Professor Warren Foster – Searching For New Biomarkers Of Endometriosis
Professor Warren Foster and his collaborators at McMaster University are interested in finding biomarkers for endometriosis, a gynaecological disease that causes pelvic pain and infertility. They hope that the discovery of a strongly predictive and reliable biomarker for endometriosis could result in a less invasive diagnosis of the disease, meaning that women could be diagnosed more easily. Such a biomarker could also potentially be used as a measurement of treatment success, whereby declining levels could indicate a successful treatment intervention.

Endometriosis – what is it and how many women does it affect?
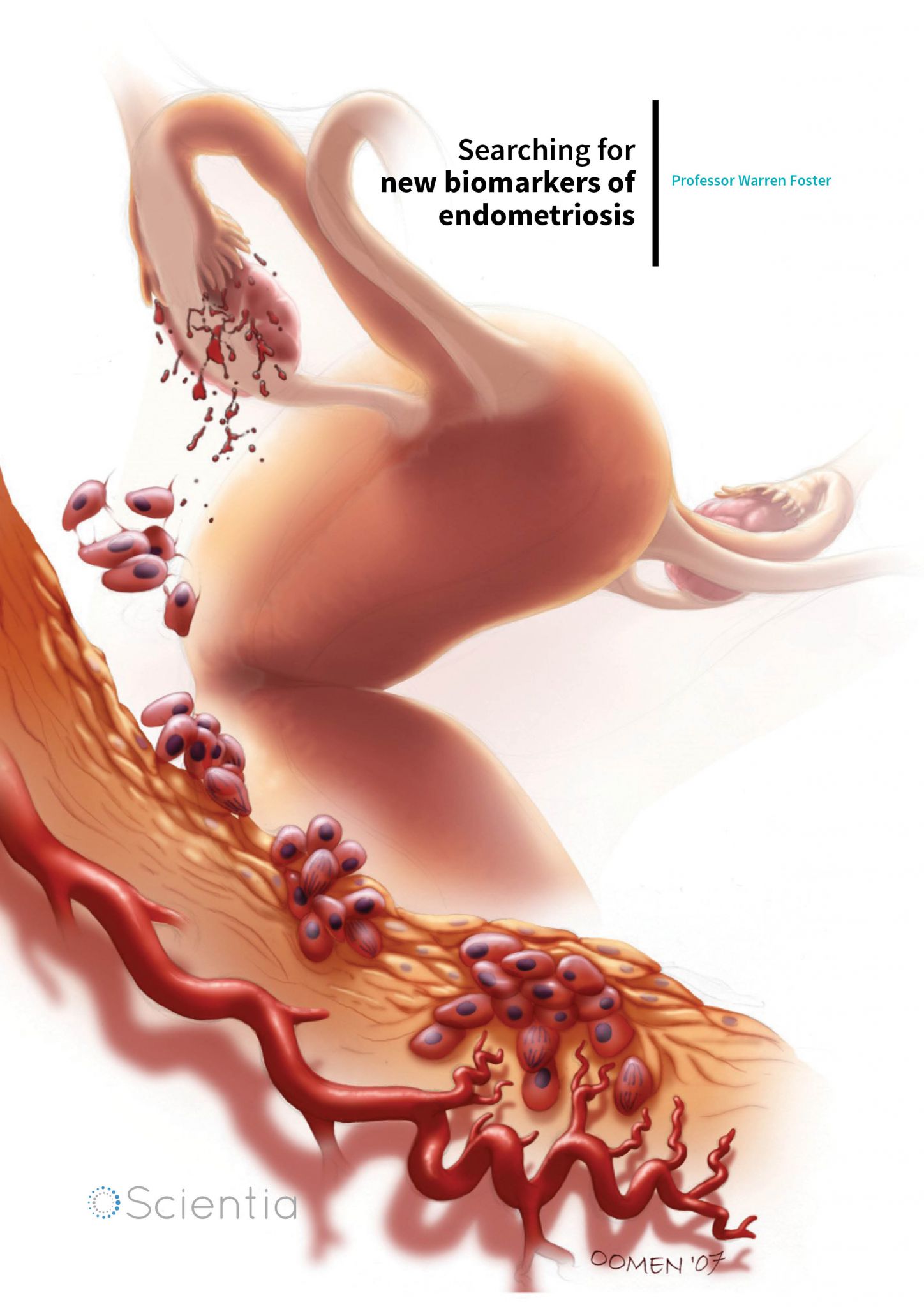
Endometriosis is a gynaecological disease, where specialised tissue, that under normal circumstances lines the interior of the uterus, grows in other locations outside the uterus. This tissue, called the endometrium, is a specialised layer that lines the interior of the uterus. Under normal circumstances, the function of the endometrium is to provide a surface for the implantation of a fertilised egg. Under the influence of female sex hormones, it grows and thickens to form a blood and nutrient-rich lining during the menstrual cycle, and functions as a permissive and nurturing environment for early embryonic development, should conception occur. The outermost layers of this tissue are routinely shed during the menstrual cycle, provided conception has not occurred. In endometriosis, fragments of this tissue arise outside of the uterus, in the abdominal cavity. This is called ectopic endometriotic growth (ectopic means in an abnormal position). This can result in pelvic pain, pain during sex and infertility. Depending on the location of the lesions, the disease can be classified as stages 1–4, which correspond to minimal, mild, moderate, and severe disease. The precise cause of endometriosis is unknown, but the disease is relatively prevalent. Approximately 10% of women of a reproductive age are affected by the disease, which applies across all ethnicities. In women with chronic pelvic pain, the prevalence increases to 30% and in infertile women up to 50% are affected. This translates to 176 million women worldwide who suffer from endometriosis and 5.5 million in the United States alone.
Societal impacts and current treatments
The societal impacts of this disease are numerous and significant. Expensive surgical procedures, drug treatments and lost time from work all contribute to a substantial economic cost. In the United States alone, the annual cost of treating endometriosis is estimated to be a staggering 22 billion dollars, which surpasses expenditure on other chronic conditions such as asthma or Crohn’s disease. The cause of endometriosis is unknown and there is no known cure. Current treatments aim to manage the disease and provide relief from its symptoms. Surgical interventions to remove the endometriotic lesions from the abdominal cavity are often implemented and can provide a measure of relief from pelvic pain. A frequent aim of treatment is to preserve or enhance fertility, where possible. In some cases, a hysterectomy (complete removal of the uterus) is necessary, or feasible in women who do not wish to have children. Treatment with pain relieving drugs is also often required, but the long term use of such drugs can be associated with significant side effects. Hormonal treatments can be used to counteract or block the actions of estrogens, which helps to reduce the growth of the endometrium during the menstrual cycle. This can help to limit the negative effects of ectopic endometriotic growths on patient well being, but can also cause significant side-effects and can interfere with fertility and the chances of conception. Additionally, it is currently difficult to determine when the disease will ‘flare up’, causing pain and discomfort, and so hormonal therapies are often required to be used all the time, meaning that affected patients need to endure their side effects almost constantly and may not be able to conceive.

‘Our work is the combined effort from a group of very talented and dedicated investigators. Their experimental results have allowed us to build a really strong research program in endometriosis that has enabled us to make important contributions in defining suitable clinical markers of endometriosis.’
Current difficulties diagnosing endometriosis
One of the major challenges in the treatment and management of endometriosis lies with the difficulty in identifying it in a timely manner. The symptoms associated with endometriosis, such as pelvic pain can sometimes be confused with other conditions, which cause pain in this area, such as irritable bowel disease or pelvic inflammatory disease, and the use of oral contraceptive pills can also suppress some symptoms, confusing a potential diagnosis. In fact, the delay between the first onset of symptoms and an accurate diagnosis of endometriosis is estimated to be 8–10 years. This means that affected women have to endure the symptoms of endometriosis without appropriate treatment for this extended period. Additionally, the harmful inflammatory processes inherent to endometriosis proceed unchecked during this period, which can lead to neuropathic pain syndromes, which can be devastating for women who have exhausted present supports trying to get help. A major factor that contributes to the delayed diagnosis of endometriosis lies with the invasiveness and cost associated with the current gold standard method of diagnosis. This approach entails a laparoscopy, where a small incision is made in the abdominal wall and a fibre-optic camera is inserted to view the abdominal cavity and identify endometriotic lesions that are present. Not only is this procedure expensive, invasive and requires specially trained medical personnel, but it can sometimes be difficult to provide a clear diagnosis. Endometriotic lesions do not always look the same and patient-topatient variability can make a conclusive diagnosis difficult. Additionally, this method is only of use in identifying larger lesions and microscopic disease may not be identified. Consequently, significant efforts have been devoted to identifying reliable biomarkers for endometriosis.
Biomarkers for endometriosis
Identifying biomarkers for endometriosis would greatly improve the ability of physicians to reliably diagnose the disease in a minimally invasive way, perhaps through a simple blood test, leading to fast track diagnosis and appropriate management. This is the motivation underlying the work undertaken by Professor Foster and his team. A variety of factors have been identified by different researchers as differentially expressed in patients with endometriosis compared with healthy controls, which is perhaps unsurprising given the inflammatory response present in such patients. However, to date, none of these factors has proven to be strongly predictive or sensitive enough to function as a robust and reliable biomarker.
Professor Foster and his team recently undertook a clinical study in which they examined the levels of four factors called neurotrophins in the blood of women with endometriosis, compared with the levels in the blood of healthy control patients. Of the four factors examined, one, called bone derived neurotrophic factor (BDNF), proved to be highly expressed in endometriotic patients, compared with healthy controls. In addition, levels of this factor were a sufficiently sensitive and specific metric to predict whether the endometriosis was stage 1 or 2. Most excitingly, levels of BDNF in patients who were receiving certain types of therapy, including oral contraceptives, or 3 months after surgical removal of lesions, were similar to levels in the control group, suggesting that BDNF is a treatment-sensitive biomarker for endometriosis. Previously, the team showed that BDNF is expressed in the endometrium and a recent proteomics study showed that it is more highly expressed in the endometrium of women with endometriosis. While the exact role of BDNF in the pathophysiology of endometriosis is incompletely understood it appears to be a promising biomarker candidate for diagnostic purposes.
 Future work and acknowledgements
Future work and acknowledgements
The team wish to further examine the relationship between levels of BDNF in the blood and disease stage and severity and will investigate potential correlations between BDNF levels and levels of pain, quality of life scores, and lesion appearance.
Previous work from the team has compared levels of BDNF in the blood of patients with surgically confirmed endometriosis with levels in healthy controls. However, to fully understand the diagnostic potential of BDNF, the team aim to carry out a comparison to determine if levels of BDNF can be used to distinguish between patients with surgically confirmed endometriosis and patients with chronic pelvic pain but no symptoms of endometriosis at surgery. If BDNF levels could be used to measure such a distinction, then patients with chronic pelvic pain but no endometriosis might not have to undergo an invasive laparoscopic procedure and similarly, may not have to undergo ineffectual treatment with endometriosis medications, saving money and preventing patient morbidity.
The team plan to examine other potential markers of endometriosis (CA-125, VEGF, and IL6) to see if the sensitivity and specificity of a panel of markers is superior to the predictive value of a single biomarker, such as BDNF alone. Finally, the team have proposed a novel way to treat endometriosis, partially based around their findings regarding BDNF. Patients with endometriosis are often faced with a dilemma, whereby they have to choose between hormonal therapies that suppress their symptoms and forgoing such treatments in order to attempt to get pregnant. For patients in the latter category, the team hypothesise that treatment with melatonin represents a viable alternative that could help with the symptoms of endometriosis while facilitating pregnancy. In a small clinical trial melatonin was shown to reduce BDNF levels and improve pain scores in patients with endometriosis. The team will examine the effects of melatonin on endometriosis and fertility in a mouse model of endometriosis to determine if this approach is viable for patients attempting to get pregnant. Professor Foster sees biomarkers for endometriosis and smartphone technology working in tandem in the future to help some endometriosis patients to monitor their disease severity and modulate their therapy appropriately. “Patients could use an app on their phone that alerts them to an impending flare up and the need to start medicating to prevent symptoms. This would be a relatively novel way of managing endometriosis and may be suitable for many (but not all) sufferers.”
Professor Foster is keen to acknowledge that this work has been a team effort. ‘Our work is the combined effort from a group of very talented and dedicated investigators,’ he tells Scientia, ‘others involved in this work are Dr Nick Leyland and Dr Sanjay Agarwal, my student Dr Jocelyn Wessels along with the tireless contributions of Annette Bullen. Other students involved in seeing this project through are Maria Haikalis, Allegra Drum and Eli Crapper, each of whom has carried out assays that have allowed us to show that brain-derived neurotrophic factor is potentially a very suitable candidate marker for the diagnosis of endometriosis. Their experimental results have allowed us to build a really strong research program in endometriosis that has enabled us to make important contributions in defining suitable clinical markers of endometriosis. This is very much a team effort that could not have had a modicum of its current success without the efforts of everyone mentioned above.’
Meet the researcher

Professor Warren G. Foster
Department of Obstetrics and Gynecology
Division of Reproductive Biology
McMaster University, Canada
Professor Warren Foster is a Professor in the Reproductive Biology Division in the Department of Obstetrics and Gynaecology at McMaster University. Professor Foster has expertise in toxicology and reproductive biology. His research program is focused on the molecular and biochemical mechanisms underlying the female reproductive system and related disease processes. In particular, he is interested in endometriosis and the key strands of his research in this regard involve discovering novel biomarkers for endometriosis and developing novel therapeutic strategies for endometriosis. He has received funding support for his research activities from the Canadian Institutes of Health Research. Professor Foster is a recipient of a Canadian Institutes of Health Research/Ontario Women’s Health Council mid-career award, an Ontario Women’s Health Council career award and several student/ postdoctoral fellow supervision awards.
CONTACT
T: (+1) 905 525 9140
W: http://obgyn.mcmaster.ca/faculty_member_foster/
W: http://endometriosis.mcmaster.ca/
KEY COLLABORATORS
Dr Nick Leyland, McMaster University
Dr Sanjay Agarwal, University of California
Dr Jocelyn Wessels, McMaster University
Annette Bullen, McMaster University
Dr Leyla Soleymani, McMaster University
Marina Bockaj, McMaster University
Maria Haikalis, McMaster University
Allegra Drum, McMaster University
Eli Crapper, McMaster University
FUNDING
Research funding for this project has been provided by an operating grant from the Canadian Institutes of Health Research (MOP 142230).
Dr. Wessels received support from a Vanier Scholarship from the Canadian Institutes of Health Research and Maria Haikalis is supported by an Ontario Graduate Student Scholarship
REFERENCES
Wessels JM, Kay VR, Leyland NA, Agarwal SK and Foster WG, Assessing brain-derived neurotrophic factor as a novel clinical marker of endometriosis, Fertil. Steril., 2016, 105, 119–128.
Agarwal SK, Daniels A, Drosman SR, Udoff L, Foster WG, Pike M, Spicer DV and Daniels JR, Treatment of endometriosis with the GnRHa deslorelin and add-back estradiol and supplementary testosterone, BioMed. Res. Intl., 2015, 934164.
Agarwal SK and Foster WG, Reduction in endometrioma size with three months aromatase inhibition and progestin add-back, Biomed. Res. Intl. (Obstet. & Gynecol.) Special Issue: The Impact of Endometriosis on the Health of Women, 2015, 878517.
Hughes CL, Foster WG and Agarwal SK, The impact of endometriosis across the lifespan of women: Foreseeable research and therapeutic prospects, Biomed. Res. Intl. (Obstet. & Gynecol.) Special Issue: The Impact of Endometriosis on the Health of Women, 2015, 158490.
Wessels JM, Leyland NA, Agarwal SK and Foster WG, Estrogen induced changes in uterine brain-derived neurotrophic factor and its receptors, Hum. Reprod., 2015, 30, 925–936.