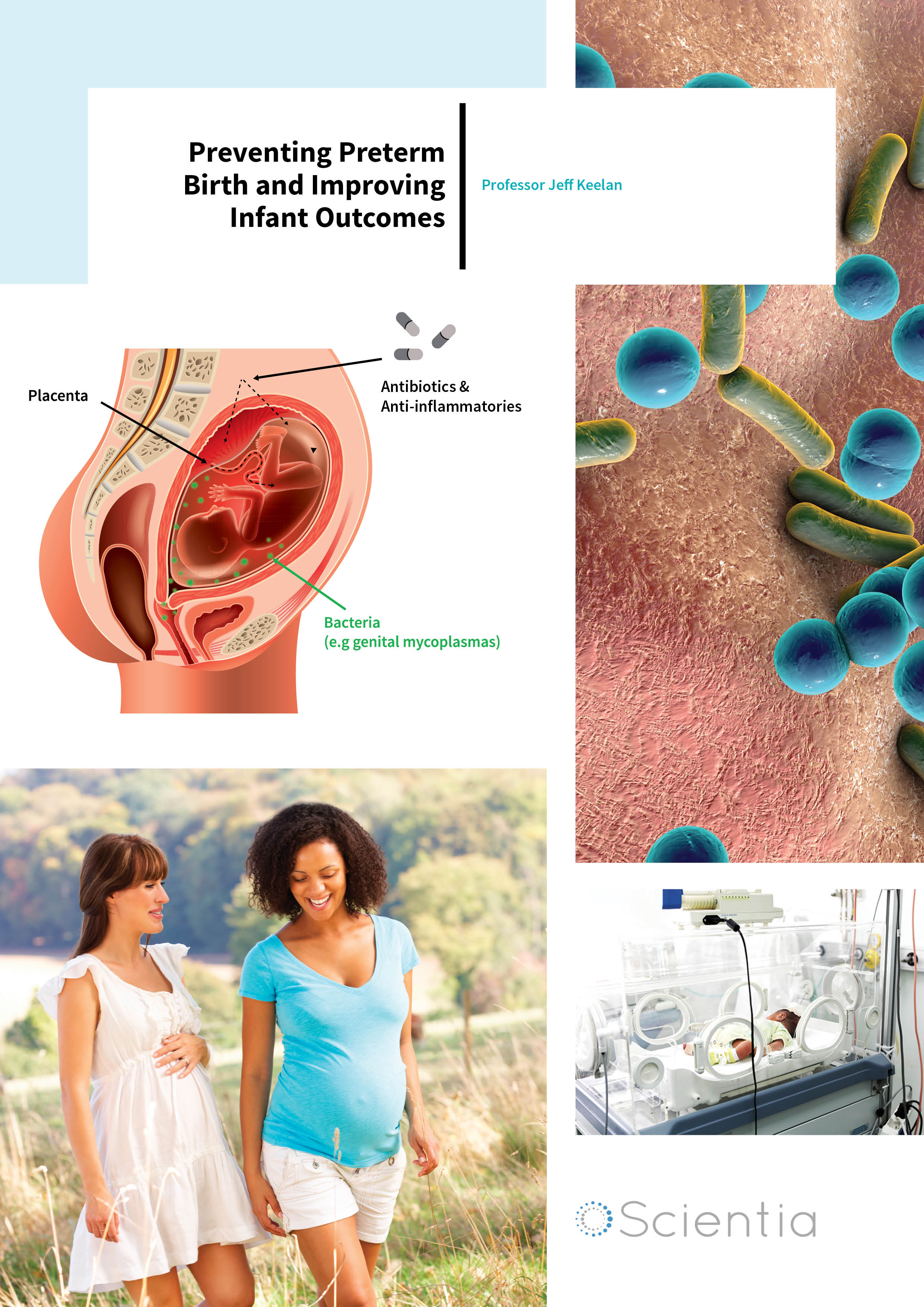
So what triggers PTB? Well, there are many different causes, but in about 10–20% of cases, the cause is intrauterine infection (IUI). IUI is caused by microbial invasion of the amniotic cavity (the fluid-filled sac which protects the fetus during pregnancy). This occurs when the bacteria residing in the vaginal mucosa breach the cervical barrier and colonise the fetal membranes and amniotic fluid. This then triggers an inflammatory response from mother and fetus, which can lead to chorioamnionitis (inflammation of the fetal membranes), preterm contractions, rupture of membranes, fetal inflammatory reaction and a range of serious neonatal complications including cerebral palsy. Importantly, the risk of IUI increases markedly as gestational age at delivery decreases. The majority of extremely preterm babies (those born more than 12 weeks premature) have evidence of an infectious aetiology – these babies typically have the greatest risk of death or life-long disability.
But which pathogens are responsible? ‘A variety of bacterial culprits that commonly cause infection-driven preterm birth have been identified – some are relatively innocuous bacteria commonly found in the reproductive tract of pregnant women, some are more typically associated with the oral microbiota, while others are only found in women with abnormal vaginal flora,’ explains Professor Keelan. ‘However, the bacterial species most commonly isolated from the amniotic cavity with preterm birth are Ureaplasma, tiny microorganisms present in around 50% of pregnant women. It remains a mystery why, in some women, vaginal Ureaplasma colonisation – which is usually harmless and undetected – transforms into intraamniotic infection. However, it is known that the ascension process does not occur until the second half of pregnancy, which means that we have the first 20 weeks to give any antibiotic therapy and prevent the complications of IUI-associated PTB.’
Identifying Women at Risk
Before one can treat the infections leading to PTB, one has to know who to target for treatment. Most women with Ureaplasma, for example, will not deliver early, while the ‘classic’ vaginal microbial abnormalities are poorly predictive of preterm delivery, and only present in a small percentage of women. Antibiotics need to be administered conservatively in pregnancy in order to prevent antibiotic resistance and also to preserve the ‘healthy’ bacterial environment that is required for normal fetal development. Professor Keelan and his colleagues, based at King Edward Memorial Hospital in Perth, Western Australia, have been developing a test to identify the presence of high risk vaginal microbiota in mid-pregnancy based on molecular profiling of bacterial DNA at 18–20 weeks’ gestation. Through this microbial profiling, they believed it should be possible to identify and treat women who screen positive with appropriate antimicrobial drugs or probiotics to prevent IUI and its consequences. This novel test employs polymerase chain reaction (PCR) to detect and quantify a specific set of bacterial DNA sequences in self-collected vaginal swabs. Excitingly, their research has identified a specific genotype of Ureaplasma that is associated with increased risk – but only when present in combination with two other bacterial species. Based on these observations, the team has developed a new microbial DNA test that is far more sensitive and specific than standard microbiological methods for identifying women destined to deliver preterm. Moreover, the test allows the identification and treatment of women positive for Ureaplasma at high risk of PTB, for the first time. The team plans to commence trials to prevent IUI-driven PTB based on screening of women using the bacterial DNA test in the near future.
Novel Antibiotic Therapy Against PTB
As it stands currently, trials that have given antibiotics to women to prevent PTB have had limited success. There are several reasons why antibiotics have been ineffective: many antibiotics do not cross the placenta at sufficient levels to kill particular bacteria; antibiotics were administered too late in the pregnancy to prevent infection; antibiotics were ineffective against the specific organisms associated with IUI; and finally, there has been little attempt to mitigate inflammation in the fetus or amniotic cavity.
The team has identified a new antibiotic, called solithromycin, which has great potential for preventing and treating perinatal infections. The drug was created by a small USA-based biotech company, Cempra Inc., by adding three molecular groups to a well-known macrolide antibiotic – clarithromycin – in order to change the uptake of the drug and its ability to overcome resistant bacteria. Professor Keelan’s team discovered that solithromycin is unique in that it can cross the placenta much more efficiently than other macrolide antibiotics. Using a pregnant sheep model, they found that solithromycin was capable of reaching effective concentrations in the fetal circulation and amniotic fluid within four hours of a single maternal dose. These findings were subsequently confirmed in a human placental model. Solithromycin is active against all the major microorganisms associated with PTB, including Ureaplasma, making it an effective treatment for IUI that can be administered orally. It is also highly effective against a wide variety of antibiotic resistant bacteria, and has the added bonus of being an effective anti-inflammatory agent.
Professor Keelan explains the potential of the new drug: ‘We’ve done some measurements in sheep and human placentas and estimate that the crossover is about 50% compared to just 2–4% for the older antibiotics. It’s also 10 to 100 times stronger, and works on antibiotic resistant bugs. It really does open the door for the effective treatment of a range of intrauterine infections and offers significant advantages over existing antibiotics’. Their next step is to carry out preliminary studies in pregnancy to assess the degree of fetal exposure and also solithromycin’s effects of vaginal bacteria. In an unexpected turn of events, work on solithromycin was recently halted after the approval of the drug for the treatment of community-acquired pneumonia was blocked by the FDA on safety grounds. Professor Keelan and his team are waiting to hear from the manufacturer on their strategy going forward before progressing their studies.
Exploring Cytokine Supressive Anti-inflammatory Drugs
While intrauterine infection is undeniably an important and preventable cause of PTB, inflammation without signs of infection is almost twice as common in preterm deliveries. Inflammation is a normal part of labour and delivery, but in some pregnancies, it is excessive and causes early birth. The triggers of so-called ‘sterile inflammation’ may include oxidative stress, placental breakdown and senescence, uterine stretch, and hormones produced by maturing fetal organs. The answer to effectively treating excessive inflammation may lie in a group of drugs known as cytokine supressive anti-inflammatory drugs (CSAIDs). Cytokines are a broad group of proteins that act as signalling molecules to regulate immune responses and inflammation. Cytokines and bacterial products activate common signalling pathways inside cells that turn on inflammation-related genes and activate cellular inflammation. One of the key pathways that coordinates this response is called the NF-KB pathway – activation of two enzyme complexes (IKK2 and TAK1) is critical in the regulation of this pathway.
Professor Keelan and his colleagues have examined the potential use of CSAIDs to target pathways that lead to intraamniotic inflammation and PTB, using a variety of in vitro and ex vivo models. After screening many different compounds and performing concentration-response studies, they identified two drugs that appear to be particularly effective in blocking activation of inflammation-associated genes and which are likely to be safe and effective in pregnancy. The first is an IKK inhibitor called TPCA-1. This drug, which has been investigated in several animal models of inflammation, inhibited the expression of multiple inflammation-associated genes in human placental cells stimulated with bacteria. Another drug, (5Z)-7-Oxozeaenol (OxZ, a TAK1 inhibitor), was found to be even more effective than TPCA-1 in blocking placental inflammation. Importantly, neither drug caused placental cell death, which can be a side-effect of some anti-inflammatory drugs.
In a recently published preclinical evaluation, Professor Keelan and his team found that TPCA-1 and OxZ were both effective in reducing inflammation in human extraplacental membranes delivered following spontaneous preterm labour. Production of prostaglandins – lipidderived factors produced in response to inflammation which active the uterus and trigger contractions – was significantly inhibited by the two drugs. TPCA-1 and OxZ were equally effective in membranes delivered with or without evidence of IUI, suggesting that they could be used to block the effects of both sterile and infection-driven inflammation. ‘This is important,’ says Professor Keelan, ‘because clinically it is difficult to diagnose an IUI until after delivery, when placental histology and microbiology can be performed and the diagnosis made’.
The effects of two CSAIDs were explored in a sheep model of endotoxindriven chorioamnionitis. Both drugs (administrated via intraamniotic injection) prevented signs of inflammation in the fetal lung and fetal membranes, lowering cytokine concentrations in both the fetus and the mother and levels of prostaglandins and cytokines in amniotic fluid. Studies are now underway in rodent models to confirm that the drugs prevent PTB and improve fetal outcomes at safe doses. Additional studies are required to assess fetal toxicity at high and/or chronic doses, and determine the extent of passage across the placenta. These drugs, if they are introduced clinically, would be used in conjunction with antibiotics to ensure that an underlying infection is not allowed to flourish once the inflammation has been suppressed.
Next Steps in PTB Research
The team are also working on the efficacy and safety of a novel peptide drug called rytvela as a therapeutic strategy for protecting the fetus from IUI. Rytvela is an allosteric antagonist that partially blocks the activity of the IL-1 receptor. IL-1 is a key cytokine responsible for amplifying inflammation in the pregnant uterus and neonate. Rytvela suppresses IL-1 driven inflammation in diseases such as inflammatory bowel disease and ischemic retinopathies, and in mouse models has shown remarkable results in stopping PTB and preventing inflammation-driven damage in the fetus. The team must now determine a number of factors: whether or not rytvela can cross the placenta, how efficacious it is in humans, the dose and mode of administration that best reduce inflammation with minimal side effects, and the short and long term benefits of the drug on fetal organ development and physiological function. The team, which involves collaborators across Australia and Canada, will use a number of animal and human tissue models to answer these questions.

The potential advantages to rytvela are numerous. Pilot studies have demonstrated that the drug has greater specificity, pharmacological stability, potency and efficacy than its counterparts that are currently sold and marketed around the world. Its selectivity reduces the risk of adverse effects and its increased bioavailability means it can be administered through minimally invasive routes. It is also likely to be less costly than similar available drugs. The data from this study will inform the design of clinical trials of antenatal rytvela administration. The team hopes that this will result in another anti-inflammatory option to complement antibiotic therapy directed at fighting IUI and PTB.
While the above drugs are being investigated for the treatment of preterm labour and protection of the neonate from the harmful effects of perinatal inflammation, Professor Keelan also has plans to explore alternative therapies for chronic administration throughout the second half of pregnancy. For example, the team is currently considering several plant-based dietary supplements (nutraceuticals) which have proven anti-inflammatory effects, to see if these effects can be replicated in the human placenta at feasible doses. This approach could be a safe and patient-friendly way of suppressing inflammation during pregnancy in women at high risk. The results of the preliminary studies will be known by the end of 2017.
Meet the researcher

Professor Jeff Keelan
School of Women’s and Infants’ Health
University of Western Australia
Perth, Australia
Professor Jeff Keelan is Professor of Obstetrics and Head of Laboratories at the School of Women’s and Infants’ Health at King Edward Memorial Hospital (KEMH), University of Western Australia. He is also Director of the Women and Newborn Health Research Network at KEMH, Deputy Director of the Women and Infants Research Foundation, Scientific Director of the Western Australia Preterm Birth Prevention Initiative and Co-Chair of the WA Microbiome Consortium.
After finishing his undergraduate degree in Applied Biology in the UK, Professor Keelan went on to complete a PhD in Obstetrics and Gynaecology at the University of Auckland. Throughout his career, he has received numerous distinctions. He was an invited member of the Faculty of 1000 Biology in 2007, became a fellow of the Society of Reproductive Biology in 2011 and was named on the NHMRC External Assessor ‘Outstanding Contribution’ Honour Roll in 2014. He teaches undergraduate and postgraduate courses in the areas of human reproductive biology, pharmacology and obstetrics. His current research is centred on the pharmacological treatment of intraamniotic infection and inflammation, nanoparticle-based drug delivery in pregnancy and the intrauterine microbial and endocrine environment.
CONTACT
E: jeff.keelan@uwa.edu.au
T: (+61) 8 6458 1880
W: http://www.web.uwa.edu.au/people/Jeff.Keelan
KEY COLLABORATORS
Professor John Newnham, University of Western Australia
Dr Matthew Payne, University of Western Australia Associate
Professor Matthew Kemp, University of Western Australia
Dr Demelza Ireland U University of Western Australia WA
Dr Peter Mark, University of Western Australia
Professor Brendan Waddell, University of Western Australia
Professor David Olson, University of Alberta
Professor Sarah Robertson, Robinson Institute, University of Adelaide
FUNDING
WIRF: Women and Infants Research Foundation
NHMRC: National Medical and Health Research Council of Australia
Channel 7 Telethon Trust
REFERENCES
DJ Ireland, EA Nathan, S Li, AK Charles, LF Stinson, MW Kemp, JP Newnham, JA Keelan, Preclinical evaluation of drugs to block inflammation-driven preterm birth, Innate Immunology, 2017, 23, 20–33.
DJ Ireland, MW Kemp, Y Miura, M Saito, JP Newnham and JA Keelan, Intra-amniotic pharmacological blockade of inflammatory signalling pathways in an ovine chorioamnionitis model, Molecular Human Reproduction, 2015, 21, 479–489.
D De Silva, MD Mitchell and JA Keelan, Inhibition of choriodecidual cytokine production and inflammatory gene expression by selective I-kB kinase (IKK) inhibitors, British Journal of Pharmacology, 2010, 160, 1808–1822.
JA Keelan, S Khan, F Yosaatmadja and MD Mitchell, Prevention of inflammatory activation of human gestational membranes in an ex vivo model using a pharmacological NF-kappaB inhibitor, Journal of Immunology, 2009, 183, 5270–5278.
JP Newnham, SW White, S Meharry, HS Lee, MK Pedretti, CA Arrese, JA Keelan, MW Kemp, JE Dickinson, DA Doherty, Reducing preterm birth by a state-wide multifaceted program: an implementation study, American Journal of Obstetrics & Gynecology, 2017, 216, 434–442.
JA Keelan, MS Payne, MW Kemp, DJ Ireland, JP Newnham, A New, Potent, and Placenta-Permeable Macrolide Antibiotic, Solithromycin, for the Prevention and Treatment of Bacterial Infections in Pregnancy, Frontiers in Immunology, 2016, 1, 111.
M Nadeau-Vallée et al., Antenatal Suppression of IL-1 Protects against Inflammation-Induced Fetal Injury and Improves Neonatal and Developmental Outcomes in Mice, Journal of Immunology, 2017, 198, 2047–2062.