Rebecca Tallmadge, Phd | Maria Julia Bevilaqua Felippe, Mv, Ms, Phd – Antibodies, Epigenetics, And A Bit Of Hay
Dr Julia Felippe and Dr Rebecca Tallmadge of Cornell University’s College of Veterinary Medicine are attempting to understand the complex world of the equine immune system. We talk to them about their discoveries and plans.

Imagine a gingerbread house, tall as a mountain, warm and cosy, full of all the proteins and nutrients which you could possibly need to grow, reproduce, and start a giant family. Sounds tempting, doesn’t it? This is you, from the point of view of the millions of microbes swarming over your skin, your hair, in your gut and in your nose. A delicious mountain of food protected only by the tireless defence offered by the bodies’ own immune system. Small wonder then that those who lose this vital protection rapidly succumb to diseases which would barely even irritate a healthy individual.
There are many ways in which we can be left without a functioning immune system. Perhaps the most famous is AIDS, in which infection with HIV leads to a progressive loss of immune capability and eventual death from minor secondary infections. Otherwise known as immunodeficiencies, these situations are also brought about by cancer, by chemotherapy, by infections with various pathogens, even just by being born.
Just by being born?
Though we don’t often think about it, an immune system develops – it expands in capabilities over time. Infections trigger expansions in antibody-producing cells which can target those pathogens, a memory of sorts which remains long after the infection is gone, protecting against the next infection (this, of course, is the basis of immunisation). The flip side of this, however, is that an immune system which has not yet had time to develop is also far weaker than it could otherwise be.
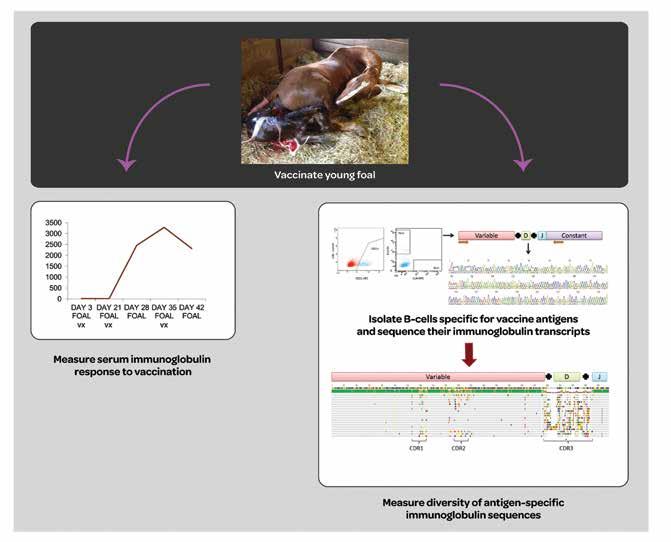
Although a large portion of the bodies’ immune system is developed while in the womb, babies tend to be born in an immunologically ‘naïve’ state – they have not yet had any exposure to the outside world, full of bacteria and other antigens. This means that the intrinsic protection provided by exposure has not yet been developed and so newborn mammals have a much higher risk of catching disease than those just a few months older. To offset this window of vulnerability, the newborn receives the antibodies produced by its mother – these antibodies continue to circulate for a while after birth and so effectively act as a borrowed immune system. Borrowed, but not as effective as it could be; newborns ranging from human babies through to horse foals are highly susceptible to pathogenic attack.
Something old, something new, something borrowed
A cavalcade of complexity is thus occurring in a newborn, mixing new-developing with decaying-borrowed immune systems, all reacting to the colonisation of bacteria which occurs moments after being born. Sounds impossible to untangle? Even more complex when your subject is up and running around within hours of being born – as is the case with those who study the immunology of horses.
Untangling this complexity is the lofty goal of Dr Julia Felippe and Dr Rebecca Tallmadge, both of Cornell University. Dr Julia Felippe is one of the top researchers in the field of equine immunology, having spent the last 27 years either as a practicing equine veterinarian, studying equine clinical medicine, or researching equine immunodeficiencies and immunodevelopment. Dr Tallmadge is newer to the field than Dr Felippe, but nonetheless brings an exceptionally broad array of knowledge and experience derived from both equine immunology and genetics. As she comments, her current role allows her to ‘provide insight from my training in equine immunogenetics – combining data we obtain in the lab with genome sequences to characterise the strength or limitations of an immune system, whether it be an immunodeficient horse or a young foal.’
Their studies focus on the role of the developing immune system in diseases such as those caused by pathogenic infections or developmental difficulties. The two have been, in Dr Tallmadge’s words, ‘gaining insight into the development and function of the immune system, then applying that knowledge to the benefit of immunodeficient horses and young foals during that period of time where they are especially susceptible to pathogens.’
These pathogens include particularly nasty examples such as R. equi, a dust-borne bacterium which, like the agent responsible for Tuberculosis, is able to avoid destruction by hiding within the lungs’ own immune cells. By hijacking these cells, known as macrophages, R. equi is able to grow without fear of detection and thus can rapidly spread throughout the lungs, causing an often severe pneumonia. The bacterium is exclusively dangerous to newborn horses and foals, as (despite their loaned set of antibodies), they have yet to develop the innate immunity which would otherwise protect them from infection – this same effect can be seen in immunocompromised human or adult horse patients, who can be susceptible to the same disease.
The trick, then, is to develop a way to target a bacterium which can hijack and kill the very immune cells which are meant to be killing it. This sounds difficult, and indeed it is – despite many different approaches the only sure-fire way to induce a robust immune response in foals is to infect them with the microbe itself. As part of the road to better understand how the pathogen deceives the immune system, the group demonstrated the ability to make a miniature lung segment – complete with the ability to clean itself of inhaled dust, and then used this to watch in detail the manner in which R. equi infected macrophages. Knowledge of these processes allows the further development of targeted treatments, and indeed Dr Felippe has collaborated with other groups assessing effective non-infectious vaccines.
‘The overall focus of our work is to gain insight into the development and function of the immune system, then applying that knowledge to the benefit of immunodeficient horses and young foals during that period of time where they are especially susceptible to pathogens.’ – Dr Tallmadge
Epidemics of epigenetic
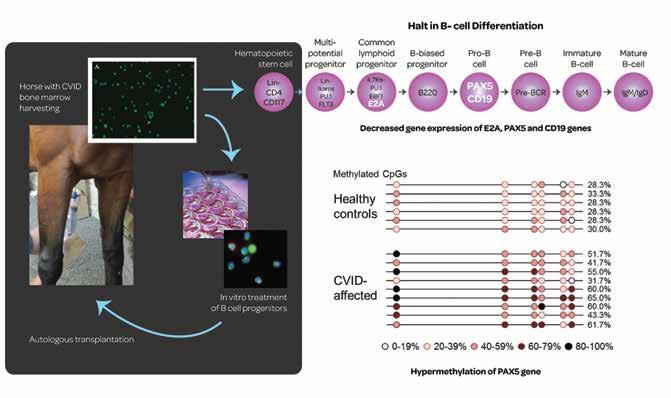
Another major focus of their work is known as common variable immunodeficiency (CVID for short). One mechanism involved is the sudden halt in the production of B cells within the bone marrow. As B-cells are the major source of antibodies within the body, this leads to a rapid drop in circulating antibodies and thus significantly weakens the ability of the affected horse to resist disease. CVID is a disease found both in horses and humans, though the origins of the symptoms often remain mysterious, as Dr Felippe explains: ‘though known in human patients for the last 6 decades, only about 10% of patients have been described with a genetic mutation, and the cause of disease is unknown for 90% of patients.’ The lack of a genetic source led her group to follow other possibilities, with the eventual discovery that, in her words ‘target gene expressions were downregulated in affected equine patients, and aberrant epigenetic mechanisms were associated with gene silencing, with the resulting halt in B cell production.’
Fine words, I hear you say, but what does this actually mean? The traditional view of genetics was that each trait held by a person or animal (strong legs, black hair, brown eyes) was the product of one or more genes, passed down from parents to children and set in stone from the moment of conception. This view turns out to be incomplete. There are a number of ways in which DNA can be modified at a later stage, changing the expression of the genes involved without modifying their basic code. The study of these secondary genetic factors is known as epigenetics, and this is the field in which Dr Felippe found herself.
Because CVID involves epigenetic mechanisms, can epigenetic factors also help to provide a cure? Bone marrow transplants are rarely used in humans with CVID to provide a fresh source of the precursor cells which can differentiate into B-cells – essentially overwriting the patients’ immune system with one from a healthy donor. Dr Felippe’s group is studying a novel approach, attempting to take the patient’s own derived bone marrow and reactivate the affected B-cell precursors using epigenetic modifiers. These cells can then be retransplanted into the affected horse, essentially allowing them to be the healthy donor for themselves.
Epigenetics are also involved in mediating the overall effect of other diseases such as equine herpesvirus-1 (EHV-1), a highly contagious virus which not only leads to neurologic damage but remains latent – ‘once infected, infected forever’ – even once apparently cured. Latent viruses such as EHV-1 tend to hide their genetic components within the bodies’ own cells, safe from immunological response but able to reactivate at a later date. By testing the use of epigenetic modifier proteins (such as histone demethylase inhibitors), the group was able to control the expression and number of these genetic copies which occurred – an excellent first step towards developing therapeutics. Indeed, Dr Felippe is already thinking along these lines, as she comments ‘our results are encouraging, and we are moving along in the path toward in vivo trials, then clinical trials.’
‘In the last few years, we have developed tools and knowledge on equine hematopoietic regenerative medicine in order to promote B cell differentiation in vitro from bone marrow precursors of CVID-affected horses.’ – Dr Felippe
Beyond a cure
Dr Tallmadge agrees with these plans, commenting that ‘while we use basic science and not each experiment can be directly applied clinically, our projects address questions with biological relevance. Further, what is learned in one project can often be applied to the other project; that synergy reinforces the value of our efforts and findings, and further drives my motivation.’ Both enjoy the opportunities which this work grants for teaching and educating the next generation of researchers. ‘Training and enabling students and veterinarians in research is a major component of our work,’ says Dr Tallmadge, ‘it is not unusual for colleagues (often international) to visit our laboratory for specific training.’ Dr Felippe notes that her work, as well as including teaching and lecturing, often involves her acting as a consultant for clinical veterinarians with particularly tricky cases.
Dr Felippe’s work is not limited to horses, but extends to far larger mammals as well. In her own words, ‘In the last decade, our knowledge and research tools have served projects in wildlife conservation and education in conservation. I am part of a team that studies disease risk factors for a rare population of Javan rhinos in Indonesia; through this program we also promote conservation education in the local schools and improve the capacity of local veterinarians.’ The pair also does a significant amount of work studying those viruses threatening elephants as well – as Dr Tallmadge explains: ‘I have had amazing opportunities to contribute to projects addressing a variety of species, including rhinos, elephants, and cats in addition to horses.’
There’s an old joke that human medicine is much easier than veterinary medicine – after all, how hard can learning about a single species be? The wide range of projects, species, and talents that the two researchers at Cornell share between them would seem to prove this more than ever.
Meet the researchers

Rebecca Tallmadge, PhD
Department of Clinical Sciences
College of Veterinary Medicine
Cornell University
USA
CONTACT
T: (+1) 607 253 3493
Dr Tallmadge has been in the field of equine research for many years, beginning with her first scientific employment as a technician taking part in the horse genome project. This quickly led to other roles in equine science: a PhD project examining immunological proteins, postdoctoral work studying infectious diseases and immunodeficiencies, and her current role investigating CVID. Along the way she has placed her name on over 20 publications, several book chapters, aced a couple of mentoring roles and given more conference presentations than can be counted – Dr Tallmadge has certainly had her share of success.

Maria Julia Bevilaqua Felippe, MV, MS, PhD
Department of Clinical Sciences
College of Veterinary Medicine
Cornell University
USA
CONTACT
T: (+1) 607 253 3100
Dr Felippe is a veterinary internist and immunologist, and faculty at Cornell University College of Veterinary Medicine. She oversees the Equine Clinical Immunology Laboratory, and has served as the editor of the Equine Clinical Immunology book. Inspired by her horse patients, she has dedicated the last 27 years of her career to learning about immunity, and the training of students and veterinary professionals. Dr Felippe’s research program currently studies mechanisms of immunodeficiency in the search for cure, including the use of epigenetic modifiers and gene transduction in hematopoietic regenerative medicine. And she investigates neonatal immunocompetence to better understand response to vaccines. A Nature lover, she is committed to applying her professional expertise to conservation medicine and education.
KEY COLLABORATORS
Noha Abou-Madi, DVM, MS, Cornell University
Kurnia Khairani Thalib, DVM, Cornell University
Mary Beth Matychak, Cornell University
Steven Miller, MS, Cornell University
Robin Radcliffe, DVM, Cornell University
Ute Schwab, PhD, Cornell University
Noah Cohen, VMD, MPH, PhD, Texas A&M University
Paul Soloway, PhD, Cornell University
Qi Sun, PhD, Cornell University
Lishuang Shen, PhD, Children’s Hospital Los Angeles
Charlotte Cunningham-Rundles, MD, PhD, Mount Sinai Hospital
Richard Koya, MD, PhD, Roswell Park Cancer Institute
FUNDING
National Institutes of Health New Director’s Innovator Award
USDA National Institute of Food and Agriculture
Harry M. Zweig Memorial Fund for Equine Research
Morris Animal Foundation



