Professor Steven E. Wilson – Clearing the Haze: Understanding the Process of Scarring Following Corneal Injury
Any injury such as trauma, surgery or infection to the cornea in the eye may result in persistent scarring (clinically referred to as fibrosis) due to the wound healing response. Professor Steven E. Wilson at the Cole Eye Institute of the Cleveland Clinic Foundation has identified that defective epithelial basement membrane (EBM) regeneration plays a central role in the development of scar producing myofibroblast cells. Critically, Professor Wilson suggests that the pathophysiological consequences of defective EBM regeneration are also likely to have wider relevance to the fibrosis that occurs in other organs, such as the lungs, heart, kidneys, and skin.
Three Decades of Corneal Research
The cornea is the transparent outer surface of the eye and lies directly in front of the iris and pupil. It acts as a protective structure and is integral to the refraction power of the eye and the precise focusing of light on the retina. Maintaining the cornea’s integrity and health is essential if the eye is to function effectively. Unfortunately, trauma and other damage to the cornea can commonly result in fibrosis (also referred to as haze or scarring) that leads to visual impairment caused by the loss of corneal transparency.
Professor Steven E. Wilson at the Cole Eye Institute at the Cleveland Clinic Foundation has spent more than three decades at the frontier of research exploring how injury and the defective regeneration of the cornea’s basement membranes may lead to the development of scar-producing cells called myofibroblasts.
The cornea provides an ideal laboratory model for which to research the mechanisms of fibrosis both within the eye and in other organs due to its easy accessibility and simple physical structure. It comprises a number of layers, including the epithelium, Bowman’s layer, stroma, Descemet’s membrane and endothelium. The corneal epithelium is an extremely thin, multicellular tissue layer of continually regenerating cells. Beneath the epithelium, the epithelial basement membrane is a highly organised layer of perlecan, nidogen-1, nidogen-2, laminins 511, 521 and 332, and collagen type IV, that attaches the epithelium to the underlying stroma and performs many other critical roles in communications between the epithelium and stroma. The stroma is the thick, transparent, middle layer of the cornea, made up of a regular array of collagen fibres, interwoven with a sparse distribution of keratocytes (resident stromal fibroblastic cells), which secrete a highly regular and organised extracellular matrix that includes collagen type I and proteoglycans. Keratocytes also produce large amounts of intracellular crystalline proteins that increase the transparency of the cells.
Professor Wilson’s research has focused specifically on the critical role that keratocytes play in epithelial basement membrane (EBM) repair and function and, in particular, the factors that lead keratocytes and other bone marrow-derived progenitor cells (fibrocytes) to develop into fibrosis-producing myofibroblasts. He is also interested in how keratocytes interact with corneal endothelial cells (that line the posterior surface of the cornea) in the production and maintenance of Descemet’s basement membrane — to which endothelial cells adhere. It is these two critical basement membranes that Professor Wilson has identified as determining whether defective tissue regeneration occurs after injury to the cornea, leading to fibrosis.
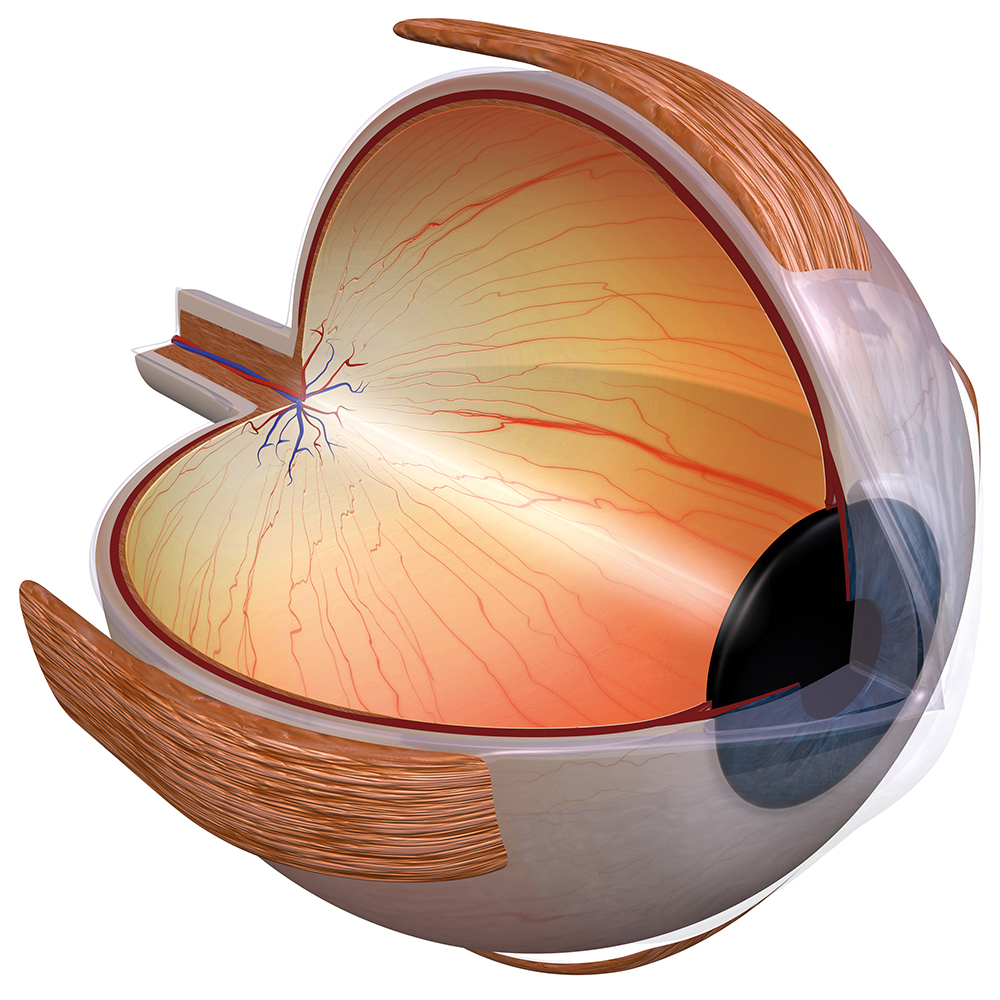
Defective Epithelial Basement Membrane Regeneration and Myofibroblast Development
In nearly all tissues and organs, fibrosis occurs through the production of myofibroblasts formed from differentiated fibroblast precursors. Critically, these fibroblastic cells are opaque in nature, and they produce a disorganised extracellular matrix within the stroma that disrupts the regular organisation and distribution of collagen fibres that are essential to maintain the transparency of the cornea critical for vision.
In their research, Professor Wilson and his team have pursued the hypothesis that following severe injury, myofibroblast development in the cornea is integrally linked to the defective regeneration of the EBM. Their research has shown that by the timely restoration of the structural and functional integrity of the EBM, fibrosis can be prevented, and corneal transparency maintained. However, delay in the regeneration of the EBM leads to the development of stromal myofibroblasts and fibrosis.
Thus, the corneal EBM plays a critical function and is sandwiched between the basal epithelium and underlying connective tissue of the stroma. Primarily made up of collagen, laminins, the heparin sulfate proteoglycan perlecan and other proteins, it forms a three-dimensional extracellular matrix that acts to maintain the cell’s structural form, but also, importantly, acts to regulate the two-way communications between epithelial and keratocyte cells, and the bone marrow-derived cells that invade the cornea after injuries.
One function that appears critical in cellular wound healing and homeostasis (maintaining a relatively stable equilibrium), is the EBM’s ability to control cellular functions by binding and mediating the local concentrations of key cytokines, such as transforming growth factor (TGF) β-1 and TGF β-2 and platelet-derived growth factor (PDGF).
In a healthy cornea, the EBM (or more specifically, components in the basement membranes, such as perlecan, nidogen-1, nidogen-2, and collagen type IV), act as barriers to bind and prevent the passage of TGFβ and PDGF from the epithelium, tears and/or aqueous humour (fluid in the anterior chamber behind the cornea) into the central cornea. However, when the EBM ‘barrier’ is punctured, through injury or trauma, large quantities of these cytokines are able to enter the stroma. After penetrating the stroma, the cytokine growth factors activate keratocytes close to the injury, transforming them into corneal fibroblasts, and ultimately into myofibroblasts if the EBM and/or Descemet’s basement membrane doesn’t regenerate. Part of the fibroblast’s normal function is to contribute key localised components to the EBM during this regenerative healing process.
When the corneal injury is minor, such as in the case of an abrasion, corneal fibroblasts in the stroma may then revert back to the keratocyte phenotype as the wound healing response subsides. However, after severe injuries, when EBM regeneration is defective, sustained exposure to TGFβ-1 and TGFβ-2 triggers corneal fibroblasts to differentiate into myofibroblasts.
Professor Wilson has identified that myofibroblasts are also formed in the cornea from bone-marrow-derived fibrocyte precursors. The myofibroblasts then produce high levels of disorganised extracellular matrix – which along with the fibroblasts themselves, create the opacity seen in the corneal stroma of scarred corneas. Regeneration of the basement membrane(s), which may take months or even years, eventually cuts off the supply of TGFβ-1 and TGFβ-2, which in turn, triggers interleukin-1 (IL-1) from adjacent cells or the myofibroblasts themselves to trigger myofibroblast apoptosis (controlled cell death). Keratocytes can then return back to the fibrotic area of the cornea where the disorganised extracellular matrix materials secreted by the myofibroblasts are removed, and transparency of the cornea is restored.
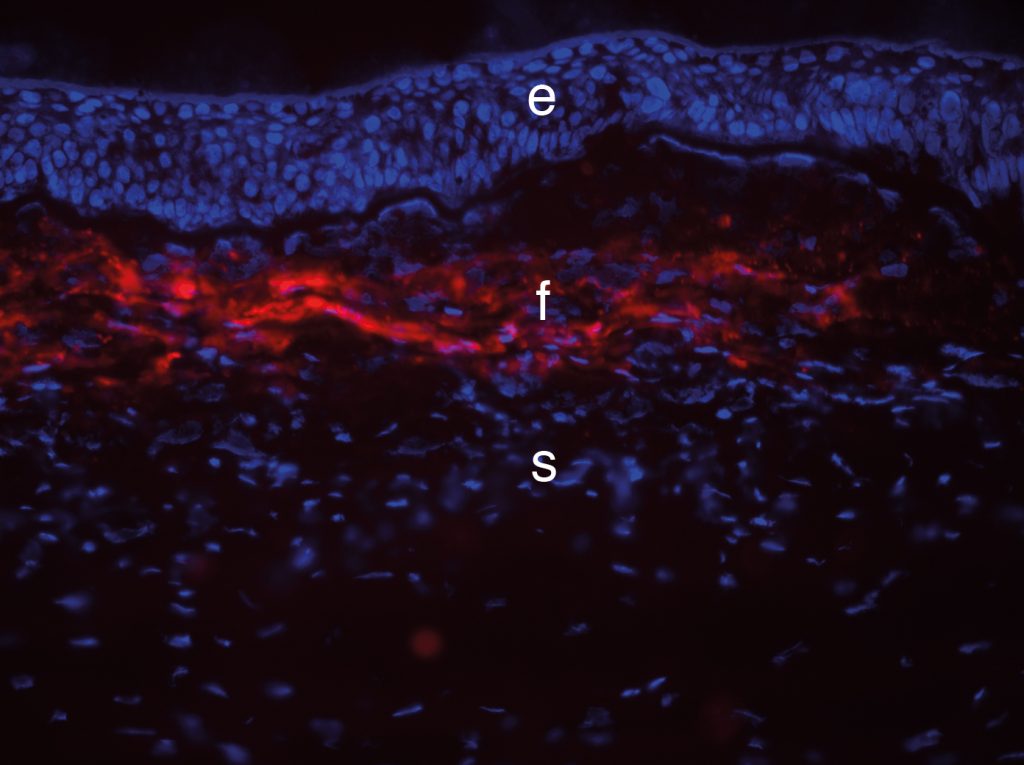
Fibrosis in the rabbit cornea.
Credit Steven Wilson.
The Key Role of Keratocytes
Now that the cause of the stromal scarring and resulting haze was apparent, the next challenge for Professor Wilson and his research team is to determine the underlying aetiology of defective EBM regeneration. He is keen to specifically explore whether a deficiency or abnormality of deposition of the EBM components, perlecan, nidogen-1, nidogen-2, laminins 511, 521 and 332, and collagen type IV, lead to defective EBM regeneration, and ultimately scarring fibrosis.
In earlier experiments, Professor Wilson and colleagues had established that the defective regeneration of the EBM probably occurred due to inadequate numbers of keratocytes in the anterior stroma. This followed from their discovery decades ago that anterior stromal keratocytes underwent significant apoptotic death after corneal epithelial injury, leading to the consequential lack of components such as perlecan and nidogen-2, that the keratocytes produce to regenerate the damaged EBM, or anomalous localisation of these components. They found that in the case of more extensive stromal injury, large numbers of mature myofibroblasts are then formed, which, as we have described, secrete a disordered extracellular matrix that forms a physical tissue barrier. Surviving keratocytes, which undergo mitosis to re-establish their numbers, are thus blocked from getting into close enough proximity to the newly forming EBM to supply and/or organise their essential components and the healing processes are delayed.
Following a minor injury to the EBM, a layer of self-polymerising laminin 511/521 is laid down by the epithelium to begin the regenerative process. However, complete regeneration of the EBM also requires adjoining keratocytes to contribute perlecan, nidogen-1, nidogen-2 and perhaps other components to rebuild the lamina lucida and lamina densa layers, which together form the mature EBM that is needed to modulate pro-fibrotic growth factors such as TGFb and PDGF.
Extensive corneal damage, therefore, results in both defective EBM regeneration and the formation and accumulation of myofibroblasts and the disordered extracellular matrix that they produce, and this can persist for many months or even years. A similar process occurs in the posterior cornea if the Descemet’s basement membrane is also injured. Eventually, in most scarred corneas, after the injury is eliminated for a long period, recovery begins with small areas of clearing called ‘lacunae’ that appear in the stromal fibrosis. In these clear areas, normal keratocytes have repopulated the anterior stroma and facilitated the regeneration of mature EBM.
Ultimately, this process facilitates the apoptosis of the underlying myofibroblasts, that will be increasingly deprived of adequate levels of TGFβ and PDGF they need to survive. Over time, the lacunae tend to coalesce and enlarge, as more surrounding EBM regenerate, eventually leading to the restoration of full corneal transparency. In the case of Descemet’s basement membrane injuries, little capacity for regeneration of this structure has been noted and corneal transplantation is typically needed to restore transparency.
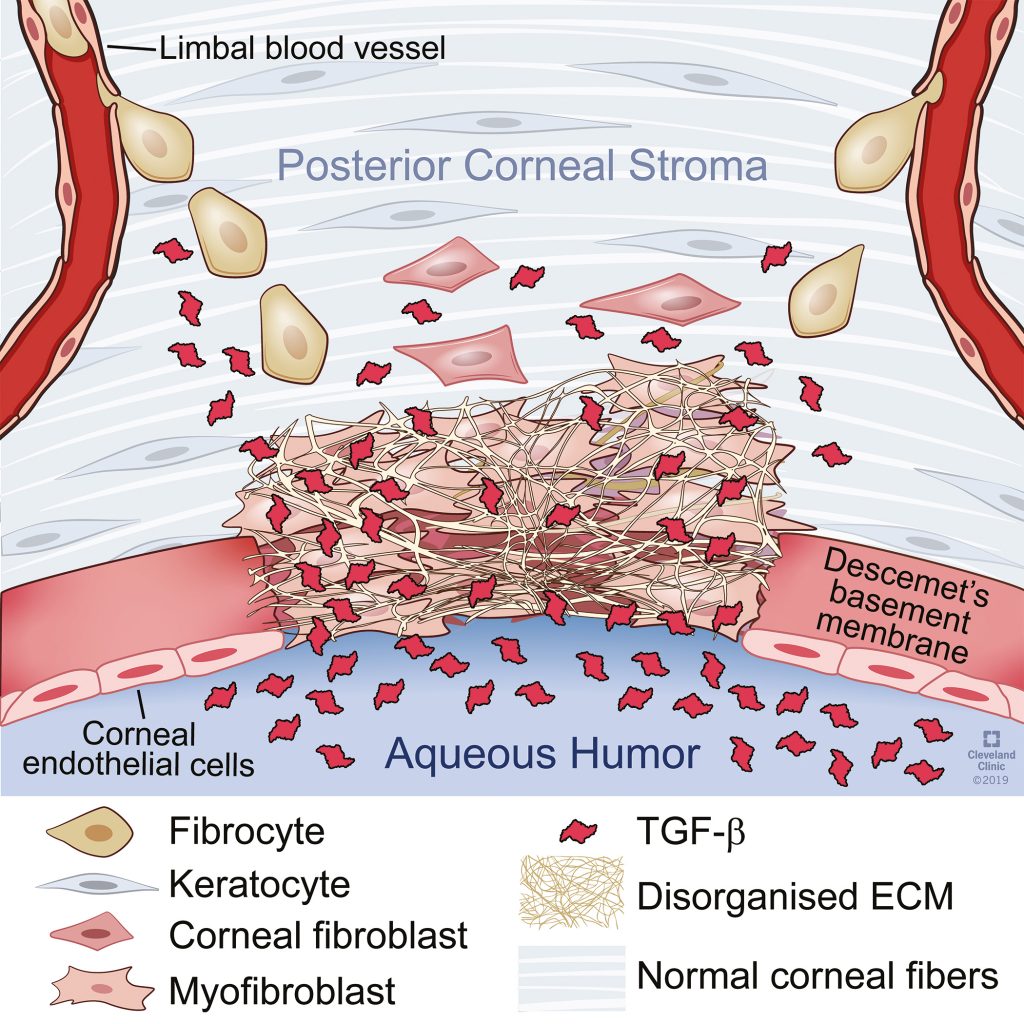
Originally published in Investigative Ophthalmology & Visual Science, Medeiros et al (2019).
Reproduced with author permission.
Keratocyte Apoptosis: A Critical Finding
Professor Wilson’s discovery that keratocyte apoptosis occurs in the anterior stroma when there is an injury to the overlying epithelium was a major breakthrough. Prior to his discovery in 1994, keratocytes were thought to be relatively dormant and inactive cells. However, Professor Wilson noted that injury led to the anterior stroma becoming decellularised and that the stroma often did not return to normal cell density as quickly as might be expected after the injury. He argued that keratocytes need to actively migrate within the anterior stroma to repopulate the area and that they need to proliferate in order to restore normal stromal keratocyte density and contribute to healthy EBM regeneration.
‘The corneal model of basement membrane injury and defective regeneration associated with mature myofibroblast development and persistence […] likely has a role in the development of fibrosis in other organs where epithelial or parenchymal cell injury is linked to disease.’
The Wider Application of the Corneal Model
The research of Professor Wilson and his team has far-reaching potential beyond having achieved a better understanding of corneal healing. The physiological changes that accompany fibrosis in many organs, such as the lungs, kidneys, and skin, are likely to have many similarities to that identified in corneal injury. Professor Wilson hypothesises that ‘The corneal model of basement membrane injury and defective regeneration associated with mature myofibroblast development and persistence, and attributable, at least in some diseases, to insufficient fibroblastic/mesenchymal contributions of EBM components, likely has a role in the development of fibrosis in other organs where epithelial or parenchymal cell injury is linked to disease.’ For example, chronic alveolar epithelial cell damage caused by toxins, such as smoke, silica dust or heavy metal dust, could lead to injury and defective regeneration of the underlying basement membrane, anomalous TGFb modulation, myofibroblast development and fibrosis, that parallels the events in corneal fibrosis.
It is, therefore, clear that a greater focus on BM injury and defective BM regeneration in other organs could provide important insights into our greater understanding of the pathophysiology of many fibrotic diseases. Importantly, when a corneal injury is interrupted, the EBM can repair itself and resolve fibrosis. It, therefore, seems probable the fibrosis can be similarly prevented or lessened in other organs, by promoting basement membrane repair.
Reference
https://doi.org/10.33548/SCIENTIA479
Meet the researcher

Steven E. Wilson, MD
Professor of Ophthalmology
Director of Corneal Research
The Cole Eye Institute/i32
The Cleveland Clinic Foundation
Cleveland, OH
USA
Professor Steven E. Wilson is the Director of Corneal Research at the Cleveland Clinic Foundation and Professor of Ophthalmology at the Cleveland Clinic School of Medicine, Cleveland, Ohio — posts he has held since 2003. In a widely acclaimed career spanning five decades as a physician and scientist, Professor Wilson is established as a world leader in corneal research. After achieving his MD at the University of California in 1984, he took up an Ophthalmology residency at the Mayo Clinic, Minnesota (1985–88) and then a Fellowship at the LSU Eye Center, New Orleans (1988–1990). He took up a number of professorships between 1990 and 2003, including Professor of Cell Biology, Neurobiology, and Anatomy at the Cleveland Clinic Foundation Health Sciences Center at Ohio State University, and Professor and Chair of the Department of Ophthalmology, University of Washington, Seattle. His most recent honours include the Richard L. Lindstrom Career Award (2013), International Society of Refractive Surgery Recognition Award (2018), and the Barraquer Award from the International Society of Refractive Surgery-American Academy of Ophthalmology (2020).
CONTACT
W: https://my.clevelandclinic.org/staff/1882-steven-wilson
KEY COLLABORATORS
Four postdoctoral fellows made outstanding contributions to this research:
Andre A. M. Torricelli, MD, Assistant Professor, University of Sao Paolo, Sao Paolo, Brazil
Gustavo Marino, MD, Sao Paulo, Brazil
Luciana Lassance Dibbin, PhD, Cleveland, USA
Carla S. Medeiros, MD, PhD, Rio de Janeiro, Brazil
Rodrigo Carlos de Oliveira, MD, Cleveland, USA
FUNDING
EY10056 from the National Eye Institute of NIH (1994 to 2021)
VR180066 from the Department of Defense, USA (2019 to 2022)
Research to Prevent Blindness, New York, NY, USA
FURTHER READING
SE Wilson, Coordinated modulation of corneal scarring by the epithelial basement membrane and Descemet’s basement membrane, Journal of Refractive Surgery, 2019, 35(8), 506–516.
SE Wilson, GK Marino, AAM Torricelli, CS Medeiros, Injury and defective regeneration of the epithelial basement membrane in corneal fibrosis: A paradigm for fibrosis in other organs?, Matrix Biology, 2017, 64, 17–26.
GK Marino, MR Santhiago, A Santhanam, AAM Torricelli, SE Wilson, Regeneration of defective epithelial basement membrane and restoration of corneal transparency after photorefractive keratectomy, Journal of Refractive Surgery, 2017, 33(5), 337–346.
CS Medeiros, P Saikia, RC de Oliveira, L Lassance, MR Santhiago, SE Wilson, Descemet’s membrane modulation of posterior corneal fibrosis, Investigative Ophthalmology & Visual Science, 2019, 60, 1010–1020.
![]()
Creative Commons Licence
(CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
More articles you may like
Grandmothers: Innovation Through Tradition
Grandmother Project – Change through Culture (GMP) is an organisation dedicated to documenting the role of grandmothers and demonstrating the effectiveness of grandmother-inclusive strategies in improving the health and well-being of women, children, and adolescents. GMP’s groundbreaking work challenges conventional wisdom to transform community-based interventions in Africa and beyond, harnessing a powerful but often overlooked resource: the wisdom and influence of grandmothers.
Dr Robert Larkin | Cultivating Change to Improve Soil Health and Increase Potato Yield
Environmental quality and food production are facing the pressing challenges of climate change and global population growth. Dr Robert Larkin from the United States Department of Agriculture-Agricultural Research Service (USDA-ARS) and a team of plant scientists developed and tested a range of crop management systems to help overcome these compounding challenges. Their work is improving soil health and increasing the yield of potato crops, contributing to the future food security of nations.
Professor Giorgio Buttazzo | Artificial Intelligence and a Crossroads for Humanity
Where do we stand with artificial intelligence? Might machines take over our jobs? Can machines become conscious? Might we be harmed by robots? What is the future of humanity? Professor Giorgio Buttazzo of Scuola Superiore Sant’Anna is an expert in artificial intelligence and neural networks. In a recent publication, he provides considered insights into some of the most pressing questions surrounding artificial intelligence and humanity.
Dr Ralf Adam | New Technologies Shaping the Future of Oral Hygiene
Understanding the efficiency of various toothbrush technologies is essential for achieving optimal oral health. Dr Ralf Adam, who leads a dedicated team at Procter & Gamble in Germany, is keen to investigate the complexities of these technologies. His team have provided new insights into the best toothbrush types for plaque removal and the maintenance of gum health. By highlighting the importance of informed oral care decisions and ongoing investigations, this vital research works towards ensuring everyone can achieve a brighter, healthier smile.




