Professor Yubin Zhou – Let There Be Light!
Professor Yubin Zhou, from the Center for Translational Cancer Research at the Texas A&M University Institute of Biosciences & Technology, USA, is developing ways to use light to control cellular function. The researcher and his team are responsible for a series of breakthroughs in this field – known as optogenetics – including the exciting concept of using this method to develop novel ways to treat cancer.
The ability to control what cells do is vital to understand cellular mechanisms. It may not be the most obvious option, but control with light has become a popular approach in a field known as optogenetics (Figure 1). Early work involved neurons genetically modified to express channels in the cellular membrane that were sensitive to light. This represented a simple level of control with light on to open the channel and light off to close it.
In the past few years, there’s been a boom in this field and many researchers are now starting to consider whether this can be applied in other areas. One of them is Professor Yubin Zhou, from the Center for Translational Cancer Research at the Texas A&M Institute of Biosciences & Technology. ‘My group is working on developing innovative tools by combining optogenetics and cell signalling,’ explains Professor Zhou. ‘These light-sensitive tools allow us to control the flow or localisation of signalling molecules, proteins, and enzymes, so that we can manipulate biological pathways and gene expression in a non-invasive and reversible manner.’
The idea is sound, but with immune cells instead of nerve cells as their main target, the team had a major challenge to overcome first. Stationary nerve cells are easy to reach, but what about fast-moving immune cells? How to develop a way to shoot accurately at a moving target?
Calcium Signalling
Even before the optogenetics idea, one of the early interests for Professor Zhou’s laboratory has been a deeper understanding of cell signalling with a focus on calcium signals. This ion may be present only in small amounts in the cell, but it is crucial in controlling a variety of processes, from muscle contraction and communication between neurons, to gene expression and even cell death. Not surprisingly, abnormal activity has been linked to many different conditions, including cancer and cardiovascular problems.
As the signal relies on sharp and effective releases of calcium from its cellular stores (the endoplasmic reticulum being the most important) or sudden calcium influx from the extracellular space, it is crucial to maintain the correct calcium levels. To achieve this, cells resort to specialised sections linking the cellular membrane and the endoplasmic reticulum. Curiously, these structures don’t even touch, but recently researchers are beginning to understand their importance in regulating calcium signals.
‘With these tools, we can now not only answer fundamental questions of science that we never could before but also translate them into the clinic for disease intervention’
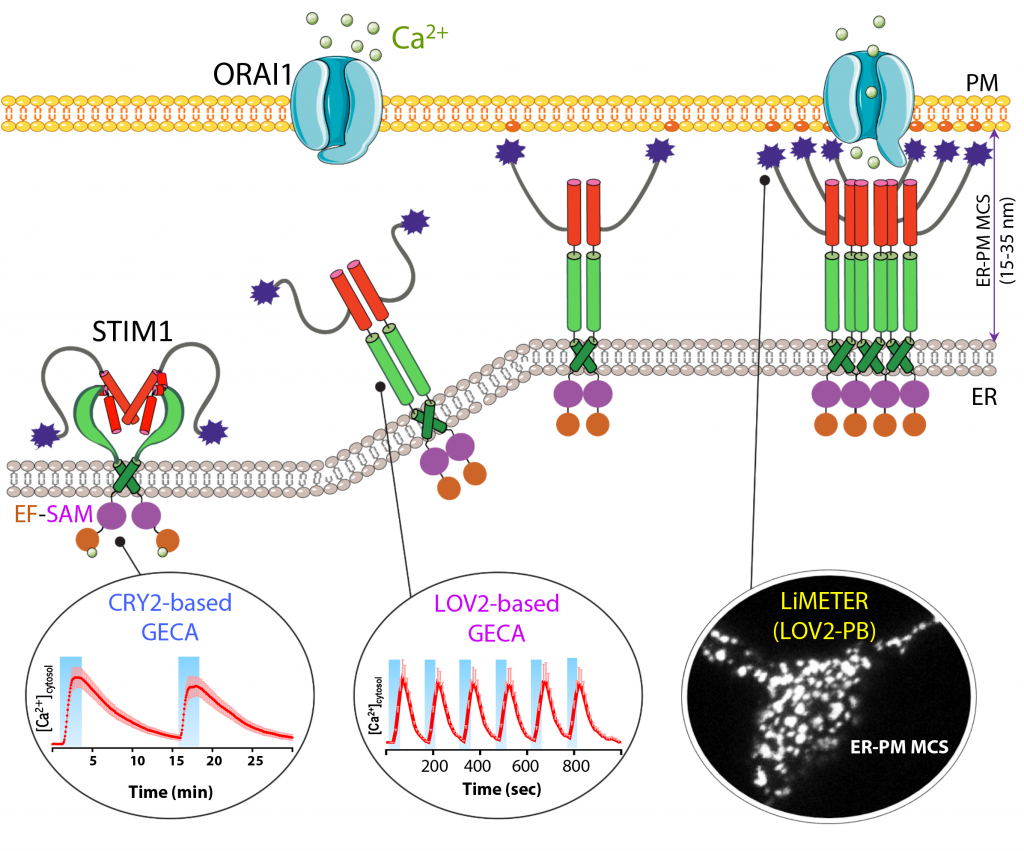
Figure 1. Optical control of calcium signalling at membrane contact sites. Credit Yubin Zhou
Understanding these signals has been on Professor Zhou’s agenda since 2015. The first step included the identification of all the proteins involved in transporting calcium, which when put all together form a multi-protein structure spreading across the membrane (Figure 2). Further studies unravelled conformational changes in this complex to regulate calcium influx. During rest, everything stays closed and inactive. When there’s a need for a burst of calcium, however, there’s a quick change with two arms extending to open the gates.
Surprisingly, Professor Zhou also found different sensitivities to calcium in these structures. Some can be fast but need large calcium fluctuations to work, whereas others have a weaker binding affinity to calcium and are more responsive to small fluctuations. Professor Zhou and his team believe this combination makes for efficient regulation of calcium levels in the cell.
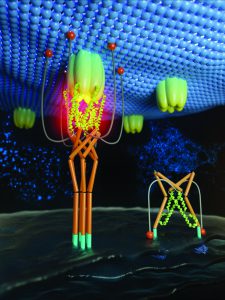
Figure 2. Transporting Calcium. Credit Yubin Zhou.
The Power of Light
It turns out this early work studying calcium channels was essential in solving Professor Zhou’s big challenge: that is, to develop a new system to remotely control immune cells using light (Figure 3). Eventually, the team managed to design a way to mimic the conformational changes in calcium signalling described earlier. In essence, Professor Zhou’s team created a form to trigger calcium release at will in immune cells following light stimulation.
As a test, the team genetically engineered immune cells with a specific protein involved in calcium signalling becoming light sensitive (designated ‘Opto-CRAC’). Through collaboration with a nanotechnologist, Dr Gang Han, the team then used a laser to penetrate deep (in this case, deep means about a centimetre or two) into the mouse’s thigh, where nanoparticles converted the laser light into blue light. Exposing immune cells to blue light from the nanoparticle opened the calcium gates to release calcium, whereas darkness stopped the flow. The system even has a built-in fine-tuning mechanism, where more light increases calcium release and less light means a gentle trickle.
It’s not difficult to imagine that these light-based techniques could allow ‘remote control’ of immune responses within the body, in particular in combination with immunotherapy to fight cancer at a specific time and location. ‘My group is among the first to advocate the concept of optogenetic immunomodulation to advance next-generation anti-cancer immunotherapies with reduced off-target cytotoxicity,’ notes Professor Zhou.
The fact that it can be switched on and off easily means this method could represent a much more accurate approach to treat cancer, with a low incidence of the type of side-effects common in other therapies. ‘It’s quite a cool technology. With these tools, we can now not only answer fundamental questions of science that we never could before but also translate them into the clinic for disease intervention,’ added Professor Zhou.
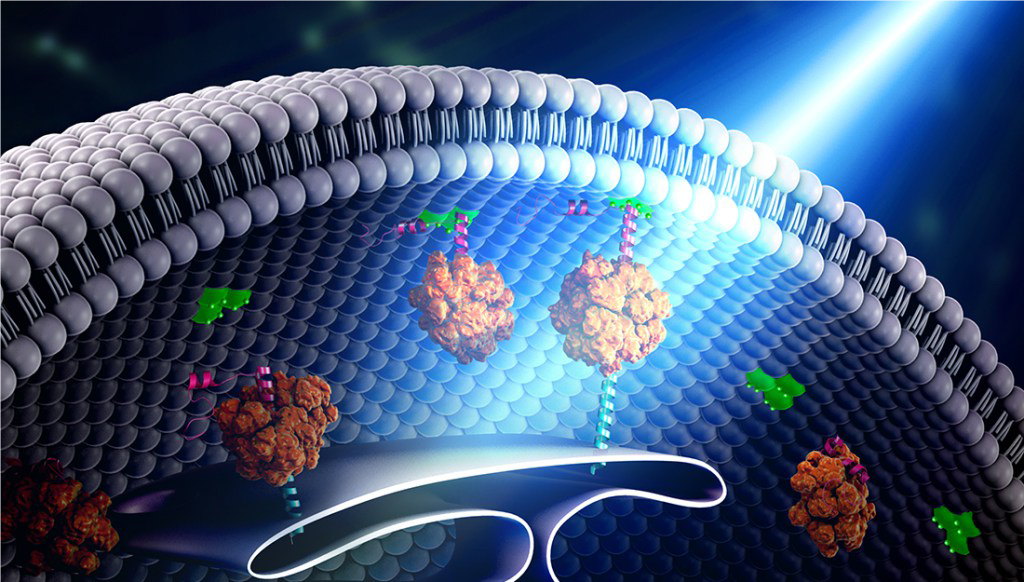
Figure 3. Engineered photosensitive proteins serve as the building blocks of the ‘bridge’ between organelles in the cell. Credit Yubin Zhou.
Cellular Communication
Professor Zhou and his collaborator Dr Yun Huang very quickly realised there was a variety of potential uses for their light-induced method. Their first approach was to couple light-sensitive tags to proteins bound to membrane lipids which are involved in forming the bridges between cellular organelles (Figure 4).
Following blue light illumination, this new system – named OptoPB or LiMETER by the researchers – forces the target to undergo conformational changes to expose the region that binds to membranes. This new method proved to be quite a success and the team even found that they could manipulate the contact region and the distance between membranes, as well as control the movement of proteins in the membrane with a simple blue light.
This work focused on the link between the cell membrane and the endoplasmic reticulum, but the team’s ambition is to build bridges between other organelles, such as between the endoplasmic reticulum and mitochondria. For Professor Zhou, this is just a glimpse of what can potentially be achieved in the future in cell biology and other fields.
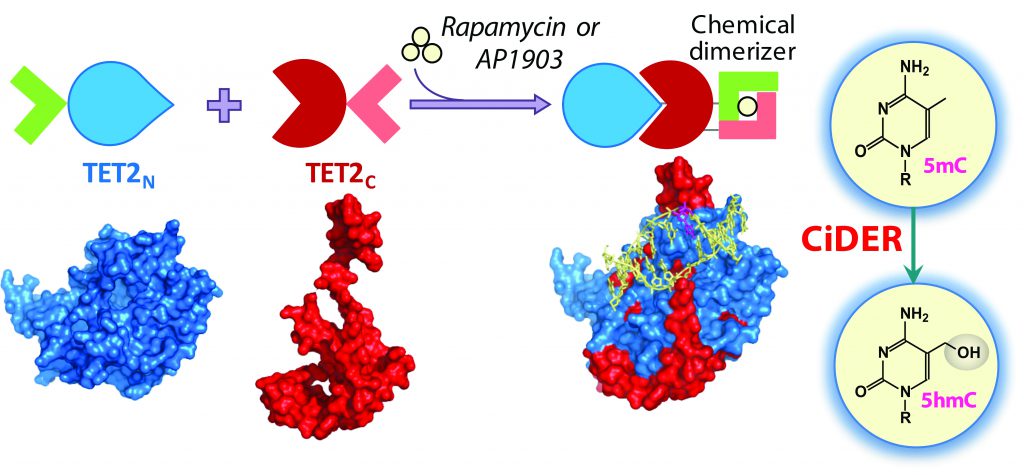
Figure 4. Design of a Chemical-Inducible Epigenome Remodeling (CiDER) Tool Based on a Split TET2 Enzyme. Credit Yubin Zhou.
Time for a Cider
The next use of light and optogenetics involved controlling epigenetic changes. To put it simply, these changes modify the activity of certain genes, but only on a superficial level and do not involve modification to the actual genetic code sequence. Researchers know these are involved in multiple conditions, including cancer, Alzheimer’s disease, and developmental disorders, but it’s still not obvious whether these changes are the cause or a consequence of such conditions.
This question has been impossible to answer because the tools to generate the desired DNA modifications are very limited. But Professor Zhou has developed a system that may provide a workable solution. The new tool allows researchers to control with precision specific epigenetic changes. Crucially, the system can be controlled with a drug and reverses without it, giving Professor Zhou and his team absolute control over epigenetic changes. They named it CiDER, which stands for chemical-inducible epigenome remodelling tool (Figure 5). By replacing the chemical-responsive modules with photosensitive domains, the human epigenome can be conveniently re-sculptured with a simple flash of light.
At the moment, the system still lacks control over where the changes occur, but the researchers want to use the genetic tool CRISPR to improve specificity. Once this issue is overcome, Professor Zhou foresees enormous potential for this tool in any field involving epigenetic changes, and not just for cancer studies.
Figure 5. Non-invasive Control of Voltage Gated Calcium Channel
to Intervene with Cardiac Arrhythmia.
After Cider Comes the Carrots
Keen to pursue the CRISPR avenue, the team’s next step was to design a method to use the same light-induced calcium signalling to carry this genome-engineering tool. The method, which the team called CaRROT (for calcium-responsive transcriptional reprogramming tool) – is simple. In the dark, nothing happens. However, when the light is switched on, the passageway controlling calcium signals opens, which in turn triggers expression of specific genes through CRISPR.
This approach allows the researchers to decide exactly how, when, and where genes are programmed to perform (or not) a desired function. The future goal is to control cellular differentiation in stem cells, which can then be used in regenerative medicine. It remains possible that the day will come when doctors can grow a new organ by simply shining a light on a petri dish.

Professor Yubin Zhou and His Team at the Texas A&M University Institute of Biosciences & Technology.
The Opposite Trick
Finally, Professor Zhou is also the mastermind behind a new optogenetic tool with the opposite objective: Instead of triggering a calcium signal, when the light shines, calcium influx can be turned off.
There are already several calcium-channel blockers commonly used to treat cardiovascular conditions including high blood pressure, arrhythmia, and coronary artery disease. However, with side-effects including headache, dangerously low blood pressure, and palpitations, a new approach to complement traditional medication is eagerly awaited.
There may be good news coming soon on this front, as tests in cardiac muscle cells have shown a significant reduction or even stopped calcium release altogether in response to the blue light, and then restarted when the light was switched off.
This approach – which the researchers termed optoRGK – promises unprecedented accuracy and, in combination with all the other light-based tools, is ideal for regulating many processes in many different cellular mechanisms (Figure 5). The team believes their work will one day drive a new generation of optogenetic devices for treating a variety of diseases, including cancer and cardiovascular conditions.
Translational Research
Chimeric antigen receptor (CAR) T-cell based immunotherapy has demonstrated curative potential for the treatment of leukemia and lymphoma. Recently, the FDA has approved two CAR T-cell therapies (Yescarta and Kymriah) to treat certain types of blood cancer. Nevertheless, due to a lack of predictable and precise control of the dose, location, and timing of T cell activity, CAR-T cell therapy has been associated with significant safety issues, as notably exemplified by ‘on-target, off-tumor’ toxicity and cytokine storm syndromes. These side-effects have had devastating consequences for certain patients in clinical trials, and pose significant limitations on the use of the current CAR-T therapy. The team aims to tackle this challenge by developing the next generation ‘optogenetic immunotherapy’ in which precise control over anti-tumor immune responses will enable personalised tuning of the amplitude, duration, and location of the treatment (Figure 6). Such intelligent cell-based therapy makes it possible to tell when and where to launch a fierce attack toward tumor cells while sparing healthy ones, thereby mitigating the potential toxicity encountered in current anti-cancer treatment.
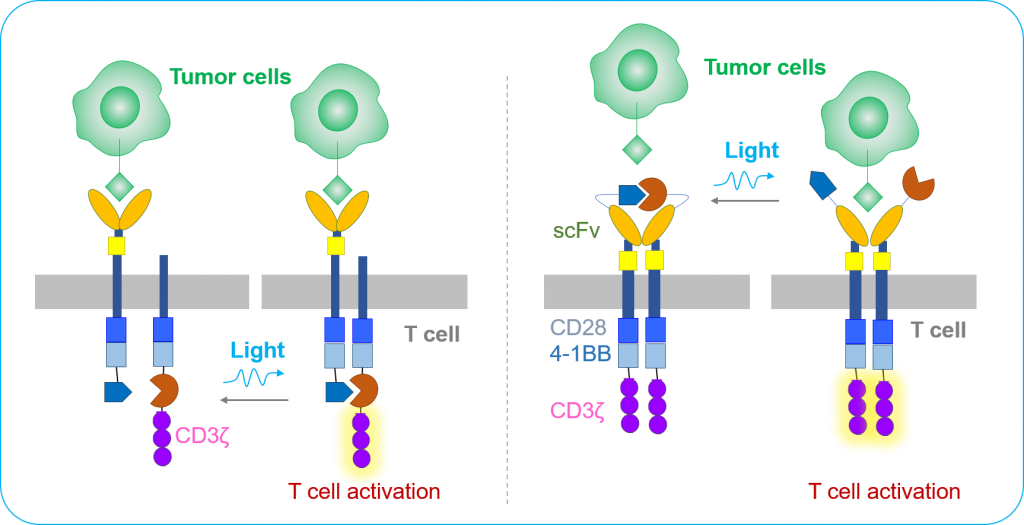
Figure 6. Design of Photo-switchable CARs for Optogenetic Immunotherapy. Credit Yubin Zhou.
A Rewarding Journey
For Professor Zhou, the optogenetic engineering of calcium channels turned out to be an extremely rewarding journey. It started with calcium signals and ended with a series of optogenetic and chemical biology tools, including OptoPB, LiMETER, Opto-CRAC, CaRROT, CiDER, and optoRGK, to harness the power of light to control calcium entry and gene expression in cells, and ultimately in living organisms.
‘We have been constantly translating what we have learned from basic studies on ion channels and signalling proteins toward the development of molecular toolkit and drug candidates. The invention of innovative optogenetic tools has enabled life scientists not only observe but also to perturb biological processes at unprecedented precision. We are more like Bob the Builder, very keen to craft new toys for biomedical researchers to play with for the purpose of technical and conceptual innovation,’ concludes Professor Zhou.
Meet the researcher

Professor Yubin Zhou
Center for Translational Cancer Research
Institute of Biosciences and Technology
Texas A&M University
Houston, TX
USA
After completing his medical training in 2003 and a PhD in chemistry at Georgia State University in 2008, followed by a 2-year postdoctoral position at Harvard Medical School, Professor Yubin Zhou joined La Jolla Institute for Immunology in 2010 and eventually moved to Texas A&M University where he currently holds the position of Associate Professor. Research on calcium signalling and optogenetics has rewarded Professor Zhou with almost 100 published manuscripts in high-impact journals and four patents. In addition, he has received multiple awards including the John S Dunn Foundation Collaborative Research Award in 2018 and the High-Impact/High-Risk Research Award, Cancer Prevention & Research Institutes of Texas in 2017. Professor Zhou also teaches at Texas A&M University and has mentored a long list of undergraduate and postgraduate students, many of whom have progressed to receive awards themselves.
CONTACT
E: yzhou@ibt.tamhsc.edu; yubinzhou@tamu.edu
T: ZhouLab_TAMU@ibtzhoulab
KEY COLLABORATORS
Yun Nancy Huang, Institute of Biosciences and Technology, Texas A&M University
Gang Han, Department of Biochemistry and Molecular Pharmacology, University of Massachusetts Medical School
FUNDING
National Institutes of Health (R01GM112003; R21GM126532; R01CA232017; R01HL134780)
Cancer Prevention and Research Institute of Texas (RP170660)
American Cancer Society (RSG-16-215-01-TBE; RSG-18-043-01-LIB)
Welch Foundation (BE-1913)
John S Dunn Foundation
FURTHER READING
G Ma, J Liu, Y Ke, et al, Optogenetic control of voltage gated calcium channels, Angewandte Chemie, 2018, 57, 7019–7022.
N Nguyen, L He, M Moczygemba, et al, Rewiring calcium signaling for precise transcriptional programming, ACS Synthetic Biology, 2018, 7, 814–21.
S Zheng, G Ma, L He L, et al, Identification of molecular determinants that govern distinct STIM2 activation dynamics, PLOS Biology, 2018, 16, e2006898.
L He, J Jing, L Zhu, et al, Optogenetic control of membrane tethering and interorganellar communication at nanoscales, Chemical Science, 2017, 8, 5275–81.
M Lee, J Li, Y Liang, et al, Engineered split-TET2 enzyme for inducible epigenetic remodeling. Journal of the American Chemical Society, 2017, 139, 4659–62.
P Tan, L He, G Han, Y Zhou, Optogenetic immunomodulation: shedding light on the immune system, Trends in Biotechnology, 2017, 35, 215–26.
J Jing J, L He, A Sun, et al, Proteomic mapping of ER-PM junctions identifies STIMATE as a novel regulator of Ca2+ influx, Nature Cell Biology. 2015,17,1339-47.
L He, Y Zhang, G Ma G, et al, Near Infrared photoactivatable control of Ca2+ signaling and optogenetic immunomodulation, eLife, 2015, pii:e10024.