Dr Beate Aurich | Identifying, Understanding and Managing Treatment-related Risks of Medicines Prescribed to Children
The relatively new field of paediatric pharmacovigilance aims to improve the clinical care of children by understanding and appropriately managing the risks of medicines administered to this group of patients. Dr Beate Aurich is an established expert in this field, and with colleagues, has published an article on the practical aspects of paediatric pharmacovigilance. She notes that the assessment of the benefit-risk balance of available treatment options should be based on multidisciplinary efforts and include both children and their families.
The Importance of Paediatric Pharmacovigilance
Research into the risks of medicines is called pharmacovigilance. It helps to identify, understand and reduce treatment-related risks – so-called adverse drug reactions or ADRs. Pharmacovigilance is a relatively young speciality that began to emerge in the 1930s following several major drug safety disasters.
Pharmacovigilance is a continuous process of monitoring treatment-related risks throughout the life cycle of medicinal products worldwide. It starts at the time of pre-clinical studies, i.e., before a new product is administered for the first time in humans. It is continued as long as there are still patients taking the medicine. To better understand certain long-term risks, pharmacovigilance may even be continued after a drug has been taken off the market.

Paediatric pharmacovigilance combines the understanding of general pharmacovigilance with the special requirements of children and how ADRs may affect the growing body. The overall aim is to help parents, children and healthcare professionals to make evidence-based treatment decisions by informing them about the possible risks of medicines and how these can be managed.
Dr Beate Aurich is a pharmacovigilance and drug safety consultant with extensive experience. One of her peer-reviewed publications, ‘c4c: Paediatric pharmacovigilance: Methodological considerations in research and development of medicines for children – A c4c expert group white paper’, was written in collaboration with colleagues from the conect4children (c4c) network. This white paper describes some of the points to consider with regards to the detection and proactive management of treatment-related risks in paediatric studies and paediatric clinical practice.
‘the inclusion of children and their families for the effective management of treatment-related risks should be standard practice for paediatric pharmacovigilance and risk management activities.’

Identifying Treatment-related Risks in Children
Treatment-related risks in children are often different compared to adults. They can also vary between different paediatric age groups, for example, babies and adolescents. These differences can be due to factors such as age-related changes in the body’s metabolism, comorbidities (additional illnesses) and comedications (additional medicines). A further challenge is a higher risk of medication errors because formulations are frequently not adapted to children’s needs. Medicines administered to children may need to be prepared from drugs intended for adults, for example, by crushing a tablet and this can be a source of errors. The identification of treatment-related risks in children is based on the age-group-specific safety specification.
The safety specification summarises the scientific evidence for known and potential treatment-related risks and which safety data is currently missing. The analysis of existing data may include, for example, safety data from pre-clinical studies, modelling and simulation, pharmacology, interventional and non-interventional studies, registries, safety and electronic health care databases and the literature (e.g., drug class effects, information from health authorities). The safety data is then combined with the specifics of children including, for example, paediatric pharmacology, how ADRs present clinically in children, the risk for medication errors, possible interactions with the child’s nutrition and other medicines, and the challenges of collecting and analysing safety data in children.
Managing Treatment-related Risks in Children
The risk management of medicines administered to children has similarities to risk management in other domains of our daily life. For example, car seat-belt systems are adapted to the age and size of a child. This is based on data from real and simulated accidents and the understanding of the effects of the laws of physics on a child’s body. However, many medicines prescribed to children have very limited child-specific information on treatment-related risks. The management of treatment-related risks uses standard and additional risk management tools. The product’s package insert (label) is used for standard risk management. However, this is often difficult to understand, even for adults, and frequently not adapted to the needs of children. Informing children and parents about any important updates, in particular for medicines which are taken regularly, is a challenge. Additional risk management strategies may, for example, include informed assent/consent by the prescribing doctor or targeted testing for certain ADRs (e.g., liver function, genetic risk factors).

The Importance of Patient involvement
Paediatric pharmacovigilance and risk management require a multidisciplinary effort. Dr Aurich proposes that ‘the inclusion of children and their families for the effective management of treatment-related risks should be standard practice for paediatric pharmacovigilance and risk management activities’.
SHARE
DOWNLOAD E-BOOK
LISTEN TO THE AUDIO
REFERENCE
https://doi.org/10.33548/SCIENTIA830
MEET THE RESEARCHER
Dr Beate Aurich
Pharmacovigilance and Drug Safety Consultant
Dr Beate Aurich obtained her medical degree in 1996 from Humboldt University, Berlin, Germany. In 2001, she obtained the Membership of the Royal College of Paediatrics and Child Health (MRCPCH), London, UK. Between 2002 and 2006, she worked at the Drug Safety Research Unit in Southampton, UK, as a Medical Research Fellow. She obtained a medical doctorate in pharmacoepidemiology at Portsmouth University (UK) in 2006. From 2006 to 2015, she was the Director of Global Pharmacovigilance and Drug Safety at GlaxoSmithKline and Novartis, leading the Global Pharmacovigilance Team for marketed drugs and drugs in clinical development. Between 2015 and 2021, she worked as a Project Leader in the Department of Paediatric Clinical Pharmacology at Robert Debré Hospital in Paris, France.
She has over 20 years of experience in pharmacovigilance, pharmacoepidemiology and risk management in academic research and the pharmaceutical industry. This includes the detection and assessment of safety signals, risk minimisation, safety profiling, the review of individual case safety reports and aggregate safety data and writing risk management plans. She is also experienced in reviewing protocols, informed consent forms and paediatric investigation plans, planning safety data analyses, advising on the Medical Dictionary for Regulatory Activities (MedDRA) coding and contributing to regulatory documents (e.g., clinical safety summaries, study reports, development safety update reports, periodic safety update reports).
Since February 2020, she is a member of the conect4children (c4c) Expert Group for Paediatric Pharmacovigilance, writing articles on paediatric pharmacovigilance, teaching paediatric pharmacovigilance and drug safety and advising CDISC on the particularities of capturing clinical trial data in children. Dr Aurich is currently working as a pharmacovigilance and drug safety consultant in adult and paediatric research.
CONTACT
LinkedIn: https://www.linkedin.com/in/ba-010264145/
FURTHER READING
B Aurich, D Apele-Freimane, T Banaschewski, et al., C4C – Paediatric pharmacovigilance: Methodological considerations in research and development of medicines for children – A c4c expert group white paper, British Journal of Clinical Pharmacology, 2021, 1-20. DOI: https://doi.org/10.1111/bcp.15119
B Aurich, E Vermeulen, V Elie, et al., Informed consent for neonatal trials: practical points to consider and a check list, BMJ Paediatrics Open, 2020, 4(1), e000847. DOI: https://doi.org/10.1136/bmjpo-2020-000847
B Aurich, T Martin-Montoya, D Zhang, E Jacqz-Aigrain, Reporting of offspring data in diabetes, HIV infection and hypertension trials during pregnancy: a systematic review, Archives of Disease in Childhood – Fetal and Neonatal Edition, 2020, 105(2), 215–221. DOI: https://doi.org/10.1136/archdischild-2019-316982
REPUBLISH OUR ARTICLES
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence (CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
SUBSCRIBE NOW
Follow Us
MORE ARTICLES YOU MAY LIKE
Dr Hatim Hassan | Proteins identified in gut bacteria that reduce oxalate levels
New research has identified proteins from gut bacteria, called Sel1-like proteins, that have the potential to help the body get rid of excess oxalate, an organic substance linked to kidney stones, kidney disease, and other health problems. Sel1-like proteins help the cell in assembling large molecular complexes important for cell function. Dr Hatim Hassan from the Division of Nephrology and Hypertension, Mayo Clinic, Rochester, Minnesota, United States, is part of a team of scientists researching whether these proteins and their derived peptides could reduce blood and urinary oxalate levels to prevent and/ or treat hyperoxalemia (high blood oxalate), hyperoxaluria (high urine oxalate) and related disorders (including kidney stones).
Dr Norio Mitsuhashi | Measuring Respiratory Motion to Improve Precision in Lung Radiation Therapy
Dr Norio Mitsuhashi, former Professor of the Department of Radiation Oncology at Tokyo Women’s Medical University, leads revolutionary clinical research into optimising stereotactic body radiation therapy for lung cancer. Dr Mitsuhashi and his colleagues examine whether routinely available patient and tumour characteristics can predict respiratory tumour motion, a critical source of uncertainty in high precision radiotherapy. Their findings suggest that respiratory motion cannot be reliably inferred, and must instead be measured directly in every patient.
Professor Terry C. Hrubec | Clean is good – but is too clean better?
Quaternary ammonium compounds are a large class of compounds used as disinfectants in hospitals, restaurants, healthcare and animal care facilities, and are popular as household cleaners. With disease outbreaks increasing our fears about infections, the use of disinfectants has skyrocketed in recent years. Understandably, we all want to feel safe. However, as Professor Terry Hrubec from the Department of Biomedical Sciences of E. Via College of Osteopathic Medicine discovered, such products may be causing more harm than good.
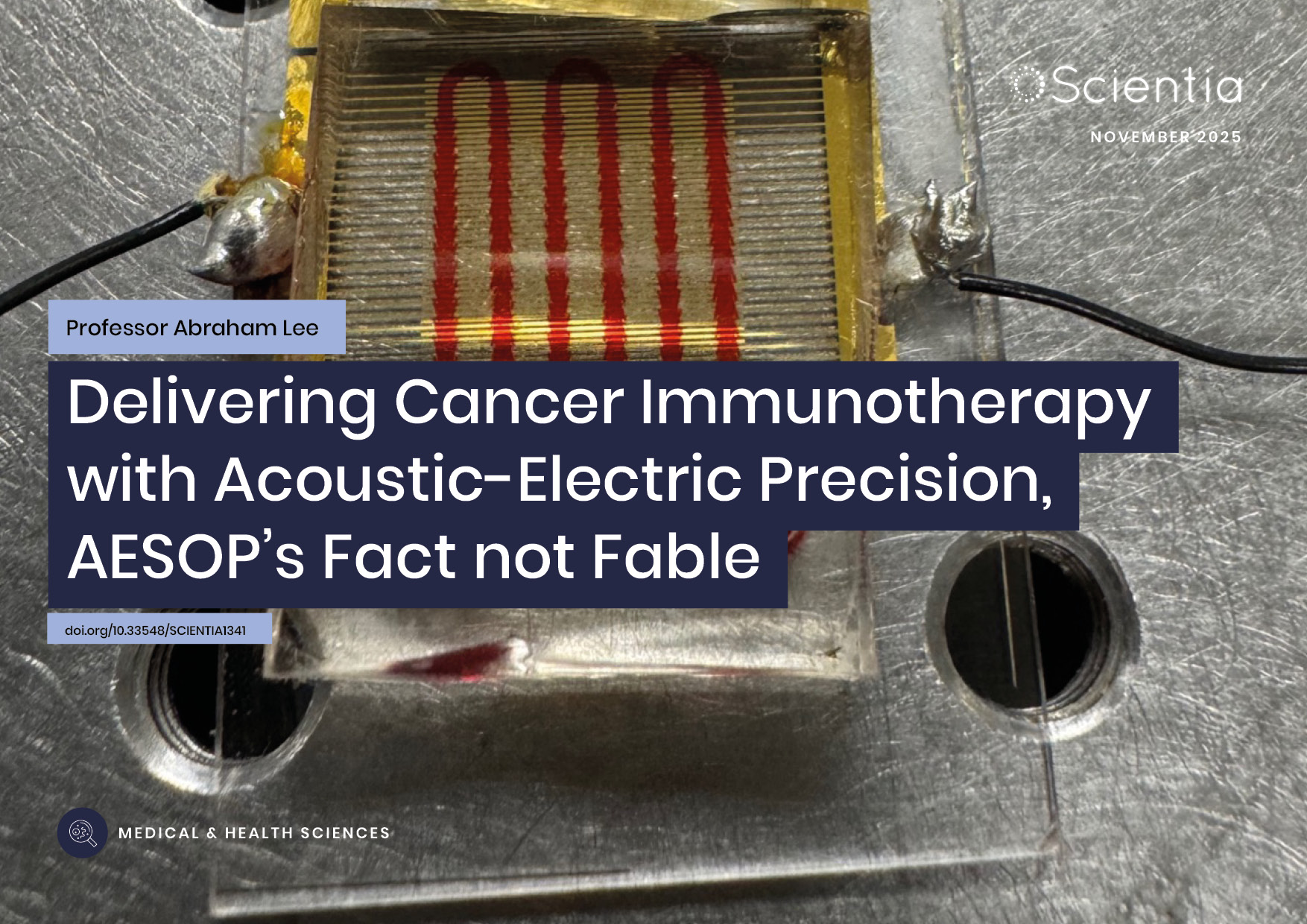
Professor Abraham P. Lee | Delivering Cancer Immunotherapy with Acoustic-Electric Precision, AESOP’s Fact not Fable
Chimeric Antigen Receptor (CAR) T-cell therapy offers life-saving potential, particularly against blood cancers, but severe side effects such as cytokine release syndrome (CRS) limit its safety. These toxicities are linked to uncontrolled CAR expression levels on the T-cell surface. Led by Professor Abraham P. Lee, researchers at the University of California, Irvine, have developed an advanced microfluidic system, called the Acoustic-Electric Shear Orbiting Poration (AESOP) platform, to precisely control the dose of genetic material delivered into primary T cells. This innovation promises safer, more homogeneous, and highly effective cellular immunotherapies.