Professor Varda Shoshan-Barmatz – Mitochondria – A Novel Target in the Fight Against Cancer
There is an urgent need for new therapies to target challenging, difficult-to-treat cancers such as glioblastoma. To meet this need, Professor Varda Shoshan-Barmatz, a renowned researcher in the field of mitochondrial biology from Ben-Gurion University of the Negev in Israel, is exploring VDAC, a key protein in the metabolic and apoptotic adaptations during cancer development, with the hope that it will present a novel therapeutic target.
The Problem with Glioblastoma
For the majority of patients, it starts with frequent headaches that, as time progresses, are accompanied by bouts of nausea, vomiting and changes in vision. Many individuals go on to experience seizures, problems with speech, and shifts in mood and personality. Investigation by magnetic resonance imaging (MRI) then reveals a lesion or mass in the brain, leading to the fear-inducing diagnosis of a malignant tumour.
Glioblastoma is the most common form of brain cancer. It is highly aggressive and particularly difficult to treat, which is reflected in its survival rate. Approximately 88% of patients die within 14–36 months of diagnosis despite undergoing surgery, chemotherapy, targeted therapy and radiotherapy. This high mortality rate results largely from the tumour’s ability to resist treatment.
Heterogeneous in nature, a glioblastoma comprises a number of different cellular population subsets, one of which consists of glioma stem cells. These are self-renewing cells that have the potential to become cancerous. Typically, glioma stem cells remain dormant until activated and, as such, they are unaffected by conventional chemotherapies that target dividing cells. Like sleeper agents, they wait patiently until the onslaught is over; only then do they reawaken to re-establish the tumour. This covert action results in great disappointment and frustration for patients and doctors alike. The bleak prognosis that accompanies glioblastoma highlights that there is an urgent need for the development of new therapies that specifically target and eliminate the tumour as a whole. Professor Varda Shoshan-Barmatz developed treatments that either kill cancer stem cells or lead to their re-programing, reversing their properties to those of non-cancerous cells.
The Hallmarks of Cancer Development
A cell undergoes changes and acquires several traits that mediate its transformation from normal to malignant. These acquired traits, often referred to as the hallmarks of cancer, enable the cell to thrive when under normal circumstances it would die. Briefly, the cell gains the ability to produce its own growth factors and reprogram its metabolism to support persistent development. In addition, it adopts strategies that enable self-replication and evasion of signals instructing it to die. This uncontrolled growth leads to the formation of a mass. Once the mass reaches a certain size (approximately 2 mm) it induces the creation of blood vessels, which provide it with the nutrients and oxygen supply required to expand and invade into surrounding tissues. Given that these adaptations are central to cancer development, it is essential to understand the mechanisms involved in order to create potential strategies for treatment. In her research, Professor Shoshan-Barmatz ‘hijacks’ two important hallmarks of cancer development and survival, turning them against cancer cells.
The Mitochondrion – The Cellular Power Plant
Metabolic reprogramming and avoidance of cell death are two of several acquired traits of cancer cells that rely on the rewiring of an organelle called the mitochondrion. Mitochondria are housed in the cytoplasm, or in association with other organelles (for example, the endoplasmic reticulum), where they communicate with other cellular compartments via multiple activities. They play a fundamental role in metabolising different substrates to produce chemical energy, in the form of the energy-carrying molecule adenosine triphosphate (ATP), which the cell requires to carry out normal processes. Mitochondria are also involved in a number of other functions, including the synthesis of many compounds, such as cholesterol, regulation of the cell redox state, calcium homeostasis, cell signalling events, inter-organelle communication, cell proliferation, cellular ageing, and disease development. As cancer cells possess reprogrammed metabolism including that of the mitochondria, Professor Shoshan-Barmatz explored a new code for reprogramming cancer cell metabolism, thus reversing tumour oncogenic properties.
In addition to serving as the cell’s powerhouse, mitochondria also play a pivotal role in regulating programmed cell death, commonly referred to as apoptosis. This is an evolutionarily conserved and genetically regulated process that begins during embryonic development, which maintains homeostasis within body tissues throughout adulthood. Apoptosis allows the efficient removal of unnecessary or menacing cells. Consequently, deficiencies in the regulation of apoptosis are linked to numerous diseases, including neuronal degenerative diseases, tumorigenesis, autoimmune disorders, and viral infections. In cancer, resistance to apoptosis contributes not only to tumour development, but also to resistance to conventional anticancer therapies, such as radiation and chemotherapy. In her research, Professor Shoshan-Barmatz discovered a new mechanism for activating apoptosis and developed novel molecules to activate this mechanism as a strategy for treating cancer.
VDAC1 – A Novel Molecular Target
The search for new treatments to tackle the cancer heavyweights such as glioblastoma has prompted the research of Professor Shoshan-Barmatz, at Ben-Gurion University of the Negev in Israel, noted for her work on the mitochondrial protein voltagedependent anion channel (VDAC) 1, as a definitive therapeutic target in cancers such as glioblastoma, liver cancer and others.
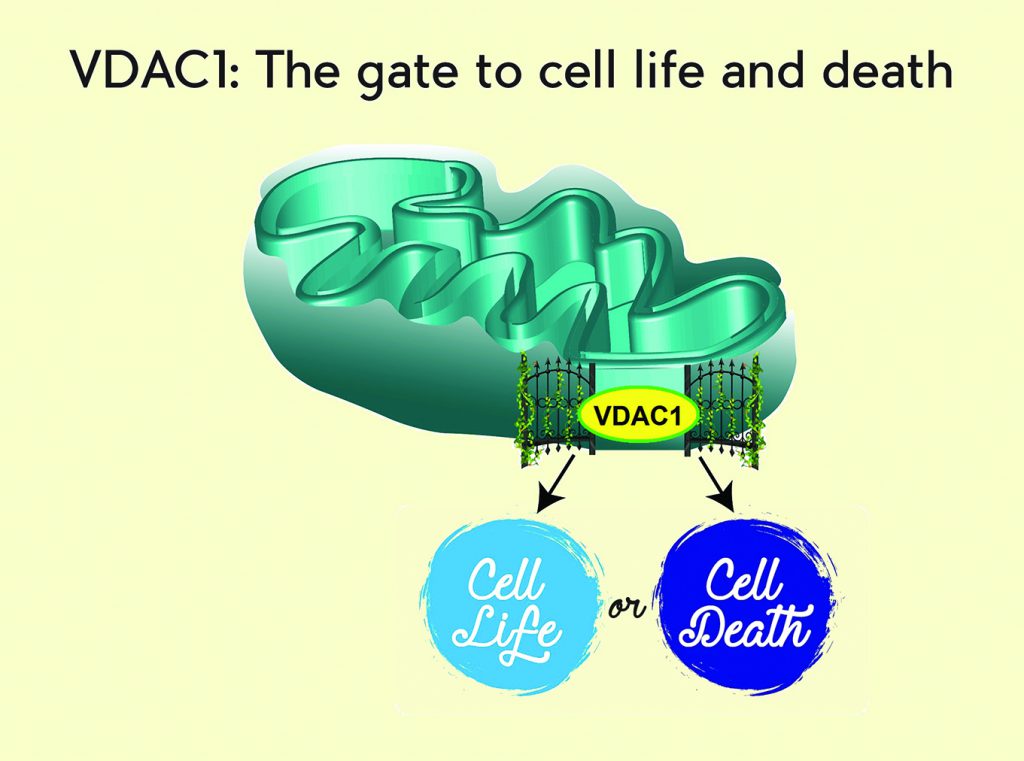
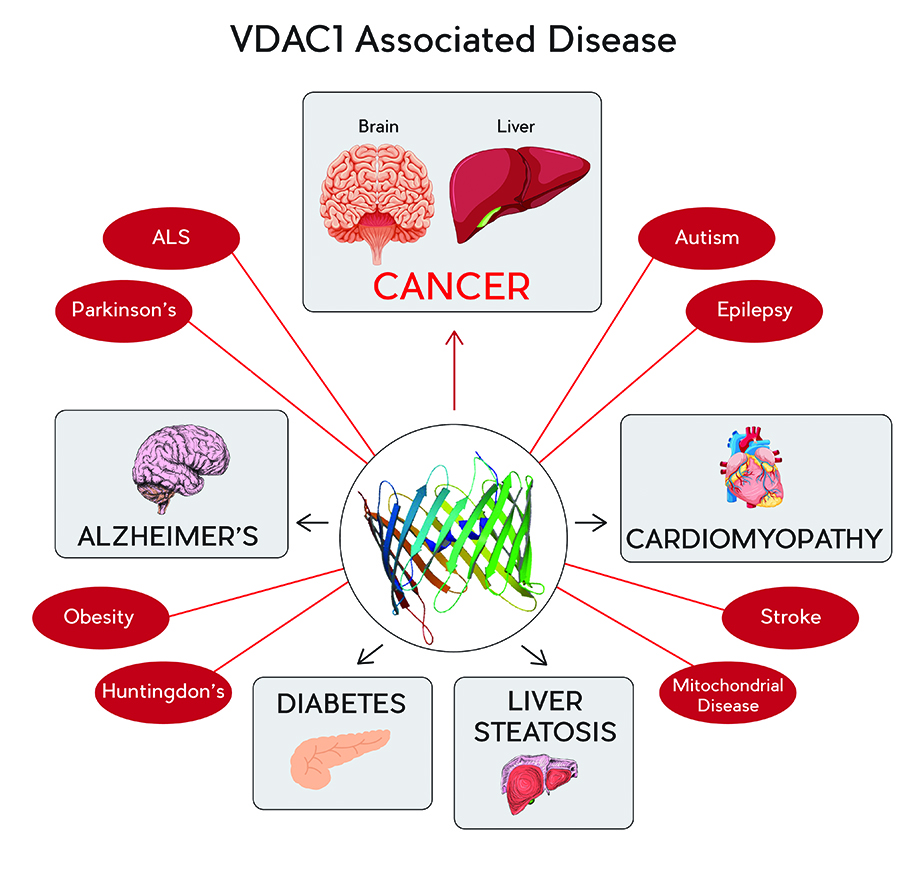
VDAC1, a protein that resides within the mitochondrial outer membrane, is a beta barrel protein containing a large pore that enables the movement of metabolites and ions into the mitochondrion from the cytoplasm and vice versa. As the gatekeeper of the mitochondrion, VDAC1 assumes a crucial position in the cell, serving as the main interface between mitochondrial and cellular metabolisms, and controlling crosstalk between mitochondria and the rest of the cell. Consequently, VDAC1 is responsible for regulating mitochondrial energy production and maintaining metabolism. VDAC1 is over-expressed in cancer cells. Professor Shoshan-Barmatz has shown that it is required for their development and survival. VDAC1 has also been recognised as a key protein in mitochondria-mediated apoptosis by assisting in the release of inter-membranal apoptotic proteins from the mitochondrion into the cytosol and due to its association with pro- and anti-apoptotic proteins. The expression levels of VDAC1 are increased in many cancers, including breast, lung and glioblastoma. Thus, VDAC1 is emerging as a promising target for controlling apoptosis.
Importantly, VDAC1 serves as a hub protein, interacting with diverse sets of cytosolic, endoplasmic reticulum, mitochondrial and other proteins that together regulate cell survival and cellular death pathways. The most notable of these proteins belong to the family of hexokinases; important enzymes that catalyse the first step of glycolysis, the process of metabolising glucose. One of the signature characteristics of highly malignant, poorly differentiated tumours is their high rate of glycolysis, a property that is frequently dependent on the marked over-expression of VDAC1-bound hexokinase in cancer cells. Subsequently, the glucose consumption of cancer cells is greater than that of non-malignant cells, a characteristic that is the basis for the PET scan. Recently, however, there has been a shift in the understanding of the role that interaction between hexokinases and VDAC1 plays in mitochondrial function. Two hexokinases, hexokinase-1 (HK-1) and hexokinase-2 (HK-2) are overexpressed in cancers and by interacting with VDAC1 they gain direct access to the mitochondrion generated ATP, thus enhancing glucose metabolism. Interestingly, HK-1 and HK-2 interaction with VDAC1 also impedes apoptosis and it is this relationship that may be instrumental in mediating not only how cancer cells reprogram metabolism to gain the energy they require to grow, but also how they acquire the ability to evade cell death. It is suggested that the increases in VDAC1 and HK-1 and HK-2 expression levels in cancer contribute to the acquisition of two of the hallmarks of cancer: reprogrammed metabolism and evasion of apoptosis. Professor Shoshan-Barmatz identified the sites in VDAC1 serving for its interaction with HK-1 and HK-2. These sites were used as the basis for developing decoy peptides interacting with HK-1 and HK-2, preventing their interaction with VDAC1 and thus the advances cancer cells gain through these interactions.
Novel Strategies for Cancer Therapies
Further research by Professor Shoshan-Barmatz and her team has led to emergence of several potential strategies for cancer therapy. The first, involves the screening, identification and development of several novel, patent protected small molecules that can activate the proapoptotic activities of the protein. By targeting VDAC1-bound HK-1 and HK-2, and VDAC1-mediated apoptosis, these small molecules are able to bring about cell death. These molecules led to the establishment of a company, ViDAC Pharma, Ltd., that is currently carrying out a phase II clinical study on non-melanoma skin cancers, and treatments for other cancers are under development.
A second strategy involves VDAC1-based peptides, a chain of amino acids derived from VDAC1 sequence serving as the binding sites for HK-1, HK-2 and other anti-apoptotic proteins, which impair energy homeostasis and minimise the anti-apoptosis self-defence mechanisms of cancer cells. To date, over 40 versions of cell penetrating VDAC1-based peptides have been designed and screened, and the three shortest, most stable, and effective at inducing cell death in cancer cell lines but not in non-cancerous cells have been identified. The peptides were tested in mouse models of cancer and were found to prevent energy production in the tumour, inhibit cell proliferation and invasion, induce cell death, including of cancer stem cells from which the cancer re-develops. This multi-pronged attack on tumours was obtained regardless of the cancer type and mutation status, with perceived specificity towards only cancerous cells.
Investigations with these peptides have been conducted in animal models of lung, breast, and liver tumours, all showing equal success in inhibiting both tumour growth and the metastasis of melanoma to the lung or the brain. Favourable results, including dramatic cell death induced by the peptides in B-cell chronic lymphocytic leukaemia (CLL), and selective killing of lymphocytes obtained from CLL patients, have been obtained, while sparing those obtained from healthy donors. This work was prompted by a prestigious three-year award from the American Leukemia & Lymphoma Society.
Professor Shoshan-Barmatz and her team have more recently demonstrated the ability of the engineered VDAC1-based peptide, Tf-D-LP4, to effectively induce cancer cell death in a panel of genetically characterised glioblastoma and glioblastoma-derived stem cell lines and in animal models of human glioblastoma. Ultimately, the Tf-D-LP4 peptide was able to cross the blood-brain barrier and launch a multipronged attack, disrupting glioblastoma cell metabolism, dramatically reducing intra-cranial tumour growth, invasiveness, proliferation and, importantly, increasing mouse survival by over 5 months (equivalent to several years in humans) even after treatment had ended. Thus, the TfD-LP4 peptide offers an innovative therapeutic strategy that is capable of preventing the replication of not only cancer cells, but also glioma stem cells in the tumour, thereby preventing tumour regrowth and the relapse of the patient.

A third strategy is based on the overexpression of VDAC1 in cancers and involves the silencing of VDAC1 expression using molecules called small interfering ribonucleic acids (siRNAs). Rather than using molecules to interfere with cellular metabolism and activate cell death pathways at the protein level, Professor Shoshan-Barmatz’s team have shifted their attention to disabling it at the source. Deoxyribonucleic acid (DNA) is the genetic blueprint from which all proteins are constructed. In a series of well-orchestrated steps, genes are transcribed, or copied, into messenger RNA (mRNA), which is subsequently translated into proteins. Therefore, if this process is interrupted at any point, the protein will not be produced and all cellular functions associated with that protein would be affected. Specific siRNAs (modified for stability) have been developed that can interrupt the translation of the VDAC1 protein from its mRNA template. Research has shown that this results in the depletion of VDAC1 within the cell, thereby inhibiting growth, disrupting energy production, and disabling the abnormal metabolic behaviour of these cancer cells. Research using a cervical cancer, lung cancer, and a glioblastoma mouse human xenograft model, showed that siRNAVDAC1 inhibited tumour growth and caused regression of established tumours.
Using glioblastoma as a platform for ‘proof of concept’, the potentiality of siRNAs was tested in several glioblastoma cell lines and patient-derived cells, and subcutaneous or intracranial-orthotopic glioblastoma xenograft mouse models. Silencing of VDAC1 expression resulted in significant inhibition of tumour development. When encapsulated within a nanoparticle, siRNA was able to reach the brain, where it reversed cancer-induced metabolic reprograming, preventing the proliferation of cancer cells, and transformed glioma stem cells, the perpetrators of disease recurrence, into normal, non-proliferating neuronal-like brain cells.
Implications of VDAC1-Based Therapeutics
Professor Shoshan-Barmatz’s work has demonstrated that VDAC1 plays a fundamental role in mediating how cancer cells acquire the metabolic and apoptotic adaptations necessary for development and progression. These findings represent a major breakthrough in the development of anti-cancer strategies that are capable of simultaneously targeting numerous hallmarks of cancer development. The results implicate VDAC1 as a significant control point in oncology and thus, an emerging cancer drug target with enormous potential. Professor Shoshan-Barmatz’s new anti-cancer agents have broad therapeutic impact and are expected to result in huge clinical benefits, not only for glioblastoma, but for other cancers as well.
Meet the researcher

Professor Varda Shoshan-Barmatz
Department of Life Sciences
Ben-Gurion University
Beer-Sheva, Israel
Varda Shoshan-Barmatz is a full Professor in the Department of Life Sciences, Ben-Gurion University (BGU), and the Hyman-Kreitman Chair in Bioenergetics. She received her PhD in Biochemistry from the Weizmann Institute of Science in Israel. She went on to carry out post-doctoral research at the University of Wisconsin-Madison in the USA and Toronto University in Canada. Since 1982, Professor ShoshanBarmatz has been a faculty member of the Department of Life Sciences at Ben-Gurion University in Israel, where she was promoted to full Professor in 1995 and served as Chair (2000–2004). Professor ShoshanBarmatz was founder of the National Institute for Biotechnology in the Negev (NIBN), served as Deputy Director (2004–2006) and Director (2006–2015). She is the Principal Investigator on 40 research grants and has authored over 140 articles and reviews. Her work on VDAC1 formed the basis for the establishment of a company, ViDAC Pharma, and she holds 13 patents on novel strategies for human cancer therapies and biomarkers. Over the course of her career, Professor Shoshan-Barmatz has mentored over 60 graduate students and 14 post-doctoral fellows. In addition, she has served on a number of granting committees and has been an ad-hoc reviewer for 16 international scientific journals. Professor Shoshan-Barmatz has earned prestigious awards for her contributions to science, including the Hestrin Prize for Excellence in Biochemical Research, a Teva Research Award for Young Scientists (1993) and Teva Founders Award in recognition of pioneering scientific achievements in cancer treatment research (2016). She was selected by the Lady Globes Journal as one of the 50 most influential women in Israel (2009) and the five women who made breakthroughs in science in Israel (2016). She has been nominated twice by BGU and the city of Beer-Sheva for the Excellence in Teaching Award and Excellent Scientist Award, respectively.
CONTACT
E: vardasb@bgu.ac.il
T: (+972) 864 61336
W: http://in.bgu.ac.il/en/natural_science/LifeSciences/Pages/staff/Varda_Shoshan-Barmatz.aspx
FUNDING
Israel Science Foundation (ISF)
The Leukemia and Lymphoma Society (USA)
Chief Scientist’s Office, Ministry of Health
National Institute for Biotechnology in the Negev
The Israel Cancer Society
REFERENCES
T Arif, Y Kerlin, I Nakdimon, D Benharroch, A Paul, D Dadon-Klein and V Shoshan-Barmatz, VDAC1 is a molecular target in glioblastoma, with its depletion leading to reprogrammed metabolism and reversed oncogenic properties, Neuro-Oncology, 2017. DOI: 10.1093/neuonc/ now297
A Shteinfer-Kuzmine, T Arif, SS Tripathi, Y Krelin, A Paul and V Shoshan-Barmatz, Mitochondrial VDAC1-based peptides: attacking oncogenic properties in glioblastoma, Oncotarget, 2017. DOI: 10.18632/ oncotarget.15455
V Shoshan-Barmatz, D Ben-Hail, L Admoni, Y Krelin and SS Tripathi, The mitochondrial voltage-dependent anion channel 1 in tumor cells, Biochemica et Biophysica Acta, 2015, 1848, 2547–2575.
S Weisthal, N Keinan, D Ben-Hail, T Arif and V Shoshan-Barmatz, Ca2+mediated regulation of VDAC1 expression levels is associated with cell death induction Biochemica et Biophysica Acta, 2014, 1843, 2270–2281.
T Prezma, A Shteinfer, L Admoni, Z Raviv, I Sela, I Levi, V ShoshanBarmatz, VDAC1-based peptides: novel pro-apoptotic agents and potential therapeutics for B-cell chronic lymphocytic leukemia, Cell Death and Disease, 2013, 4, e809.
V Shoshan-Barmatz, T De Pinto, M Zweckstetter, Z Raviv, N Keinan and N Arbel, VDAC, a multi-functional mitochondrial protein regulating cell life and death, Molecular Aspects of Medicine, 2010, 31, 227–285.