Dr. Hamdy Awad – Eliminating Paralysis After Aortic Aneurysm Surgery
Ruptured and dissected aneurysms are medical emergencies that can have fatal consequences. There are two main surgical procedures to repair a ruptured aneurysm: open surgery and endovascular aneurysm repair. Unfortunately, both methods present a risk of developing spinal cord injury and paralysis. In addition, patients who develop paralysis after surgery have a significantly lower survival rate compared to non-paralysed patients. Dr. Hamdy Awad at The Ohio State University has spent most of his academic career focusing on the development of preclinical small and large animal models to understand the mechanisms of ischaemic spinal cord injury and discovery of novel therapeutics to prevent paralysis after aortic aneurysm surgery.
Aortic Aneurysm: Incidence and Causes
The aorta acts as the main highway for the transport of oxygen-rich blood around the whole body. To carry out its functions, the aorta is made of strong, elastic walls that enable it to withstand the high pressure of the blood flowing from the heart. An aortic aneurysm is a condition where the walls of the aorta start to weaken and dilate, causing loss of elasticity and increasing the risk of developing ruptures in the aortic wall. Aortic dissection is a condition caused by a tear in the aortic wall, which also leads to rupture.
Aortic aneurysms cause about 12,000 deaths every year in the USA. Several environmental factors increase the risk of aortic aneurysms in humans. The most important factor affecting the integrity of the aortic wall is smoking, which accounts for about three-quarters of all cases of abdominal aortic aneurysms. Other environmental factors include high blood pressure, high cholesterol and atherosclerosis. Rare inherited conditions affecting the connective tissue, such as Marfan syndrome and Ehlers-Danlos syndrome also increase the risk of an aortic aneurysm.
Dr. Hamdy Awad from the Department of Anesthesiology at The Ohio State University Wexner Medical Center has spent most of his clinical and academic career focusing on furthering understanding of the mechanism of ischaemic spinal cord injury and paralysis through the development of preclinical small and large animal models to simulate the clinical paradigm of aortic aneurysm. His aim is to reduce the number of patients who develop paralysis after the surgery, a devastating postoperative complication that is caused by ischaemic injury of the spinal cord.
Surgery of the Aorta: Life-Saving Treatment or Russian Roulette?
Acute aortic syndrome includes aortic dissection, intramural haematoma and aortic ulcer. During an aortic dissection, the inner layer of the aorta ruptures. The blood flows through the tear, causing the inner and the middle layer of the aortic wall to separate. If untreated, an aortic dissection can be fatal, because of the risk of internal bleeding.
An intramural haematoma consists of a separation of the inner and middle layers of the aorta, with blood leaking through them. Unlike an aortic dissection, the intramural haematoma does not occur because of a tear in the inner layer of the aorta. Aortic ulcers are formed when atherosclerotic plaques damage and weaken the structure of the inner lining of the aorta, making it prone to dissection.
Despite the large amounts of funding by the National Institutes of Health in the USA and other institutions around the globe, the research to develop an effective pharmacological therapy for the medical treatment of severe aortic aneurisms and acute aortic syndrome remains inconclusive, and surgery is to date the only effective method to replace a diseased aorta and/or prevent the fatal consequences of a ruptured aneurysm.
There are two main surgical options to repair a diseased aorta prior to or following a ruptured aneurysm: open surgery and the less invasive endovascular aneurysm repair. In both methods, a graft is used to replace the aneurysm. Unfortunately, both procedures also present a risk of developing ischaemic spinal cord injury and paralysis after surgery.
The mechanisms behind these devastating post-surgery complications are not well understood. However, one plausible explanation is that the clamping and unclamping procedures employed in aortic surgery affect the flow of oxygenated blood feeding the spinal cord tissue, resulting in ischaemia and reperfusion injury. Another possibility is that debris from atherosclerotic plaques can become dislodged during the surgical procedure, occluding the blood vessels that supply the spinal cord with blood. Another possibility is the starvation of the spinal cord tissue due to lack of blood flow, which carries substrates such as fatty acids, lactate and oxygen.
Dr. Awad faced the traumatising experience of caring for a patient who survived surgery but later developed ischaemic injury to the spinal cord, followed by paralysis. Dr. Awad was a cardiothoracic anaesthesiologist fellow at the Cleveland Clinic at that time. His patient was an asymptomatic 55-year-old engineer who found out about his thoracic aortic aneurysm after a routine chest X-ray. The patient debated the pros and cons of having surgery versus the risk of developing paralysis. Given the potentially fatal consequences of leaving the aneurysm untreated, he opted for surgery. Unfortunately, the patient woke up from surgery paralysed. Since then, Dr. Awad has made it his mission to find a solution to the ‘Russian roulette’-type dilemma faced by cardiovascular surgeons, anaesthesiologists and patients when opting for aneurysm aortic surgery.

Chimney graft technique for left subclavian artery revascularization. A thoracic aortic endograft is excluding a descending thoracic aneurysm and covering the origin of the left subclavian artery (LSA); a chimney graft is inserted in the LSA parallel to the proximal wall of the aortic stent. The top left figure shows a cross section of the aortic arch with the proximal end of the thoracic stent and the LSA chimney graft in place. The bottom left figure shows a cross section of the distal portion of the aortic stent sealing the descending thoracic aorta. LSA: Left subclavian Artery; L: LeftReproduced with permission from Awad et al, 2017, Spinal cord injury after thoracic endovascular aortic aneurysm repair, Canadian Journal of Anesthesia.
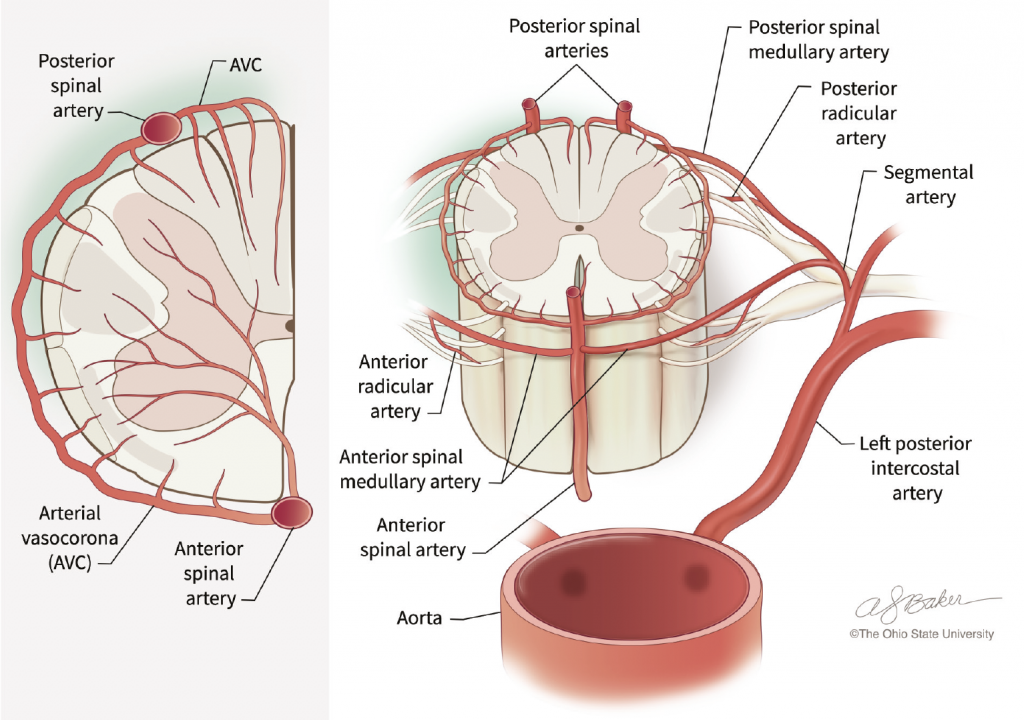
Mapping Out a Complex Network of Spinal Cord Vasculature in Normal and Diseased Conditions
The network of vessels feeding the spinal cord with blood is one of the most complex in the human body. To make matters more complicated, researchers can only rely on a very limited range of animal models that are suitable to understand the pathophysiology behind the post-surgery damage to the spinal cord.
Dr. Awad and his team published a book chapter highlighting the significant differences in blood supply to the spinal cord across different species, and described the complexity of identifying a suitable animal model that would allow vascular scientists and surgeons to simulate the ischaemic injury to the spinal cord observed in humans.
Despite the difficulties, Dr. Awad and his collaborators developed mouse and canine models to shed light on the pathophysiology of the potential ischaemic damage to the spinal cord. The team investigated the effects of cross-clamping the descending aorta on the development of spinal cord injury with delayed paralysis in mice. All mice tested in the study were able to move for up to 24 hours after the procedure, after which they all developed mild or severe paralysis of the hind limbs around 40 hours after aortic cross-clamp. The study also showed that it is not possible to predict beforehand which animals will be severely or mildly affected by the procedure, despite having a similar genetic background and surgical procedure. The two primary factors controlling paralysis were ischaemia time and temperature during aortic cross-clamp.
The team also studied the effect of post aortic surgery inflammation in large animal models on the delayed onset of paralysis. Following the aortic cross-clamping procedure, the animals displayed increased levels of oxidative stress in plasma and the cerebrospinal fluid (CSF) and increased levels of inflammation markers in CSF samples, indicating that the inflammation response is initiated within the central nervous system. Reperfusion injury of the spinal cord caused hind limb paralysis within a three hour interval and this correlated with white blood cell infiltrates three days post-ischaemia. The team used large animals to obtain CSF, which cannot be obtained in the rodent models. They were also able to compare CSF and blood samples from large animal models, as well as from humans, with and without paralysis.

Reproduced with permission from Awad et al, 2017, Spinal cord injury after thoracic endovascular aortic aneurysm repair, Canadian Journal of Anesthesia.
Spinal Cord Protective Strategies During Aortic Aneurysm Surgery
Dr. Awad and his collaborators aim to use their animal models to understand how the pathophysiology of ischaemic injury of the spinal cord differs in the open surgery procedure when compared to the endovascular aneurysm repair approach. Although the risk of developing paralysis is similar in both procedures, endovascular surgery correlates more with the incidence of delayed onset paralysis and less with acute paralysis. This indicates that the two procedures cause different types of damage to the white and gray matter tissue of the spinal cord, and Dr. Awad’s team is actively investigating the variables at play behind this difference between open and endovascular repair in the mouse and canine models available.
The team also published a review that evaluated the effectiveness of several spinal cord protective strategies during aortic aneurysm surgery. Among others, the measures adopted to prevent ischaemia include cerebrospinal fluid drainage, the induction of a mild state of hypothermia and optimal blood pressure management. However, to date, there is not a single pharmacologic agent to prevent or protect the spinal cord from ischaemic damage in humans.
There are two methods that the perioperative team utilise to protect the spinal cord from ischaemia: increasing the spinal cord perfusion pressure and drainage of CSF using a spinal drain. The procedure for the insertion of a spinal drain is not devoid of minor and major complications. Drains are usually placed using anatomical landmarks by anaesthesiologists before surgery. The procedure can be technically challenging, often leading to multiple insertion attempts. Fluorescent image-guided spinal drain insertion is an alternative to the conventional blind technique used by anaesthesiologists. It improves the accuracy of correct spinal drain insertion, reducing the potential development of post-surgery complications.
Dr. Awad and his team believe that because of the advent of these endovascular therapies, the overall numbers of patients presenting with thoracoabdominal aortic aneurysm who are now being offered therapy are increasing, potentially increasing the numbers of patients with paralysis who are being seen in health systems. This demands urgency of a solution to the issue of ischaemic spinal cord injury and paralysis in the setting of aortic aneurysm surgery. Dr. Awad and his team have made significant advances towards the understanding of this complex problem that is a major complication in these patients, contributing to both increased costs, significant patient suffering and loss of life.
Future Directions and Biobanking
The team hopes to establish a multicentre blood and cerebrospinal fluid biobank that stores biological samples from patients who have undergone aortic aneurysm surgery in North America and potentially globally, to ascertain what cellular and molecular components are altered in humans that might lead to ischaemic injury of the spinal cord. The human samples will be used to guide the preclinical work performed in animal models and contribute to understanding of the cellular and molecular mechanisms behind ischaemic spinal cord injury and paralysis in the surgical setting. The long-term goal for Dr. Awad’s team is to eliminate the onset of paralysis after aortic aneurysm and dissection surgery. Dr. Awad established and personally funded a university fund to achieve the goal. He hopes that additional donations can help fund the research and the recruitment of skilled personnel needed to understand and resolve the problem of ischaemic injury of the spinal cord after surgery of the aorta.
Reference
https://doi.org/10.33548/SCIENTIA565
Meet the researcher

Hamdy Awad, MD
Wexner Medical Center
Department of Anesthesiology
The Ohio State University
Columbus, OH
USA
Dr. Hamdy Awad received his medical degree in 1983 at the Zagaziz University, Egypt and specialised in anaesthesiology in 1987 at the same institution. He moved to the USA in the early 1990s and obtained a research fellowship in anaesthesiology at the University of California, San Diego. He then completed his residency training at the University of Texas Southwestern before completing a fellowship in cardiothoracic anaesthesiology at the Cleveland Clinic. In 1998, Dr. Awad moved to The Ohio State University where he is now a tenured associate professor in the Department of Anesthesiology. He has been actively involved in teaching medical students, anaesthesiology residents and cardiothoracic fellows throughout his career. Dr. Awad is also an extremely active clinical researcher and currently holds three USA patents. He has published extensively in prestigious journals and books, reflecting his deeply rooted commitment to developing methods to prevent spinal cord injury following aortic aneurysm repair.
CONTACT
E: Hamdy.Elsayed-Awad@osumc.edu
Mobile: +1(614) 5601505
KEY COLLABORATORS
Esmerina Tili, PhD, The Ohio State University, Wexner Medical Center
FUNDING
National Institutes of Health
FURTHER READING
H Awad, TJ Huang, E Tili, et al, Images in Anesthesiology: Spinal Subdural Hematoma after Spinal Drain for Endovascular Thoracic Aortic Aneurysm Repair, Anesthesiology, 2018, 128, 5, 1004.
H Awad, ME Ramadan, HF El Sayed, et al, Spinal cord injury after thoracic endovascular aortic aneurysm repair, Canadian Journal of Anesthesia, 2017, 64, 12, 1218–1235.
H Awad, ME Ramadan, E Tili, et al, Fluoroscopic-Guided Lumbar Spinal Drain Insertion for Thoracic Aortic Aneurysm Surgery, Anesthesia & Analgesia, 2017, 125, 4, 1219–1222.
H Awad, H Elgharably, PG Popovich, Role of induced hypothermia in thoracoabdominal aortic aneurysm surgery, Therapeutic Hypothermia and Temperature Management, 2012, 2, 119–37.
H Awad, DP Ankeny, Z Guan, et al, A mouse model of ischemic spinal cord injury with delayed paralysis caused by aortic cross-clamping, Anesthesiology, 2010, 113, 4, 880–91.

Want to republish our articles?
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence
(CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
More articles you may like
Prof Candis M. Morello – Prof Jan D. Hirsch | Recent innovations in pharmacy education
A pioneering research team from the Skaggs School of Pharmacy and Pharmaceutical Sciences, University of California, San Diego, United States, has been instrumental in developing innovative techniques for teaching pharmacy students. The Next Generation of Pharmacist Educators (NextGen-RxEd) programme is a new method of training the next generation of pharmacist educators and academics. To help pharmacists and pharmacy students visualise the complex issues experienced by their patients, the team led by Professors Candis Morello and Jan Hirsch developed an innovative educational tool, called the Medication Therapy Management (MTM) Spider Web.
Professor Michael Yarus | How RNA Started the Conversation That Built Life
The genetic code stores the instructions for building proteins, yet how it first arose remains unclear. It likely did not appear fully formed, but instead emerged step by step from simple chemical interactions. In this study, a team led by Professor Michael Yarus at the University of Colorado Boulder shows how early ribonucleic acid (RNA) molecules could bind specific amino acids, creating the first coding relationships. These early interactions are then refined through evolutionary processes such as duplication and merging of partial codes. By combining the experimental data with computer simulations, the work provides a testable pathway from prebiotic chemistry to the modern genetic code.
Identifying Nutritional Risk in Early Childhood: Insights from NutriSTEP®
Early childhood is a critical period for growth and development, yet many young children face nutritionrelated risks that can go unnoticed. Professor Janis Randall Simpson and colleagues have developed NutriSTEP®, validated and reliable screening tools that help identify potential nutritional concerns in toddlers and preschoolers. Their large-scale analysis of Canadian data reveals patterns in diet, behaviour, and food access that could help guide early interventions and support healthy development.
Professor Tian Yu Cao | Twistor Theory: A New Framework for Quantum Gravity
At Boston University, Professor Tian Yu Cao is rethinking the foundations of modern physics. His work builds on twistor theory which demonstrates that spacetime is secondarily derived from twistor constructions, but goes further to highlight the most important implication of the Penrose transform in that the primary physical agents can only be mathematically described by elements of cohomology with the defining feature having roots in spin. This view of primary agents combines with Cao’s other major claim that quantum behaviour itself may arise from the physical property of spin leads to a new consistent framework of quantum gravity in which long-standing puzzles in black holes (evaporations) and cosmology (transitions between cycles of cosmos) can be adequately addressed, with the crucial help from the on-going development of operator product expansion formular defined on twistor space.




