Travis M Whitfill | Azim Munivar – Harnessing Skin Bacteria to Fight Skin Disease
The brainchild of Azim Munivar and Travis M Whitfill, Azitra Inc. is a microbiome start-up with big ambition. Beginning with eczema and Netherton’s Syndrome, the Azitra team are set to change the landscape of skin disease treatment.
Azitra Inc. is a company with one purpose – to deliver innovative, affordable and sustainable treatments for skin disease. How are they doing this? Through utilising the potential of the microbiome. The microbiome is the combined genetic makeup of the microorganisms present in a particular environment, in this case, the skin. The microbiome comprises billions of bacteria, which play a critical role in human health, particularly in inflammation and immune regulation. By manipulating the microbiome, Munivar and Whitfill have engineered a novel bacterial vehicle capable of secreting therapeutic treatments for dermatological conditions. As Whitfill explains: ‘The microbiome is an explosive field, and we’re using cutting-edge genetic tools to enhance natural and safe bacteria as a platform for novel therapeutics.’
Microbiome start-ups are increasingly being recognised as the way forward by pharmacological companies and funding organisations alike. So far, Azitra has received $3.75 million in funding, including a $2.9 million venture round led by Bios Partners. They also make up part of Peter Thiel’s Breakout Labs programme – a funding and support mechanism for up and coming companies in scientific fields.
This funding allows the team at Azitra to push forward with developing therapies for a wide range of skin conditions, from dry skin and eczema to MRSA and rare genetic conditions. Skin diseases constitute a high level of disease burden and will affect 30 to 70% of individuals at some point in their lives. In spite of this, current treatments for skin diseases are frequently limited to antibiotics, anti-inflammatories or steroids, due to the wide variability of cause and presentation amongst skin disorders. These are often ineffective and short lasting, with potentially negative side effects. Whitfill explains why: ‘Many patients with skin disease are suffering from symptoms that are not fully addressed with current treatment options, and we hope to provide effective therapeutics that treat the root cause of eczema and eventually other skin diseases with our platform.’ The team aim to treat the underlying causes of conditions by introducing therapeutic bacteria into the affected zone, allowing them to colonise the area and begin to correct the microbial imbalance.
‘We have created enhanced probiotics for treating skin disease. These probiotics use safe, naturally occurring bacteria from skin, and we have enhanced them to make natural proteins that can have added benefit to bacteria alone.’ – Whitfill
Foundations of Novel Drug Delivery
Azitra applies sophisticated microbiological, molecular and genetic techniques to develop patient friendly treatment options for skin conditions. ‘We have created enhanced probiotics for treating skin disease,’ says Whitfill. ‘These probiotics use safe, naturally occurring bacteria from skin, and we have enhanced them to make natural proteins that can have added benefit to bacteria alone.’

The probiotic in question is a reengineered version of the bacterium Staphylococcus epidermis (SE). SE is a bacterium that naturally lives on human skin and is usually non-pathogenic. Certain strains of SE have also shown beneficial immune effects in skin. Although this species of bacteria has historically been very difficult to modify, the Azitra team (in partnership with Jackson Laboratory and Yale University) has optimised methods of manipulating SE to develop a version that can make and secrete therapeutic proteins as it colonises the skin. In this way, it is possible to treat skin diseases that are caused by a deficient protein.
Azitra’s current phase of research and development aims to demonstrate the feasibility and potential of genetically engineered SE as a vehicle for drug delivery. One of the advantages of this approach is that SE can establish residence on the patient’s skin and continuously deliver therapeutic proteins, removing the inconvenience of frequent reapplications. Another benefit is the modularisation of the delivery system, which will allow genetic targets to be controllably swapped. This will in turn allow the team to apply the technology to a wide range of skin diseases.
Finally, it is a safe and stable treatment option. ‘Our treatment is very safe. Everyone has bacteria on their skin, and in many times their microbiome is imbalanced (i.e. dysbiosis),’ says Munivar. ‘There is evidence that by applying healthy bacteria to the skin, it can restore the dysbiosis and help restore healthy skin. Additionally, these bacteria have been shown to bolster the skin’s immunity and have conferred protective effects to the skin against pathogens and antigens. We have considerable data to show that this approach is safe, but we’ve also engineered our bacteria with additional safety features.’
Tackling Genetic Skin Disorders
The main goal of the team is to demonstrate the ability of SE to secrete a range of functional proteins with real world clinical potential in two different skin diseases: atopic dermatitis and Netherton’s syndrome. This approach will demonstrate the flexibility of the system across a range of cellular properties and functions.
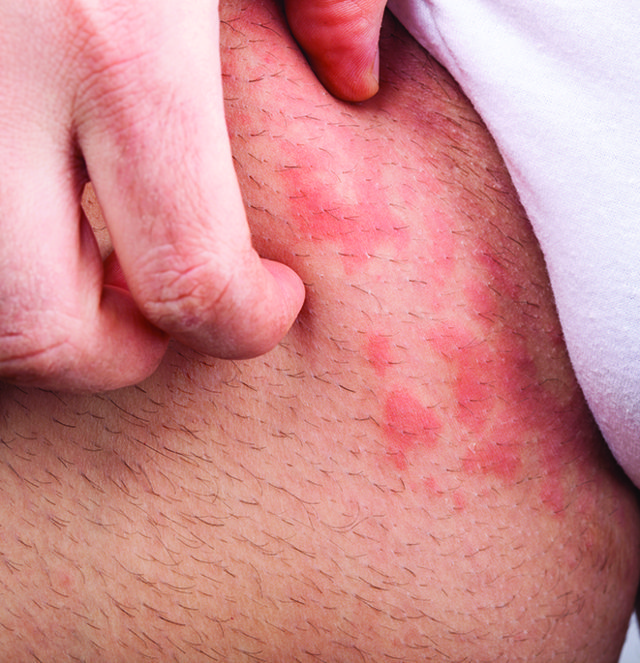
Atopic dermatitis, or eczema, is a chronic skin disease which affects 10 to 15% of children under the age of 16. Symptoms include areas of dry skin, itching, redness and, in severe cases, skin thickening, cracking and bleeding. It is associated with high costs and disease burden and there is no known cure. Mutations in the gene encoding filaggrin (a structural protein) cause the skin barrier to because disrupted, leading to inflammation and dysbiosis in the skin. ‘Studies have repeatedly shown that these patients have an imbalanced microbiome, and in the case of eczema, they often have an overgrowth of Staph aureus,’ says Whitfill. ‘There is also evidence that our strain of bacteria can kill some strains of Staph aureus, which is one of the reasons we chose it as a chassis.’
As topical filaggrin has been shown to restore the skin barrier in mice with eczema, the Azitra team have engineered SE to supplement filaggrin in the skin. Early trials confirm the potential of SE as a treatment for eczema, with patients showing increased skin water and fat content, lower skin pH and a dramatic reduction in Staphylococcus aureus (a bacterium responsible for skin infections). The treatment also colonises the skin over time, a feature that would remove the need for frequent reapplication of emulsifiers and antibiotics.
Netherton’s syndrome is a genetic disorder caused by a mutation in the gene encoding LEKTI – a protein involved in the regulation of desquamation (skin peeling). The condition presents at birth with red skin covered by fine scales, eczema and elevated systemic levels of Immunoglobulin E (an antibody associated with allergic reactions). There are currently very few treatment options for patients suffering from this condition. The Azitra team is engineering SE to secrete LEKTI onto the skin to provide an effective and convenient therapy for individuals with Netherton’s syndrome.
Key Features of the Delivery System
However, the concept of ‘bugs as drugs’ isn’t without its challenges. In order to generate a robust and flexible drug delivery system, the Azitra team must engineer some key characteristics into their design. As well as overcoming the difficulties of creating a stable chassis, there is also the issue of penetrating the stratum corneum (the outermost layer of the epidermis). Challenges that arise include susceptibility to enzymatic digestion, obstacles in solubility and diffusion due to the hydrophobic (water repellent) surface of the skin, and the layers of tightly linked cells that comprise the stratum corneum. The team have optimised human skin importability (how well the protein can permeate the layers of the skin) by engineering cell penetrating peptides (CPP) into SE. The have also maximised bacterial cell exportability, which refers to the ability of the system to export therapeutic proteins to the skin.
‘These bacteria have been shown to bolster the skin’s immunity and have conferred protective effects to the skin against pathogens and antigens.’ – Munivar
As part of Azitra’s ongoing research, the team are characterising the effectiveness of engineered SE at colonising human skin. This involves investigating the penetration depth and length of residence of SE on the skin, and evaluating different doses and application times on human explants (tissues cultured outside the body). The team’s preliminary studies in mice suggest that bacteria will colonise the surface and deep grooves of the stratum corneum and maintain a constant presence for up to a week at a time and have been engineered to rapidly die off for safety and commercial purposes. These data are essential in translating these in vitro studies to human clinical trials, and give valuable information on potential dosing strategies.
Towards the Future
So, what else is in store for Azitra? Moving to a commercial product may be a long journey, but the team has high aims for the remainder of 2017. They are establishing a well-controlled and defined cell bank and are working on developing a final strain that incorporates advanced genetic and functional features. The team is also focusing on the practicalities of developing a real product, a process which involves establishing a stable formulation at room temperature and setting up a manufacturing system that ensures products are consistent and of high quality. In the final quarter of 2017, Azitra is planning an investigator-led safety and proof-of-concept human clinical study in collaboration with their partners at Jackson Laboratory.
The coming year will see the beginning of clinical trials of SE for cosmetic indications, such as rashes, dryness and itching. Alongside this, Munivar and Whitfill will be meeting with the FDA to seek permission to carry out clinical trials of SE for eczema and other clinical indications. From there, the team can begin to carry out the studies needed to get to the clinical trial stage, which is envisioned to begin in late 2018 or 2019.
‘We are aiming to go two routes using our platform: a cosmetic application and a pharmaceutical application,’ says Munivar. ‘We are concurrently talking with the FDA for the pharmaceutical approach for treating atopic dermatitis while also heading towards a study as a cosmetic (e.g. dry or itchy skin). In the future, we hope to develop other iterations of this platform for treating additional skin diseases. We are hoping to partner with or out-license parts of this platform to pharmaceutical and consumer health companies.’
Meet the researchers

Travis M Whitfill
Azitra Inc.
400 Farmington Ave
Farmington, CT
USA
Travis M Whitfill is co-founder and Chief Scientific Officer of Azitra Inc. After training in biochemistry and microbiology at the MD Anderson Cancer Center and Duke University, Whitfill completed a Master’s of Public Health in molecular epidemiology at Yale University. He is the principal investigator on numerous federally funded grants and partnership discussions, and has raised $3.8 million in funding to date. Whitfill is also a research scientist at Yale and senior analyst and partner at Bios Research, a venture capital focused on therapeutics and medical technology. He has been presented with several awards including the Thiel Foundation Grant and SCCM Top Paper Award and the IMSH/SAEM Top Ten Papers in 2015 and 2016.
CONTACT
E: travis@azitrainc.com
T: (+1) 203 599 0700
W: https://www.azitrainc.com/

Azim Munivar
Azitra Inc.
400 Farmington Ave
Farmington, CT
USA
Azim Munivar is co-founder and Chief Executive Officer of Azitra Inc. He holds a BS in both Engineering (focusing on Chemical Biomolecular Engineering) and Economics (specialising in Finance and Healthcare Management) from the University of Pennsylvania. He also completed the Jerome Fisher Program in Management and Technology and has recently completed his medical degree at Yale University.
CONTACT
E: azim@azitrainc.com
T: (+1) 203 599 0700
W: https://www.azitrainc.com/
FUNDING
NSF
NIH
Thiel Foundation
KEY COLLABORATORS
Julia Oh PhD, Jackson Laboratory for Genomic Medicine
Leonard Milstone MD, Yale University
Jeff Bose PhD, University of Kansas Medical Center