Dr Jack Jallo – Improving Patient Outcomes in Traumatic Brain Injury
Severe traumatic brain injury (TBI) is associated with high rates of disability and even mortality. Understanding the relationship between patient outcomes and the treatment received, as well as other physiological factors such as inflammation, can improve how we approach TBI. Dr Jack Jallo and his team from the Department of Neurological Surgery at Thomas Jefferson University are researching the factors that influence TBI recovery to help design better care management protocols and optimise patient recovery.
Traumatic Brain Injury: Mechanisms of Damage
Globally, around 10 million people are affected by traumatic brain injury (TBI) each year. Treatment varies based on the severity of injury, but success is often reliant, at least in part, on managing sequelae of injury. This is because, while the immediate injury puts patients at risk of death or disability, it is often the subsequent progression of damage that is most dangerous. When the brain undergoes such trauma, it responds by triggering a cascade of inflammation in an attempt to repair the damage and provide vital nutrients and oxygen to cells. Unfortunately, this inflammation can become uncontrolled and may play a significant role in secondary injury, contributing to cell death and further dysfunction.
Inflammation can cause swelling or oedema which increases the pressure in the skull, creating what is known as intracranial pressure (ICP). The brain is extremely sensitive to this change, and ICP over 20mmHg is the most frequent cause of death in TBI patients. While inflammation is initiated by the body in an attempt to increase blood flow to the damaged tissue, uncontrolled inflammation can increase ICP and reduce cerebral blood-flow, complicating outcomes. When this occurs, treatment primarily involves neurosurgical intervention and the removal of a section of the skull to relieve the pressure, a procedure that comes with risks of its own.
Dr Jack Jallo is a neurosurgeon from Thomas Jefferson University (TJU), who is dedicated to ascertaining the factors that affect patient outcomes from TBI and establishing how treatment can be improved. To identify novel therapeutics for TBI, he has explored the effects of tissue oxygenation following injury, craniotomy and surgical intervention, as well as the role of the cannabinoid system in inflammation.
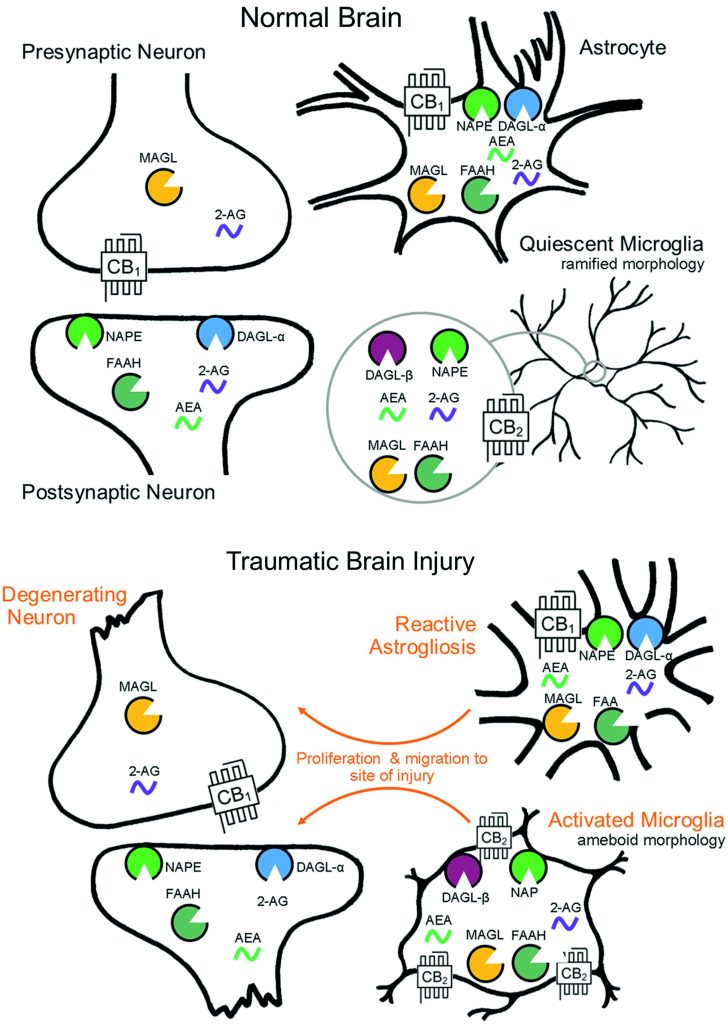
Reproduced from LD Schurman, AH Lichtman, Endocannabinoids: A Promising Impact for Traumatic Brain Injury, Frontiers in Pharmacology, 2017, 8, 69.
Care Related Patient Outcomes
As the Director of Neurotrauma and Critical Care at TJU, Dr Jallo has years of experience working in a level one adult trauma centre. His observations of trauma care and TBI have informed his key papers detailing the different outcomes among patients depending on their level of treatment.
When quantifying these differences, it is essential to take into account that there may be variances in treatment between centres. Comparatively, level one trauma centres are capable of dealing with a much higher volume of patients and tend to have more specialised and interdisciplinary teams, while level two centres are slightly less specialised. Studies have shown that, on the whole, severely injured patients have an increased survival rate when treated at level one trauma centres. Dr Jallo’s research also confirms that for TBI, patient outcomes are generally better when they receive specialised and directed care, such as craniotomy, in a level one centre.
When dealing with TBI, a mainstay of trauma centres since the 1960s has been the use of ICP monitoring. The idea is, that by keeping tabs on a patient’s ICP using intracranial devices we can better predict which patients may need surgery. This could be useful in cases where brain swelling is delayed following injury or occurs silently, allowing doctors to perform risky neurosurgery only when necessary.
However, when publications came out questioning the benefit of ICP monitoring in severe TBI, hospitals observed a decrease in the use of this technique. In a recent paper, Dr Jallo and his colleagues are hesitant to denounce the practice, and instead, recognise significant limitations, such as small sample size, associated with the previous publications. It remains clear that, as a time-sensitive condition, TBI treatment would likely benefit from ICP monitoring that allows for more personalised care, and Dr Jallo emphasises the need for more research into its use.
In a further trial on monitoring patient progression, Dr Jallo noted that brain tissue oxygenation also associated quite well with outcomes. It was found that combining oxygen monitoring with ICP monitoring improved management of severe TBI and reduced the amount of brain tissue which was starved of oxygen. A hugely important aspect of TBI care is preventing secondary damage from occurring, being able to identify when that might happen is a significant advantage.
Dr Jallo’s work has shown that it is essential to take into account the variation in care that patients receive so that we can untangle the downstream effects from physiological differences relating to the injury itself. Furthermore, the way we define and categorise TBI also has implications for patients down the line.
Defining Injury
The majority of emergency room visits for head injury are classified as mild TBI (mTBI). These cases generally have higher rates of recovery, but for some mTBI patients, chronic cognitive or neurological deficits can persist. Can these cases really be described as ‘mild’? Following the universally approved definition outlined in the American Congress of Rehabilitation Medicine, mTBI is diagnosed only if the loss of consciousness is for less than half an hour, and in general, post-trauma amnesia (PTA) should resolve within 24 hours. Nevertheless, there is room for significant variation within the mTBI group.
Clinicians have noticed that a subset of mTBI patients shows significantly worse outcomes 6 months post-injury. Researchers, therefore, coined the term complicated mild TBI (cmTBI) to describe cases where patients may have more extensive damage, such as lesions in the brain, which can cause this. Categorising patients presents a problem for treatment as it may not fully appreciate the nuances of injuries between individuals, meaning that the right level of care cannot be applied.
In an observational study of cmTBI patients, Dr Jallo noticed that only 25% of the sample had PTA for less than 24 hours. In nearly half of patients, it was present up to seven days, and for a third of patients, it was reported for more than a week. The duration of PTA seems to correlate more clearly with performance on cognitive tests, and Dr Jallo believes it should be used alongside other measures when defining TBI. This may help refine outcome predictions and improve rehabilitation treatments offered, but the limitations of the study should be noted, such as the lack of a control group. Further research into the use of PTA duration as a possible diagnostic factor is essential to help advance our understanding of TBI.

Targeting the Endocannabinoid system
Developing new treatments for TBI has been a challenge for a long time. After initial surgical attempts to prevent brain swelling, it becomes difficult to accurately target and prevent secondary injury from occurring. Novel immunomodulatory therapies have been identified which target the inflammatory response, preventing it from going into overdrive and suffocating the brain as a by-product.
The cannabinoid system, which functions to communicate certain signals in the brain and body, has been shown to play a significant role in inflammation. A particular receptor known as CB2R is present on immune cells in the brain and can help to regulate inflammation and the response to injury. Using molecules called agonists which bind to and stimulate CB2R, we can increase this anti-inflammatory effect, which may be helpful in treating TBI.
Dr Jallo and colleagues used mice to show that early administration of a CB2R agonist after TBI can reduce the expression of markers associated with inflammation. This may be due to the ability of CB2R to prevent immune cells from entering the bloodstream of the brain. Developing safe and effective drugs to do this would increase the number of tools we have available to treat TBI and hopefully reduce secondary injury.
Interestingly, one of the long-term outcomes of TBI, chronic headache, may be associated with disruptions in the cannabinoid system. Alterations in the sensitivity of neurons in regions associated with headache may be due to disturbances in the balance of chemical transmitters, such as cannabinoids. Modulating this with exogenous cannabinoids, such as THC and CBD, has been shown to improve post-traumatic headache as well as other conditions like migraine. Further research into this could support the push for legalisation of cannabis, which has been shown to have a multitude of benefits for many health conditions.
The Future of TBI Research
Dr Jallo’s work into the causes of and treatments for TBI is vitally important if we want to improve outcomes for patients. His position at the interface of clinical medicine and scientific research gives him a unique perspective on how the care we administer can affect clinical outcomes, as well as the physiological mechanisms associated with TBI.
Finding novel ways to treat injuries of the brain requires a deep understanding of the endogenous systems involved. It is common with brain diseases that the body’s attempts to heal the damage can end up doing more harm than good. Unravelling the threads that influence this balance can help us target them for more effectively and identify new tools to treat TBI.
Reference
https://doi.org/10.33548/SCIENTIA619
Meet the researcher

Dr Jack Jallo
Department of Neurological Surgery
Thomas Jefferson University
Philadelphia, PA
USA
Dr Jack Jallo is a neurosurgeon at Thomas Jefferson University who specialises in the evaluation and treatment of cervical, thoracic, and lumbar spine injuries, as well as spinal tumours. He earned his MD from George Washington University in 1990 and completed his PhD in physiology at Temple University Hospital in 2003. He completed a neurosurgical residency at Temple University as well as a fellowship in neurotrauma and critical care. In addition to being a neurosurgeon, Dr Jallo is also currently a professor at Thomas Jefferson University and the executive director for critical care. His research explores methods to improve outcomes from traumatic brain injury. He has authored a number of articles and textbooks with the goal of educating patients and surgeons. He is on the board of both the Pennsylvania Emergency Health Services Council and the Pennsylvania Trauma Systems Foundation and since 2017 has been a member of the American College of Surgeons Committee on Trauma as well as the Joint Council on Neurotrauma and Critical Care.
CONTACT
E: jack.jallo@jefferson.edu
W: https://hospitals.jefferson.edu/find-a-doctor/j/jallo-jack.html
Twitter: @JackJalloMD
LinkedIn: https://www.linkedin.com/in/jack-jallo-b2b72a1b8/
KEY COLLABORATORS
Melanie Elliot, PhD
FUNDING
080-20000-Z90701 Sponsor: NIH through the University of Pittsburgh Short Title: BOOST-3.
09/2014–08/2017: Central Pain Mechanisms and Novel Therapeutic Strategies in a Model of Closed Head Injury through the Department of Defense. The goal of this project is to study potential biomarkers, therapeutic interventions, and pathological mechanisms of chronic migraine headache in a model of concussion. Grant #: W81XWH-14-1-0594 (MR130514). Role: Co-Investigator.
9/01/12–8/31/2015: Investigator-Initiated Research Award Department of Defense PR 110398 to M. Elliott. U.S. Army Medical Research & Materiel Command through Thomas Jefferson Medical College: Trigeminal Sensitization in a Pre-clinical Model of TBI: Implications for Post-Traumatic Headache. Using a traumatic brain injury (TBI) animal model of controlled cortical impact (CCI), the investigators propose to elucidate mechanisms of central sensitisation underlying post-traumatic headache (PTH) disorders. They hypothesize that increased levels of two crucial molecular mediators – calcitonin gene-related peptide (CGRP) and inducible nitric oxide synthase (iNOS) – following TBI contribute to sensitisation of the trigeminovascular system. The well-defined experimental design is aimed at determining the role of endogenous and exogenous CGRP and iNOS in driving sensitisation in the acute and post-traumatic period. Amount: $1,095,000. Role: Collaborating Investigator
7/01/10-06/30/11: NIH through Emroy University University of Michigan through Temple University Neurological Emergencies Treatment Trials (NETT) – ProTECT III. The major goal of this project is to determine the efficiency of administering intravenous (IV) progesterone (initiated within 4 hours of injury and administered for 72 hours, followed by an additional 24 hours taper) versus placebo for treating victims of moderate to severe acute TBI (Glasgow Coma Scale score 12–4). Role: Principal Investigator.
Further Reading
N Chalouhi, N Mouchtouris, F Al Saiegh, et al., Comparison of Outcomes in Level I vs Level II Trauma Centers in Patients Undergoing Craniotomy or Craniectomy for Severe Traumatic Brain Injury, Neurosurgery, 2020, 86, 107–111.
DO Okonkwo, LA Shutter, C Moore, et al., Brain Tissue Oxygen Monitoring and Management in Severe Traumatic Brain Injury (BOOST-II): A Phase II Randomized Trial, Critical Care Medicine, 2017, 45(11), 1907–1914.
MB Elliott, SJ Ward, ME Abood, et al., Understanding the endocannabinoid system as a modulator of the trigeminal pain response to concussion, Concussion, 2017, 2(4), CNC49.
T Hart, TA Novack, N Temkin, et al., Duration of Post-Traumatic Amnesia Predicts Neuropsychological and Global Outcome in Complicated Mild Traumatic Brain Injury, Journal of Head Trauma Rehabilitation, 2016, 31(6), E1–E9.
PS Amenta, JI Jallo, RF Tuma, et al., Cannabinoid receptor type-2 stimulation, blockade, and deletion alter the vascular inflammatory responses to traumatic brain injury, Journal of Neuroinflammation, 2014, 11, 191.

Want to republish our articles?
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence
(CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
More articles you may like
Alex Spezowka | Responsible Research Writing in the Age of AI: From Detection to Transparency
Artificial intelligence is often discussed as a future challenge for research, yet it is already shaping how many papers are written. Drawing on emerging evidence and recent analysis, work led by Alex Spezowka highlights a key shift in thinking: rather than trying to detect AI use, the focus is moving towards how it can be used responsibly. This has important implications for how research is produced, evaluated, and trusted.
Laura Daniela Muñoz Duarte – Jo Philips | Rethinking Hydrogen Use in Acetogenic Bacteria: Kinetics, Thresholds, and Biotechnological Potential
Acetogenic bacteria can convert carbon dioxide and hydrogen into useful products, making them promising tools for sustainable biotechnology. Research led by Laura Muñoz and Jo Philips reveals that hydrogen consumption follows a simpler kinetic pattern than previously assumed, while also uncovering major differences between species in how efficiently they operate at low hydrogen levels. Together, these findings provide a clearer framework for selecting and optimising acetogens for industrial applications.
Dr Lynn George | A New Link Between Elongator Dysfunction and TDP-43 Loss in ALS
At Montana State University Billings, Dr Lynn George and her team are investigating the cellular mechanisms underlying amyotrophic lateral sclerosis (ALS). Using a new mouse model, their research reveals that disruption of the Elongator complex leads to nucleolar stress and the loss of the protein TDP-43 from motor neurons. These findings connect two major hallmarks of ALS and offer new insight into how the disease develops.
Dr David Hansen – Abhishek Juneja | Coming of Age: Active Balancing in Adolescence
Adolescence, the transitional stage between childhood and adulthood, comes with challenges as well opportunities for growth and learning.




