Dr Claudine Bruck | Professor Edward Morrisey | Professor Jason Burdick – A Hydrogel with the Ability to Recover Heart Function
The human heart is a muscle, and like all types of muscles, it can be injured. In humans, heart muscle is not able to regenerate after injury, and this can lead to heart disease which develops over time, eventually leading to an untimely death. A team of researchers, Doctor Claudine Bruck (Prolifagen), Professor Edward Morrisey (Department of Medicine and Cell and Developmental Biology) and Professor Jason Burdick (Department of Bioengineering) at the University of Pennsylvania, have collaborated to develop a novel therapy to regenerate damaged heart muscle.
The Challenges of Treating Heart Disease
Heart disease is a major cause of mortality around the world and the World Health Organisation estimates that it results in close to 18 million deaths annually. Heart attack, the common terminology for myocardial infarction (MI), contributes to many cases of heart disease. Although advances in management and surgical intervention have improved survival rates, chronic heart failure (HF) is a serious consequence of MI and at least half of sufferers do not survive past five years. Repair of an MI is primarily through formation of scar tissue, as heart muscle cannot regenerate after injury. HF often develops progressively after an MI, which can eventually lead to premature death.
Cardiomyocytes are the key cells of the heart muscle. They terminally differentiate after foetal development and stop proliferating around the time of birth. Upon terminal differentiation, cardiomyocytes are unable to proliferate due to their exit from the cell cycle. Thus, when the adult heart is injured, cardiomyocytes die and cannot be replaced through proliferation or regrowth from existing cells.
Traditionally, only heart transplantation has been a cure for HF but this is costly and challenging, and therefore, not an option for all patients. The regeneration of cardiomyocytes following MI would be one way of treating damaged hearts and HF. This has long been needed and much research has been focused over the years on stem cell therapy to produce new cardiomyocytes to repair damaged heart muscle. However, the development of an effective stem cell therapy has not yet been achieved due to numerous obstacles.
Professor Edward Morrisey of the Department of Medicine and Cell and Developmental Biology at the University of Pennsylvania has invented a new cardiac regeneration technology along with Professor Jason Burdick of the Department of Bioengineering at the same institution. Professor Morrisey has co-founded the biotechnology company Prolifagen, with the drug development expertise of the company’s CEO, Doctor Claudine Bruck to further develop this exciting technology for use in the treatment of HF in humans.
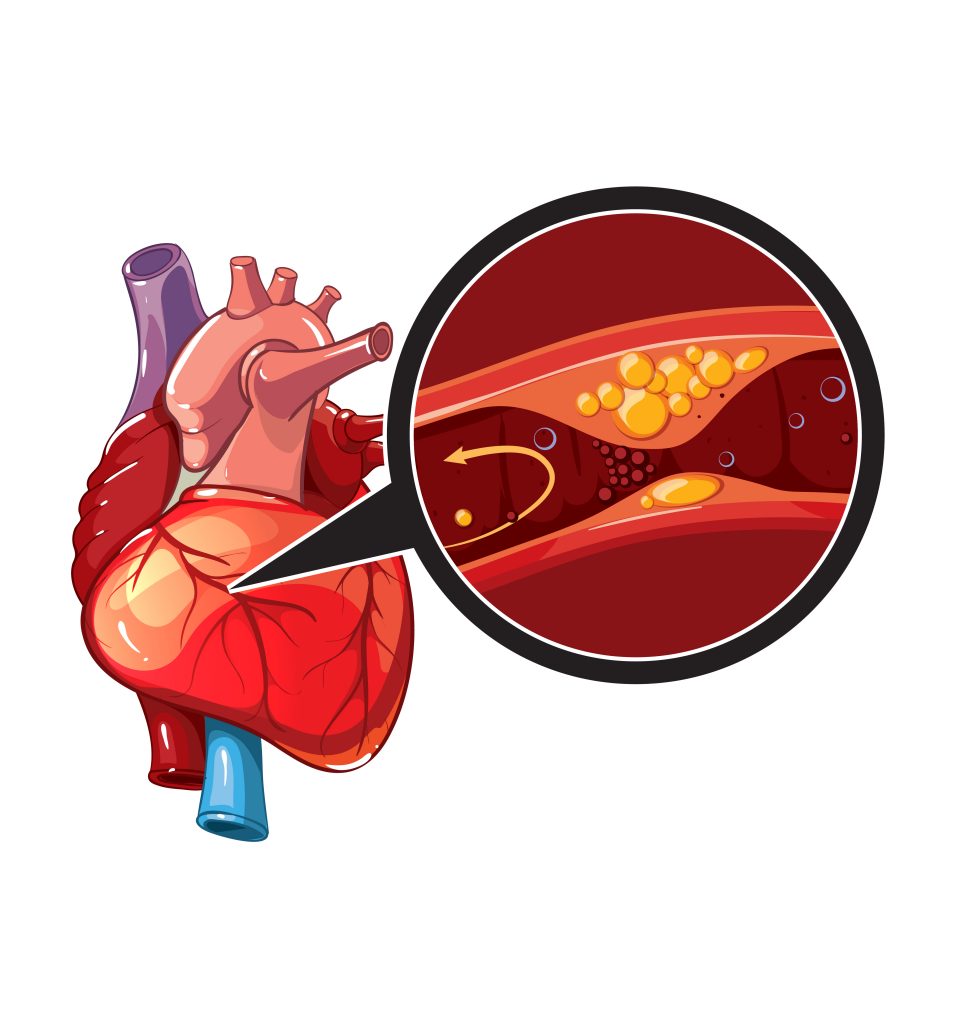
The Science Behind an Emerging Cardiac Therapy
In a 2016 research paper, Professor Morrisey and colleagues described the proliferation of cardiomyocytes in mice. MicroRNAs (miRNAs) are a class of small, non-coding RNAs that function in regulating gene expression by binding to target mRNA (messenger RNAs), destabilising them and initiating translational silencing. Professor Morrisey’s group discovered a miRNA cluster called miR302-367 that is expressed in early mouse cardiac development and plays an important role in the proliferation of cardiomyocytes during embryonic development.
The expression of miR302-367 decreases during heart development, which corresponds to decreasing cardiomyocyte proliferation. Importantly, over-expression of miR302-367 led to increased cardiomyocyte proliferation in mice as compared to controls. The researchers demonstrated that miR302-367 works by targeting components of the highly conserved Hippo signal transduction pathway. This pathway is known to control organ growth by regulating cell proliferation, apoptosis and stem cell renewal. High-throughput sequencing of mouse mRNA hybridised to miR302-367 Mst1, a core component of the Hippo signalling pathway, as a primary target of miR302-367. The team hypothesised that miR302-367 regulates cardiomyocyte proliferation by inhibiting the Hippo signalling pathway. Moreover, the importance of this pathway in cardiac regeneration has also been reported by others.
Further experiments revealed that re-expression of miR302-367 in the adult mouse heart resulted in the reactivation of the cardiomyocyte cell cycle, an increase in the number of cardiomyocytes and regeneration. When the team induced MI in these adult mice, a reduction in scar formation was observed as compared to controls. However, the team also noted that long-term expression of miR302-367 caused dedifferentiation and dysfunction of cardiomyocytes and, therefore, compromised cardiac function.
Following this, the team demonstrated that transient treatment of mice with miR302-367 mimics (miRNA that silence/repress target genes) administered daily for one week, resulted in increased mouse cardiomyocyte proliferation after MI, decreased fibrosis, and increased physiological function while at the same time not causing cardiomyocyte dedifferentiation. Importantly, there was recovery of cardiac function as measured by end-systolic, end-diastolic volume and ejection fraction. The results of these experiments led the team to conclude that there is potential for miRNA-based therapy to activate cardiomyocyte proliferation and cardiac regeneration following injury.
A Bio-engineered Injectable Therapy to Treat Heart Disease
Following the promising results of the miRNA study, Professor Morrisey and Professor Burdick collaborated to develop a miRNA-based therapy for the treatment of MI. The research was published in a 2017 paper where the team proposed delivering miR302 mimics to the heart via an injectable hyaluronic acid hydrogel. Hydrogels are water-based polymer networks, which can encapsulate drugs that are then released over time after the gel is injected into the tissue. Professor Burdick’s team developed a hydrogel with modified hyaluronic acid that exhibits shear-thinning characteristics. This allows the gel to flow during injection, and when it reaches the target site, the gel reassembles almost instantaneously.
The team studied neonatal mouse cardiomyocytes which had been treated with the gel-miR302 complex and found that the complex significantly enhanced proliferation compared to controls that were either hydrogels with no specific sequence (gel-miR-NC) or hydrogels alone. The hydrogel release profile after injection into the heart muscle indicated that the gel-miR302 complex was locally present for approximately one week from a single injected dose, whereas, in the previous study, systemic daily dosing (that is, affecting the whole body) of miR302 mimics through intravenous injection was needed.
To determine whether the cardiomyocyte proliferation following gel-miR302 treatment yielded new cardiomyocytes after MI, the team conducted a set of experiments to trace the lineage of individual cardiomyocytes using fluorescent labelling to track daughter cells from parent cells. MI was induced in mice and then injected with either gel-miR302 or gel- miR-NC (control), adjacent to the infarction site in the border zone. Mice injected with gel-miR302, but not gel- mIR-NC, showed that individual cardiomyocytes replicated into multiple cardiomyocytes in the infarcted border zone at day 28 post-injection. These data indicate that miR302 treated cardiomyocytes proliferated or grew after an MI and treatment.
The team further investigated the effect of the gel-miR302 injection on heart physiology after MI. The team selected adult mice treated with gel-miR302, gel-miR-NC or a no-hydrogel control after MI. Four weeks post-MI, echocardiography and a number of cardiac measurements were performed to assess cardiac function in treated mice. The findings showed that 4 weeks after MI, mice treated with gel-miR302 had decreased cardiac end-diastolic and end-systolic volumes (39% and 50%, respectively), improved ejection fraction (32%) and fractional shortening (64%) as compared with controls. Furthermore, echocardiography showed improved heart wall movement in gel-miR302 treated mice. The team concluded that the gel-miRNA based delivery system can be effective in promoting cardiac regeneration after MI and this method is most important as it provides a targeted and localised delivery as opposed to a systemic delivery which can lead to off-target effects.
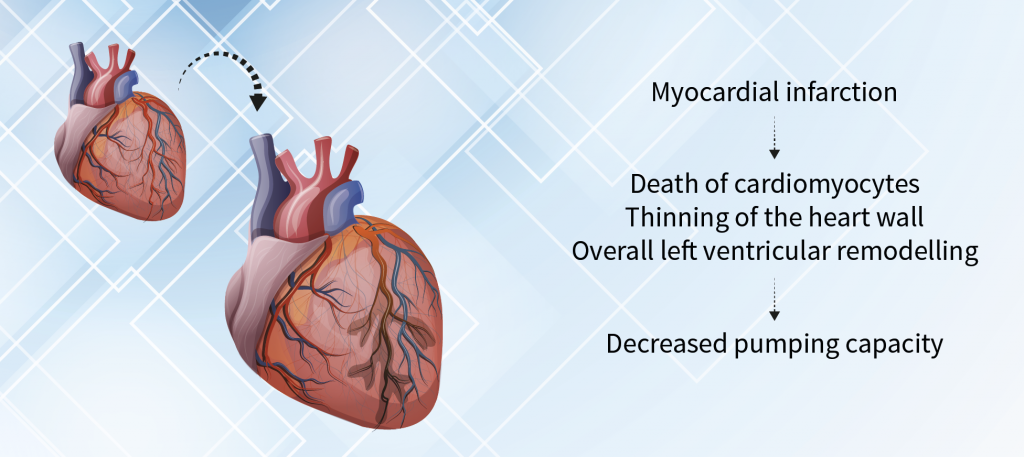
Myocardial Infarction and Heart Failure
Next Steps Towards Use in Humans
This is an exciting time for Prolifagen as they implement the next steps towards fine-tuning this new therapy as a suitable treatment in humans. Currently, the team is conducting pharmacokinetic and pharmacodynamic studies in porcine (pig) models as a proof-of-concept for human trials. For the Phase I porcine experiments, the researchers will use direct needle injections to deliver the gel-miR302 complex, however, for human trials, they propose a catheter administration.
The porcine heart is of a similar size to the human heart and will, therefore, be used to measure the induction of cardiomyocyte proliferation following injection of gel-miR302 at two different concentrations. Preliminary experiments have shown induction of cardiomyocyte proliferation following gel-miRNA302 treatment of porcine hearts. Once the porcine Phase I studies are complete the Prolifagen team aims to test the safety and efficacy of gel-miR302 in the porcine MI model.
Following success in these studies, preclinical toxicity studies will be completed ahead of early phase human trials. This novel therapy for the treatment of heart failure is a promising one with the potential of improving the outcomes for millions of patients that have suffered MI.
Reference
https://doi.org/10.33548/SCIENTIA615
Meet the researchers

Doctor Claudine Bruck
CEO and Co-founder
Prolifagen
Pennsylvania
Philadelphia, PA
United States
Doctor Claudine Bruck received her PhD in Biochemistry and Molecular Virology at the University of Brussels in 1982 and completed her postdoctoral research at Harvard Medical School. Doctor Bruck has 30 years of experience leading the pharmaceutical industry in vaccine research and drug development. She is the former vice-president and Head of GSK Ophthalmology R&D and currently the CEO and co-founder of Prolifagen where she is managing the development of the company’s cardiac regeneration technology.
CONTACT
E: clemb11@gmail.com

Professor Edward E. Morrisey
Departments of Medicine and Cell and Developmental Biology
University of Pennsylvania
Philadelphia, PA
United States
Professor Edward E. Morrisey received his PhD in Molecular and Cell Biology from Northwestern University in Illinois in 1994. He is currently Professor of Medicine and Cell and Developmental Biology, Director of Penn Centre for Pulmonary Biology, and Scientific Director of Penn Institute for Regenerative Medicine. Professor Morrisey’s laboratory focuses on cardiac and lung development mechanisms to improve the repair and regeneration of these tissues. He is also the co-founder of biotech company Prolifagen which is developing a novel therapy to regenerate cardiac tissue.
CONTACT
E: emorrise@pennmedicine.upenn.edu
W: https://www.med.upenn.edu/morriseylab/

Professor Jason A. Burdick
Departments of Bioengineering
University of Pennsylvania
Philadelphia, PA
United States
Professor Jason A. Burdick obtained his PhD in Chemical Engineering in 2002 from the University of Colorado and is currently Professor of Bioengineering at the University of Pennsylvania and the Director of the Polymeric Biomaterials Laboratory at Penn. Professor Burdick and his team focus on the development of polymeric materials for regenerative medicine and drug development. Professor Burdick holds numerous honours and awards and is a member of the editorial board of multiple bioengineering publications.
CONTACT
W: http://www.seas.upenn.edu/~burdick2/
Twitter: @BurdickLab
FURTHER READING
LL Wang, Y Liu, JJ Chung, et al., Sustained miRNA delivery from an injectable hydrogel promotes cardiomyocyte proliferation and functional regeneration after ischaemic injury. Nature Biomedical Engineering, 2017, 1, 983–992.
Y Tian, Y Liu, T Wang, A microRNA-Hippo pathway that promotes cardiomyocyte proliferation and cardiac regeneration in mice, Science Translational Medicine, 2015, 7, 279ra38.

Want to republish our articles?
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence
(CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
More articles you may like
Ms. Aikaterini Dritsoula | Looking Beyond Snoring: How Hidden Airway Problems Shape Children’s Sleep
For many parents, a child’s snoring may seem harmless, even endearing. Yet in some cases, it signals something more serious. Obstructive sleep apnoea is a condition in which a child’s breathing is repeatedly disrupted during sleep. These interruptions can affect growth, behaviour, and learning. Children with this condition may toss and turn at night, struggle to concentrate during the day, or show signs of hyperactivity and fatigue. Traditionally, enlarged tonsils and adenoids have been seen as the main culprits. Surgery to remove them has long been considered the standard treatment. However, research led by Consultant ENT Surgeon Ms. Aikaterini Dritsoula of The Leeds Teaching Hospitals NHS Trust invites us to look deeper. Her work suggests that the story is often more complex, especially in very young children.
Sara F Martin | The New Paradigm: Two Fundamental 22-year Solar Cycles Always Present on the Sun
For millennia, humans have looked up towards the life-giving Sun and sought to understand its nature. One of its earliest features noticeable before the age of technology was the presence of small dark patches scattered across its surface – sunspots. These blemishes appeared to wax and wane on a regular 11-year cycle, which was thought for over a century to be a fundamental time period governing the Sun’s magnetic activity. But new discoveries suggest a radically different understanding where sunspots are merely peak phases of two, more fundamental 22- year magnetic cycles present simultaneously in different bands of latitude.
Professor Neil Coffee – Professor Vincent Versace | Mapping Health Access: Using Address-Level Intelligence for Smarter Services
Accessing healthcare is a serious challenge for people living in rural and remote Australia. Large distances, sparse populations, and limited services can prevent residents from receiving care when they need it. Professors Neil Coffee and Vincent Versace at Deakin University’s Centre for Australian Research into Access (CARA) are leading research to model healthcare service access across the country, to provide new insights that can guide health planning and policy, as well as other services such as education. This work combines the curation of detailed address level residential dwellings and road network data to calculate access to service metrics (time and distance). These metrics are applied to the simulated residential dwelling population, to quantify the population with poor access to health services.
Prof Candis M. Morello – Prof Jan D. Hirsch | Recent innovations in pharmacy education
A pioneering research team from the Skaggs School of Pharmacy and Pharmaceutical Sciences, University of California, San Diego, United States, has been instrumental in developing innovative techniques for teaching pharmacy students. The Next Generation of Pharmacist Educators (NextGen-RxEd) programme is a new method of training the next generation of pharmacist educators and academics. To help pharmacists and pharmacy students visualise the complex issues experienced by their patients, the team led by Professors Candis Morello and Jan Hirsch developed an innovative educational tool, called the Medication Therapy Management (MTM) Spider Web.




