Dr I-Ming Chou – Building Containers for Extreme Geological Fluids
Earth’s crust plays host to many different fluids, which are often pressurised and heated to extreme degrees by the geological processes taking place around them. Harnessing his previous experience at the US Geological Survey, Dr I-Ming Chou at the Chinese Academy of Sciences designs vessels suitable for containing these fluids, while also enabling researchers to easily measure them in the lab using advanced optical techniques. Through their work, Dr Chou’s team has presented cutting-edge designs based on fused silica capillary tubes. Their designs could soon transform geologists’ understanding of the chemical processes taking place far below us.
Underground Fluids
Deep underneath the Earth’s surface, immense geological forces can elevate fluids trapped in the crust to extreme temperatures and pressures. Under the right conditions, we can see these superheated fluids as they are ejected all the way to the surface, in dramatic natural features such as geysers and hydrothermal vents on the seafloor.
By studying the unique properties and compositions of these fluids, geologists can learn much about the physical characteristics of the crust, and the chemical reactions that shape its composition. However, while these samples can be studied in their natural state by drilling holes deep into Earth’s crust, this is a costly and time-consuming process.
Instead, researchers today are aiming to recreate geological fluids in the lab directly. This poses a significant challenge: without highly advanced equipment capable of containing substances under such extreme pressure-temperature conditions, they will rapidly cool down and lose pressure. Dr I-Ming Chou at the Chinese Academy of Sciences aims to overcome this problem by developing cutting-edge new containers for measuring geological fluids. His team’s designs are now offering realistic new glimpses of the processes playing out far beneath our feet.
Investigations with Raman Spectroscopy
A key feature of geological fluids is the presence of both organic molecules such as methane (CH4), and inorganic substances such as hydrogen sulphide (H2S), contained within them. At extreme temperatures and pressures, these substances can dissolve and diffuse through water in characteristic ways, which are thought to play important roles in geological processes, as well as the chemical composition of Earth’s crust.
Researchers observe and study these unique processes through a technique named ‘Raman spectroscopy’. This method relies on the fact that the relative positions of a molecule’s constituent atoms are never static; rather, depending on the molecule’s composition, they will vibrate and rotate in characteristic ways.
When the molecule is exposed to the uniform photons produced by a laser, they will initially interact with these vibrations, either losing or gaining energy in the process. Afterwards, the photons are re-emitted with new energies and directions, in an overall process named ‘Raman scattering’. Crucially, the shifts in energy experienced by the photons are unique to the molecule they were scattered from.
So, by detecting these re-emitted photons, researchers can identify molecules by their specific Raman scattering fingerprints. However, even for the most advanced Raman spectroscopy techniques for studying geological fluids at elevated pressures and temperatures, suitable containers are still needed.

Earth’s interior structure, showing pockets of geological fluids.
Building Optical Cells
Until now, lab-based studies have largely relied on devices named ‘diamond anvil’ cells, which exploit the extreme hardness of diamond to compress samples to immense pressures. For fluids, however, this technique faces several shortcomings: including small sample sizes, an inability to directly measure or change sample pressures, and difficulties with loading gas samples onto the cells. In addition, diamond can reduce and interfere with any Raman signals passing through.
To perform Raman spectroscopy effectively, cells containing the fluids must be exceptionally robust – but must also incorporate transparent windows to let laser light in, and scattered photons out. Without extreme precautions, these windows would simply weaken the cells, allowing the pressurised, superheated liquids inside to break out.
In their earlier research, Dr Chou and his colleagues addressed the problem using a glass-like material named fused silica. Containing silicon dioxide (SiO2) arranged in irregular patterns, this material is both incredibly pure and transparent, and able to withstand considerable temperatures and pressures. Through subsequent work, the researchers have now developed two types of cells that incorporate fused silica, named the ‘high-pressure optical cell’ (HPOC) and the ‘fused silica capillary capsule’ (FSCC), finally allowing them to carry out Raman spectroscopy on geological fluids.
The High-pressure Optical Cell
The first device developed by Dr Chou’s team contained flexible capillary tubes with either square or round cross sections, each made from fused silica. The use of the square tube minimised any optical distortion, enabling better visual observations. Measuring 300 micrometres across, the tubes were sealed at one end, and connected to a high-pressure valve at the other.
To strengthen the tubes, the manufacturers coated them with a thin layer of heat-resistant polymer, a small section of which was removed by the researchers to provide a clear optical window. In previous studies, high-pressure capillary tube setups were inserted into a temperature-controlling device, with a glass cover for both temperature control and protection. In contrast, the team’s new structures were enclosed in a larger capillary tube of stainless steel, with a small part ground away to expose the optical window. The resulting HPOC has numerous advantages over any predecessors, including the diamond anvil cell.
Firstly, the HPOC allowed the researchers to directly load their sample fluids, and continuously monitor their temperatures and pressures in extended experiments. It also enabled them to perform Raman spectroscopy with very little attenuation or interference in the light passing through the transparent window – allowing for precise measurements of their energy shifts. The small volumes of the cells made them suitable for studying rarer samples, which are in limited supply, as well as poisonous samples, such as hydrogen sulphide and carbon monoxide.
Furthermore, pressures inside the capillary tube could be elevated to as high as 160 MPa – a thousand times larger than the pressure exerted by the atmosphere at Earth’s surface. Finally, the fused silica walls of the tube meant that the device as a whole was inexpensive, inert to sulphur, and highly permeable to hydrogen – paving the way for some advanced experimental capabilities, such as controlling the redox state of the sample by imposing hydrogen pressure externally through diffusion.
In their subsequent experiments, Dr Chou’s team prepared a sample of room-temperature water in the capillary tube, and then introduced a further sample of highly pressurised methane gas. In the hours that followed, the HPOC allowed them to directly observe how methane molecules passed across the gas-water interface, and diffused towards the end of the tube over time. Using Raman spectroscopy, they could then determine the diffusion rate of methane in water by measuring the concentrations of dissolved methane at different sample positions at various time. Further experiments explored other aspects of the interaction between water and organic molecules – including the solubility of methane hydrate.
From these results, Dr Chou and his colleagues showed how the capabilities of the HPOC could be extended to study processes involving larger organic molecules, including ethane, propane and butane – as well as salt solutions instead of pure water. In addition, they showed how Raman spectra could be collected across wide ranges of fixed pressures and temperatures. The successes of these early experiments have now set the stage for more complex analysis – involving fluids and mixtures that more closely resemble real geological environments.
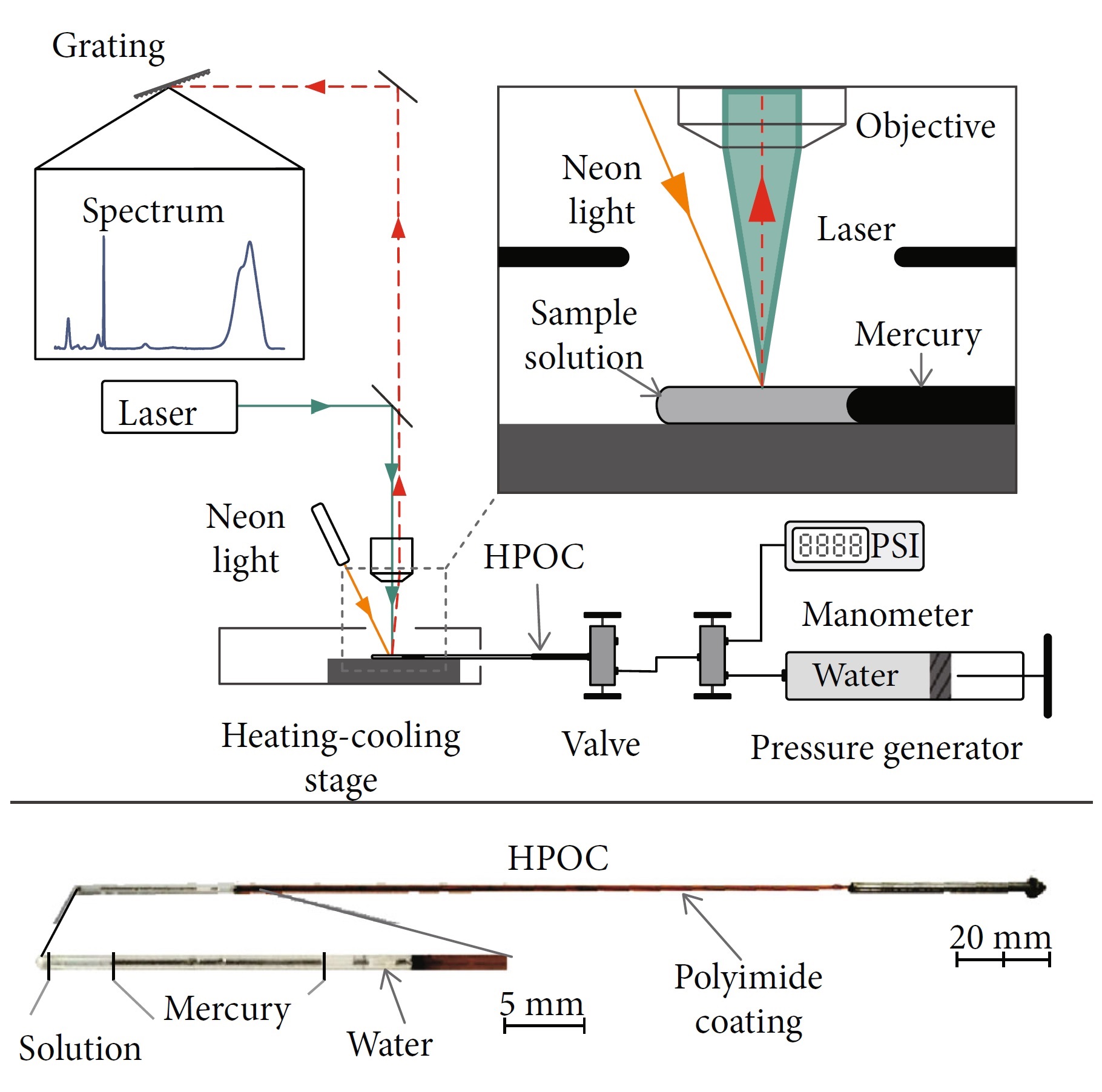
Diagram showing Dr Chou’s experimental setup. The lower part shows a prepared HPOC containing sample solution, mercury, and water as a pressure medium.
The Fused Silica Capillary Capsule
Alongside the HPOC, the second type of cell developed by Dr Chou’s team is designed for studying ‘fluid inclusions’. These microscopic bubbles of gas and liquid trapped inside crystals contain a variety of both organic and inorganic materials. They are now thought to play a key role in many geological processes – including the healing of fractured minerals, which requires fluid inclusions to be trapped in water. Previously, they could only be studied by synthesising fluid inclusions within fractures in minerals, such as quartz, garnet, fluorite, calcite, and halite. However, any subsequent experiments involving Raman spectroscopy were difficult to carry out at the pressures and temperatures required during synthesis.
In the researchers’ FSCC device, samples can be studied by first loading them into a fused silica capillary tube, which is 6 centimetres long and has one end sealed. Then the tube is centrifuged, forcing the samples to its closed end. Afterwards, the open end of the tube is connected to a vacuum pressure line, through which any air in the sample is evacuated. Gaseous samples are then added by immersing the sealed end of the tube in ultra-cold liquid nitrogen, and switching the pressure tube from a vacuum to a sample gas – which is allowed to either condense or freeze over the next several minutes. Finally, the tube is evacuated, and its open end is sealed while frozen and under vacuum.
This setup had several unique advantages for studying fluid inclusions, including the formation of large and uniform gas bubbles, which are still able to tolerate high internal pressures. In addition, since hydrogen can readily pass through the disordered molecular structure of fused silica, researchers can readily initiate useful redox reactions from outside the capsule, without otherwise disturbing the sample.
The further capabilities enabled by the team’s FSCC now open up a whole new range of intriguing geological experiments. Raman spectroscopy of fluid inclusions in the lab will be particularly useful for studying reactions between individual hydrocarbons, crude oils containing mixtures of hydrocarbons, and inorganic gases. One such process is ‘cracking’, where organic molecules containing long chains of carbon atoms break down into smaller chains when under thermal stress, altering the composition of organic material in Earth’s crust.
In the case of inorganic samples, FSCCs could be used to study the formation of ions containing uranium, within solutions of lithium chloride. This process is known to take place at extreme pressures in the crust, forming the ores that we use to source uranium for nuclear power stations – as studied by a team led by Jean Dubessy at the University of Lorraine in France. In addition, research headed by Dr Chou showed how hydrothermal circulation within the newly-formed oceanic crust is influenced by the separation of sulphate-bearing seawater – with important consequences for the transport of heat, water, and chemicals throughout Earth’s oceans and atmosphere.
Elsewhere, Dr Chou’s team showed that their capsules could be applied in calibration for thermocouples, which are a key element of the heating and cooling stages required for microthermometric and Raman spectroscopic analyses of fluid inclusions.
Improving Understanding of Geological Fluids
Over the past three decades, Raman spectroscopy has already contributed significantly to our understanding of the Earth’s composition, and the materials involved in its geological processes. Through the use of HPOCs and FSCCs, the capabilities of the technique could soon be enhanced further still, through better analysis of minerals, organic materials, silicate melts, and water-based solutions in the crust.
With transparent windows that are resilient to extreme temperatures and pressures, stable in highly acidic conditions, and inert to reactive substances such as sulphur, Dr Chou’s team hopes that their approach will soon enable experiments involving many different chemical systems, which more closely simulate the conditions present deep within Earth’s crust. With further improvements, it could even lead to cutting-edge simulations of extraterrestrial ocean waters, such as those on Europa or Enceladus.
Reference
https://doi.org/10.33548/SCIENTIA709
Meet the researcher

Dr I-Ming Chou
Institute of Deep-sea Science and Engineering
Chinese Academy of Sciences
Sanya
China
Dr I-Ming Chou completed his MS in Geology at National Taiwan University in 1968, and his PhD in Geochemistry at The John Hopkins University (JHU) in 1974. Following postdoctoral training at both JHU and NASA’s Johnson Space Centre, he became a Geologist at the US Geological Survey in 1979, where he worked for 33 years until 2013. Then, Dr Chou was appointed as Director of the Lab for Experimental Study under Deep-sea Extreme Conditions at the Chinese Academy of Science’s Institute of Deep-sea Science and Engineering in Sanya, China. His main research interests include the physicochemical properties of geological fluids, the stability of minerals at different temperatures and pressures, and the compositions of co-existing fluids. He is a Fellow of both the Mineralogical Society of America and Geological Society of America. In 2018, he organised and chaired the ‘Symposium on the combined applications of optical cells with controlled pressures and temperatures and in situ Raman spectroscopic analysis technique’, which took place in Sanya, China.
CONTACT
E: imchou@idsse.ac.cn
W: http://www.idsse.cas.cn/yjsgk2015/zzjg2015/yftx2015/shkxyjb2015/shjdhjmnyjsys2015
KEY COLLABORATORS
William A. Bassett, Cornell University, USA
Robert C. Burruss, US Geological Survey
Jill D. Pasteris & Alian Wang, Washington University, St. Louis, USA
Zhiyan Pan, Zhejiang University of Technology, Hangzhou, China
Wanjun Lu, China University of Geosciences, Wuhan, China
Jiankang Li & Yucai Song, Chinese Academy of Geosciences, Beijing, China
Shunda Yuan, China University of Geosciences, Beijing, China
Linbo Shang, Institute of Geochemistry, Chinese Academy of Sciences, Guiyang, China
Xiaolin Wang, Nanjing University, Nanjing, China
Xiaochun Xu, Hefei University of Technology
FUNDING
Energy and Mineral Programs of US Geological Survey, National Natural Science Foundation of China (Nos. 41573052 and 41973055), the Knowledge Innovation Program (SIDSSE-201302), the Hadal trench Research Program (XDB06060100), and the Key Frontier Science Program (QYZDY-SSW DQC008) of the Chinese Academy of Sciences.
FURTHER READING
IM Chou, AL Wang, Application of laser Raman micro-analyses to Earth and planetary materials, Journal of Asian Earth Sciences, 2017, 145, 309-333.
IM Chou, ZY Pan, Fused silica capillary reactor and its applications, In: Supercritical Water and its Applications in Biorefineries (Zhen Fang and Chunbao (Charles) Xu editors), Springer, 2014, Chapter 6, 157-178.
IM Chou, Optical cells with fused silica windows for the study of geological fluids. In: Applications of Raman Spectroscopy to Earth Sciences and Cultural Heritage (J. Dubessy and F. Rull, editors), 2012, Chapter 6 in EMU Notes in Mineralogy, 12, 227-247.
IM Chou, YC Song, RC Burruss, A new method for synthesizing fluid inclusions in fused silica capillary containing organic and inorganic material, Geochimica et Cosmochimica Acta, 2008, 72, 5217-5231.
IM Chou, RC Burruss, WJ Lu, A new optical cell for spectroscopic studies of geologic fluids at pressures up to 100 MPa, Advances in High-Pressure Technology for Geophysical Applications, Elsevier, 2005, Chapter 24, 475-485.

Want to republish our articles?
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence
(CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
More articles you may like
Jella van de Laak | Could Tumour- Targeting Bacteria Enhance Cancer Immunotherapy?
Cancer immunotherapy has transformed treatment outcomes, yet many patients still fail to respond. At Maastricht University, Jella van de Laak, working with Professor Ludwig Dubois and Professor Jan Theys, is exploring how tumour-targeting bacteria could improve immune responses and expand treatment success.
Distinguished Professor Michael Zhdanov | Mapping Magma and Drilling for Oil: New Methods for Geophysical Modelling
Geophysicists use a variety of different methods to peer beneath the Earth’s surface. Seismic activity, gravitational fields , and magnetic fields each offer their own windows into the world underground, but, in isolation, are incapable of giving us the full picture. Combining data from distinct geophysical surveys, however, is its own challenge. For a number of years, a team of researchers led by Professor Michael Zhdanov has worked to develop a mathematical framework capable of generating detailed geophysical models from multiphysics data.
Through application to modelling magma chambers underneath Yellowstone and searching for oil deposits in the Barents Sea, they demonstrate that their approach can produce robust and accurate predictions
Professor Deanna Sellnow | How Strategic Communication Can Save Lives When Crisis Hits
Professor Deanna Sellnow from Clemson University argues that crisis and risk communication research must evolve beyond simply describing and deconstructing problems to actively empowering people protect themselves and their communities. In an era of increasing globalisation, she proposes that researchers adopt learner- and learning-centered approaches to ensure communication does not simply inform people about dangers, but also teaches them how to respond effectively (e.g., to reduce risks, mitigate harms, improve safety, save lives) and why.
Dr David Berry | Bridging the gap between Agriculture and Automation
As automation and controlled environments reshape modern farming, the need for technicians who understand both plant science and engineering is rapidly increasing. At Virginia Western Community College (VWCC) located in Roanoke, VA, Dr David Berry is leading a programme supported by the National Science Foundation (NSF) with the aim to merge agriculture with mechatronics, preparing students for the realities of emerging industries. A distinctive example of this approach is an automated hydroponics system designed by two industry-sponsored students, demonstrating how practical experience, interdisciplinary training and strong employer partnerships can create the skilled workforce required for the future of sustainable food production.




