Dr Evelyne Deplazes – Combining Simulations & Experiments to Explore Interactions Between Membranes & Small Molecules
Many important processes in our bodies rely on the transport of small molecules and ions across cell membranes. However, these processes can be extremely intricate and complex, and are frustratingly difficult to study. Dr Evelyne Deplazes and her team at the University of Queensland and the University of Technology have been investigating these processes by combining the best elements of experimental and computational membrane biophysics. This research could help us better understand the way our bodies work, with exciting implications for biotechnology and drug development.
Movement of Ions and Molecules
Of the many complex processes that take place in our bodies, one of the most important is the transport of ions and small molecules into and out of our cells. Each of our cells is enclosed by a membrane made up of fatty molecules called phospholipids, which protects the cell’s interior from the external environment, and controls which substances can pass through. When an ion or molecule interacts with the surface of a cell, the membrane can allow it to pass through under the right conditions. However, the nature of these interactions is not yet fully understood.
Naturally, scientists have been searching for new ways to study these processes in order to learn more about them. Such improved knowledge would be invaluable for numerous different research fields – from understanding how certain diseases develop to designing effective methods for delivering drug molecules into cells.
‘The transport of small molecules across membranes is important to understand both fundamental biological processes as well as for many applications in biotechnology and drug development,’ explains Dr Evelyne Deplazes, researcher at the University of Queensland.
Unfortunately, due to their complexity, these interactions are extremely challenging to study in the laboratory. Computational chemists have developed simulations that allow for more in-depth analysis, but these are not without their own limitations based on processing power and computation time. These simulations also need to be validated, ideally by comparing their results to experimental data collected from the system being simulated. Dr Deplazes and her colleagues have been tackling these issues by using a unique combined method.
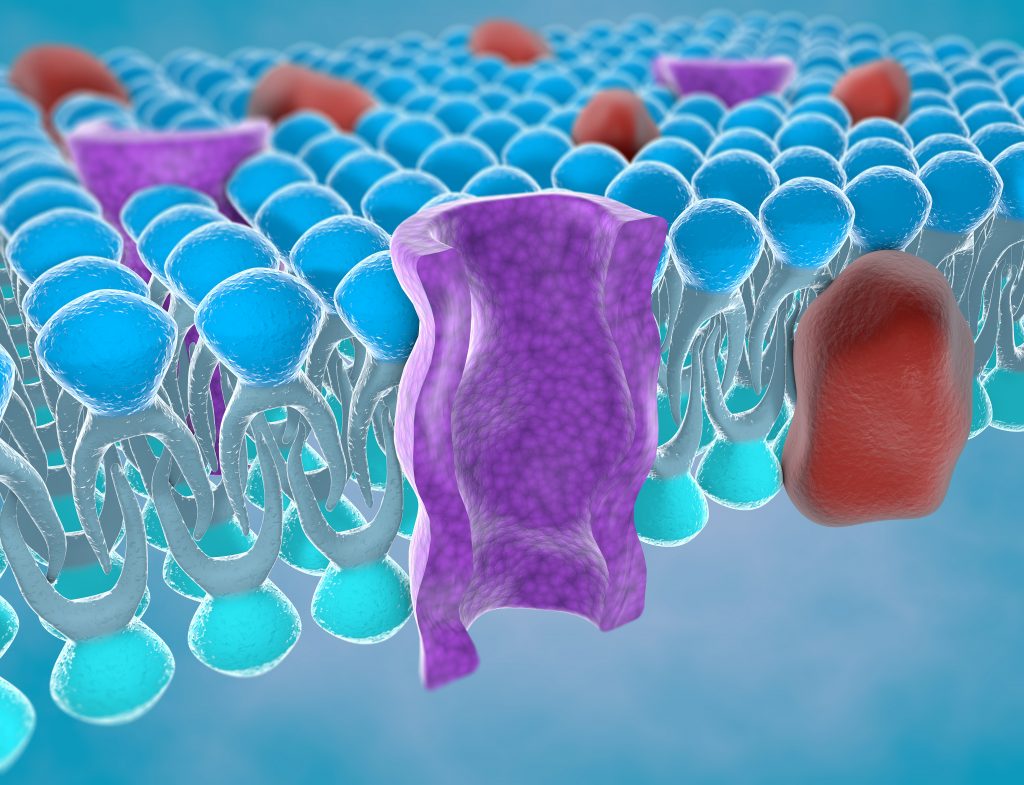
Exploring Sodium and Potassium Transport
Dr Deplazes and her colleagues began their investigations by using ‘tethered membranes’, which are essentially phospholipid molecules anchored to a substrate made of pure gold. These model membranes mimic the fluidity that would be found in a cell membrane, but in an environment where lipid composition and other variables such as pH and ion concentrations can be controlled. This allows the team to isolate different effects in a way that is usually not feasible using cell membranes.
By measuring the electrical properties of the membrane in the presence of different concentrations of sodium and potassium ions, the team was able to learn more about how these two important ions interact with the surface. Sodium and potassium ions play key roles in countless biological processes, including water regulation, muscle contraction and the transmission of nerve signals.
At low concentrations, the researchers found that sodium decreased the membrane’s electrical conductance, while potassium increased this property. However, they only observed this effect with certain types of phospholipid membranes, and not others, implying that the effect was caused by specific interactions between the phospholipid molecules and the ions.
To further investigate this phenomenon, the team implemented molecular dynamics simulations. This computational approach is used in chemistry research to simulate the ways in which molecules interact with each other. From these simulations, the team gathered data showing that sodium was more likely to interact with multiple phospholipid molecules than potassium. Sodium was also more likely to be found at the surface of the membrane. Dr Deplazes explains that these differences could be leading to a change in the way the molecules are able to move on the surface, hence a change in conductance.
This study has not only generated valuable information on the way in which our cells can interact with ions, but also made a strong case for the effectiveness of combining computational and experimental methods for probing biophysical mechanisms.
Membrane Pores
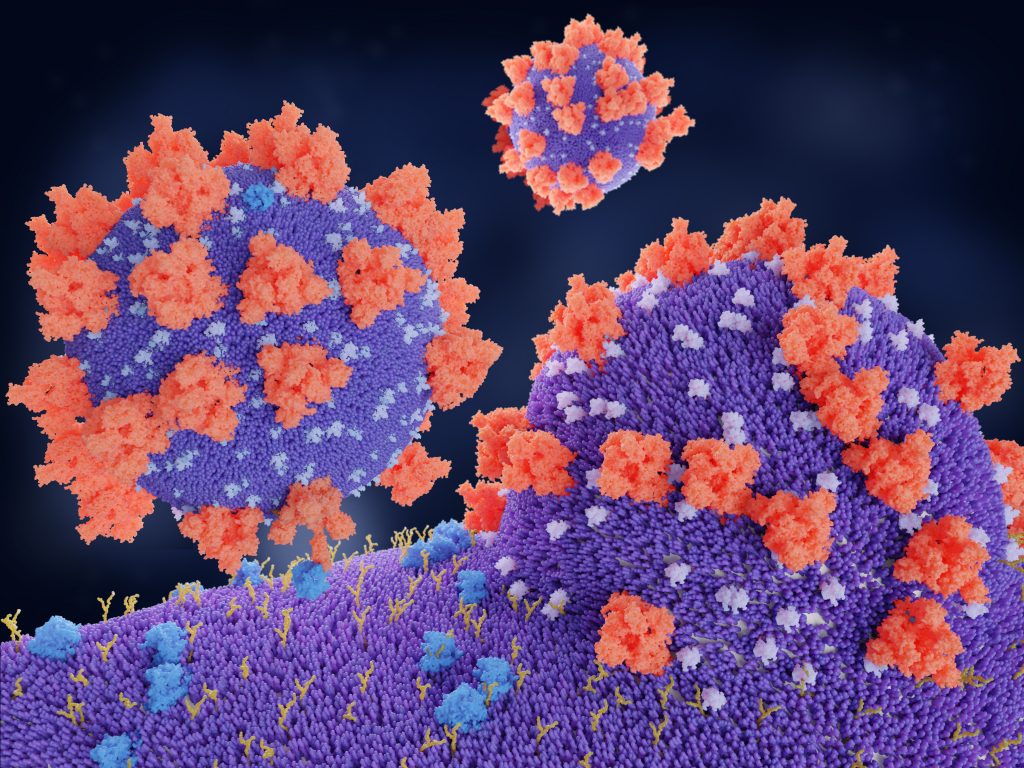
Nearly every disease-causing virus we know about interacts with our bodies in a similar way: by entering our cells through the outer membrane. Once the virus is inside a cell, it can start hijack the cell’s machinery and start replicating itself, allowing it to spread to other cells. This is done by all kinds of viruses, including HIV, measles, and the virus that causes COVID-19.
The process of viruses taking over the body’s cells is referred to as viral-membrane fusion. Scientists have studied this process extensively, to better understand how exactly viruses can pass through the cell membrane. Understanding how the virus does this may help scientists to identify ways to prevent it from happening. An improved understanding of this process would also be invaluable for vaccine research, as many of today’s vaccines – including some of those for COVID-19 – deliver genetic code to our cells inside a harmless viral ‘shell’.
Scientists have previously explored how viruses penetrate the cell membrane using a special molecule called ‘GALA’ – a synthetic protein designed to mimic the way viruses interact with phospholipid membranes. Dr Deplazes and her team have been investigating how GALA molecules are able to open pores in the cell membrane, by using a combination of experiments and molecular dynamics simulations.
It was already understood that, under acidic conditions, GALA molecules would assemble and insert themselves into the cell membrane to create an open pore. It was previously believed that each of these pores was made up of 8–12 individual GALA molecules. Dr Deplazes and the team have demonstrated that it is more likely that these pores are made up of 6 molecules instead. This is important information, as it tells us more about the size of the pores and what they could let through. ‘This combined approach, for the first time, directly provides structural information to estimate the size of pores formed by a peptide similar to the ones found in viruses,’ explains Dr Deplazes.
Furthermore, the team found the pores showed selective permeability – allowing some ions to enter more easily than others. Their results could represent an important step in learning more about how GALA, and thus viral fusion peptides, operate.
‘The transport of small molecules across membranes is important to understand both fundamental biological processes as well as for many applications in biotechnology and drug development.’
How Can Small Molecules Alter Membranes?
Understanding how small molecules interact with phospholipid membranes is important for many different applications, including drug development and biotechnology.
By integrating molecular dynamics simulations and experiments with tethered membranes, Dr Deplazes and her team have developed a powerful approach to gain valuable insights into molecule–membrane interactions at the molecular level. The lab’s ongoing research includes studying the membrane-altering effects of steroids, steroid-like molecules and phenolic acids.
Phenolic acids are molecules that are commonly found in plants. Present in many plant-based foods, phenolic acids have been extensively studied for their antioxidant, anti-inflammatory and pro-coagulant properties.
Some of these biological activities are related to the interaction of phenolic acid molecules with cell membranes. However, compared to their activities inside cells, very little is known about how these molecules interact with the cell membrane.
Dr Deplazes and her team’s tethered membrane experiments show that certain phenolic acids, such as caffeic acid, caffeic acid methyl ester and kojic acid, change the permeability of phospholipid membranes to varying degrees. In contrast, other phenolic acids with similar structures, including p-coumaric acid, gallic acid and syringic acid, do not alter membrane permeability.
To understand why certain phenolic acids can alter membrane structures, while others cannot, the research team performed experiments with different types of phospholipid, to study specific interactions between phenolic acid molecules and the lipid component of phospholipid molecules. They used the data they collected to validate simulations that provide structural information on these interactions. ‘Our results from these simulations suggest that membrane disruption is related to the phenolic acids inducing local and temporal changes at the water-phospholipid interface without changing the overall shape of the membrane,’ says Dr Deplazes.
In another study, her team investigated the interaction of steroids and steroid-like molecules with phospholipid membranes. Steroids include molecules that naturally occur in our body, such as progesterone and testosterone, and drugs used to treat inflammatory conditions including arthritis, eczema and asthma. The team’s tethered membrane experiments showed that these clinically used steroids directly alter the permeability of cell membranes. Their results will be useful for designing more effective steroid-based drugs for a range of inflammatory diseases.
A Winning Combination
Throughout her research, Dr Deplazes has demonstrated how a combination of computational and experimental chemistry can answer questions that one method alone could not. ‘Our lab is one of the few labs where biophysical experiments and molecular dynamics simulations are directly combined,’ she says.
Her team’s simulations and tethered membranes are highly effective for studying molecular interactions in systems where conditions can be controlled. To translate their findings into living systems, however, the team collaborates with microbiologists, molecular biologists and molecular biophysicists.
In collaboration with these researchers, the team’s unique approach continues to provide deep insights into the behaviour of cell membranes, with important implications in numerous fields, including drug delivery, vaccine development, and treatments for infectious diseases, cancers and neurological disorders.
Reference
https://doi.org/10.33548/SCIENTIA686
Meet the researcher

Dr Evelyne Deplazes
School of Chemistry and Molecular Biosciences
University of Queensland
Brisbane
Australia
Dr Evelyne Deplazes earned her PhD in computational biophysics from the University of Western Australia in 2012, before being awarded early career fellowships by the Swiss National Science Foundation and the Australian National Health and Research Council. She worked as a research fellow at the University of Queensland, Curtin University and the University of Technology Sydney, before returning to the University of Queensland, where she holds her current position as Senior Lecturer. As a biophysical chemist, Dr Deplazes’ research integrates wet-lab and computational approaches to study how small molecules such as drugs or peptides interact with membranes. She uses this knowledge to further our understanding of biological and (bio)chemical processes and to facilitate the development of pharmaceuticals and drug delivery systems. In addition to her research, Dr Deplazes is passionate about training up the next generation of scientists and teaching them how to be critical thinkers. Through her mentoring and leadership activities, she is actively involved in promoting diversity, equity and inclusion in STEM and higher education.
CONTACT
E: e.deplazes@uq.edu.au
W: http://researchers.uq.edu.au/researcher/8833
KEY COLLABORATORS
Dr Charles Cranfield, University of Technology Sydney
Dr Nural Cocketin, i3 Institute
Adj Prof Bruce Cornell, Surgical Diagnostics, Sydney
Prof Chris McDevitt, University of Melbourne
Dr Maria Ikonomopoulou, Madrid Institute of Advanced Studies
FUNDING
Australian National Health and Medical Research Council
Cancer Council WA (Western Australia)
University of Technology Sydney
FURTHER READING
E Deplazes, LM Hartmann, CG Cranfield, A Garcia, Structural Characterization of a Cation-Selective, Self-Assembled Peptide Pore in Planar Phospholipid Bilayers, The Journal of Physical Chemistry Letters, 2020, 11, 8152.
E Deplazes, BD Tafalla, CG Cranfield, A Garcia, Role of Ion–Phospholipid Interactions in Zwitterionic Phospholipid Bilayer Ion Permeation, The Journal of Physical Chemistry Letters, 2020, 11, 6353.

Want to republish our articles?
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence
(CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
More articles you may like
Grandmothers: Innovation Through Tradition
Grandmother Project – Change through Culture (GMP) is an organisation dedicated to documenting the role of grandmothers and demonstrating the effectiveness of grandmother-inclusive strategies in improving the health and well-being of women, children, and adolescents. GMP’s groundbreaking work challenges conventional wisdom to transform community-based interventions in Africa and beyond, harnessing a powerful but often overlooked resource: the wisdom and influence of grandmothers.
Dr Robert Larkin | Cultivating Change to Improve Soil Health and Increase Potato Yield
Environmental quality and food production are facing the pressing challenges of climate change and global population growth. Dr Robert Larkin from the United States Department of Agriculture-Agricultural Research Service (USDA-ARS) and a team of plant scientists developed and tested a range of crop management systems to help overcome these compounding challenges. Their work is improving soil health and increasing the yield of potato crops, contributing to the future food security of nations.
Professor Giorgio Buttazzo | Artificial Intelligence and a Crossroads for Humanity
Where do we stand with artificial intelligence? Might machines take over our jobs? Can machines become conscious? Might we be harmed by robots? What is the future of humanity? Professor Giorgio Buttazzo of Scuola Superiore Sant’Anna is an expert in artificial intelligence and neural networks. In a recent publication, he provides considered insights into some of the most pressing questions surrounding artificial intelligence and humanity.
Dr Ralf Adam | New Technologies Shaping the Future of Oral Hygiene
Understanding the efficiency of various toothbrush technologies is essential for achieving optimal oral health. Dr Ralf Adam, who leads a dedicated team at Procter & Gamble in Germany, is keen to investigate the complexities of these technologies. His team have provided new insights into the best toothbrush types for plaque removal and the maintenance of gum health. By highlighting the importance of informed oral care decisions and ongoing investigations, this vital research works towards ensuring everyone can achieve a brighter, healthier smile.




