Dr John T. Seykora – Promising New Targeted Therapies in the Treatment of Skin Cancer
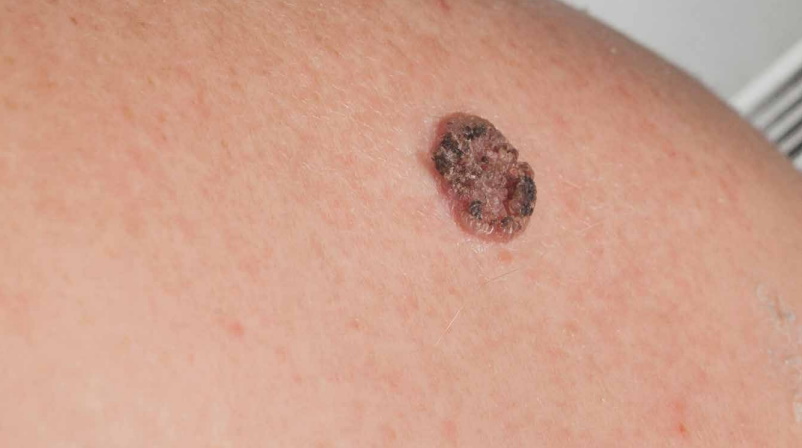
Every year, approximately 700,000 new diagnoses of squamous cell carcinoma, a type of skin cancer, are reported to dermatologists in the USA alone, constituting the second most common form of cancer. Treating pre-cancerous skin cells with a therapeutic topical cream could significantly reduce the number of harmful cells, potentially inhibiting the growth of squamous cell carcinoma. Dr John T. Seykora and his team from the University of Philadelphia have been researching the biology of skin squamous cell carcinoma and related precursor lesions to identify new therapeutic targets that could be treated using topical approaches with exciting results.
Squamous Cell Carcinoma: A Growing Problem
In our ageing yet active population, skin cancers such as squamous cell carcinoma and associated precancerous lesions are becoming increasingly common, and the need for new therapeutic strategies becomes more pressing by the day. Squamous cell carcinoma in situ is a common pre-cancerous skin condition that can progress to the more serious disease of cutaneous squamous cell carcinoma. Although the former is frequently seen by dermatologists, our understanding of the biological mechanisms behind the condition remains incomplete.
Understanding human skin cancer development requires scientific studies that elucidate the molecular pathways leading to pre-cancerous lesion formation and subsequent tumour development and disease progression. As such, laboratory mice have proven to be invaluable resources as models in this work. Currently, Dr John T. Seykora and his team from the University of Philadelphia are leading the scientific effort through their experiments on genetically engineered murine models and in-depth characterisation of human samples.
Dr Seykora has observed the clinical and molecular factors relating to skin cancers to define the progression from unremarkable epidermis to in situ lesions to cutaneous squamous cell carcinoma by utilising a variety of experimental approaches. Furthermore, insights gained from computer modelling of these lesions have identified multiple targets for treating pre-cancerous lesions.

The Development of Cancer
As is the case with other cancers, cutaneous squamous cell carcinomas exhibit a high rate of DNA mutations. The mechanisms that lead to this high frequency of DNA mutations likely result from tumour-suppressor proteins and proto-oncogenes being damaged via exposure to ultraviolet (UV) light. The UV-induced DNA mutations responsible for producing the in situ lesions in human skin were demonstrated in a 2021 study by Dr Seykora’s group on human skin specimens.
Since these initial observations of ultraviolet-induced protein mutations, other studies have confirmed the presence of mutations in significant numbers of squamous cell carcinoma patients. Previous studies by other researchers had presented a high prevalence of anti-cancer mutations in sun-exposed skin in comparison to non–sun-exposed skin, setting the stage for Dr Seykora’s own studies in this field. Consistent with these findings, Dr Seykora’s team found that 60% of patients displaying pre-cancerous squamous cell carcinoma in situ had similar anti-cancer protein mutations, indicating that the loss in these useful proteins in skin cells occurs before full cancer can develop.
Although laboratory mice are a strong, versatile model for studying skin cancer development, the key differences in skin structure between mice and humans limit any direct correlations between mice-based skin cancer studies and human disease. Additionally, there are major differences in both biochemical and genetic factors. Therefore, the correlation of observations made in mice models with those present in human skin tissue is vital for demonstrating relevance to human disease.
Multiple epidemiologic and basic research studies, including a 2019 study by Dr Seykora and his research team, have shown that cutaneous squamous cell carcinoma can also come about via chronic exposure to a systemic anti-fungal agent called Voriconazole, especially if the patient’s skin has been extensively exposed to UV radiation.
These observations suggest that Voriconazole promotes some sort of biochemical reaction in skin cells that makes them more vulnerable to UV irradiation, ultimately leading to cancerous tumour formation. Since these antifungal treatments have been found to produce cutaneous squamous cell carcinomas in UV‐exposed skin, these compounds likely promote additional DNA and cellular damage provided by UV radiation to accelerate the formation of tumours. The mechanism responsible for this process, however, was unclear until Dr Seykora’s group showed that Voriconazole directly inhibits the enzyme catalase which is a key detoxifying protein in cells that limits UV-induced damage and DNA mutations.
Research into the basic molecular mechanisms of skin cancers is the foundation for testing new therapies that are critical in combatting the disease. Back in 2002, Dr Seykora and his research team discovered that a novel cancer-preventing protein acts as a vital marker in understanding whether or not skin cells may be potentially predisposed to developing skin cancer. These studies identified a class of anti-oncogenes that target activated tyrosine kinases for degradation; this protein was named Src Activating and Signaling Molecule (Srcasm). Srcasm levels are inversely proportional to the rate of cell proliferation in cSCC and in situ lesions and Src tyrosine kinases, the target of Srcasm, are therapeutic targets for cSCC and related precancerous lesions. Therefore, molecules that enhance Srcasm function also may represent a new class of therapeutic agents to treat these lesions.
‘These innovations represent a promising and much-needed alternative to today’s topical therapies, one that directly targets the biological mechanisms driving the tumour’s growth…’
Future Potential Treatment Options
Current treatments methods for in situ and cutaneous squamous cell carcinomas are limited and invasive. Surgery is often recommended to remove squamous cell carcinomas, particularly those classified as high risk. Surgical removal involves injecting a local anaesthetic and removing the tumour from the skin along with a clear ‘safety margin’ to ensure that all of the cancer cells have been removed.
Moh’s surgery is a commonly used technique, involving the surgeon removing skin tissue layer by layer, mapping and freezing each layer, and examining the tissue for tumour cells under a microscope before proceeding to the next layer. This method is complicated and time-consuming but ensures that the entire tumour is removed with as little scarring as possible. Other options include radiation therapy, which takes a long time and can be painful, and other surgical approaches. A treatment method that does not cause pain or irritation, is non-invasive as well as cost-effective, could prove hugely beneficial for patients suffering from this type of skin cancer.
Since cutaneous squamous cell carcinomas typically arise from less-developed squamous cell carcinomas in situ, effective treatment of these affected ‘precursor’ skin lesions could potentially decrease the number of fully-developed cancer patients. Current treatments for these precursor skin conditions include cryotherapy (freezing the skin) or minor surgery, which can lead to skin pigmentation and scarring – as well as not being cost-effective. Currently available topical therapies for this stage include Imiquimod and Fluorouracil creams.
Fluorouracil and Imiquimod produce irritating, often painful inflammation, which discourages patients from completing the full course of treatment, although other topical treatment options are proven to have suboptimal pre-cancerous cell reduction rates or are very expensive for the patient or medical service provider. There remains a clear medical need for effective topical therapies that lack these serious and adverse side effects.
In 2008, Dr Seykora’s group published that Src-family tyrosine kinases are hyperactivated in human actinic keratosis, in situ carcinoma and squamous cell carcinoma. This study indicated that Src kinases are a therapeutic target for treating these lesions. Based on these studies, Dr Seykora’s group derived in vivo murine models of these lesions and tested the potential utility of topically applied kinase inhibitors to treat cutaneous squamous cell carcinomas and related precursor lesions.

A recent study of the kinase inhibitor Dasatinib shows that topical application promoted shrinkage of cancerous tumours in a similar way to topical Fluorouracil cream, but caused less painful inflammation and produced no skin ulcers. Dasatinib was applied daily to experimental mice, and was found to cause 45% and 77% reduction of cutaneous squamous cell carcinoma cells after two to five weeks of treatment, respectively.
Fluorouracil cream was also tested in this study and was found to induce a 70% regression in skin cancer cells after two weeks. However, it was found to cause skin ulcers in two out of the 15 observed tumours, and seven of the eight mice in that group died. No ulcers or deaths were observed in the Dasatinib topical treatment group. Dr Seykora, when asked in a recent interview, stated that ‘Together, these data suggest that topical application of small-molecule kinase inhibitors may be useful for treating cutaneous squamous cell carcinoma and related precursor lesions.’
In parallel experiments that were also undertaken by Dr Seykora’s research team, another topical skin cancer treatment that works in a similar way to Dasatinib, known as Dactolisib, was found to reduce the size of cutaneous squamous cell carcinomas in comparison to the currently available treatments.
Taken together, these results suggest that the topical application of new treatments targeting the proteins associated with cancer cell formation have the potential to treat cutaneous squamous cell carcinomas and other related skin conditions, and raise the possibility that topical treatments containing multiple cancer-associated protein (enzyme) inhibitors together could be more effective than utilising a single compound.
‘These innovations represent a promising and much-needed alternative to today’s topical therapies, one that directly targets the biological mechanisms driving the tumour’s growth,’ explains Dr Seykora, ‘with the number of cases of cutaneous squamous cell carcinoma steadily rising, it’s important we work to identify safer, more precise therapies to treat these cancers before they progress and spread. We believe topical kinase inhibitors are potential candidates that warrant further study.’
Reference
https://doi.org/10.33548/SCIENTIA712
Meet the researcher

Dr John T. Seykora
Perelman School of Medicine
University of Pennsylvania
Pennsylvania
USA
Dr John T. Seykora is a tenured Professor of Dermatology in the Perelman School of Medicine at the University of Pennsylvania. He received his undergraduate degree in Biology with Honors and concurrently was awarded a Masters in Biochemistry from the University of Chicago. Dr Seykora received his PhD from Rockefeller University, and then an MD qualification from Cornell University Medical College. After undertaking residency and fellowship at the University of Pennsylvania, Dr Seykora began teaching and performing research in the School of Medicine at the University of Pennsylvania, and later went on to become a tenured Professor of Dermatology, a position he still holds today. From being a National Merit Scholar at the University of Chicago in 1982 to receiving the Dean’s Award for Excellence in Basic Science Teaching from the Perelman School of Medicine in 2019, Dr Seykora has received an abundance of awards and honours throughout his career.
CONTACT
E: seykora@pennmedicine.upenn.edu
W: https://www.med.upenn.edu/apps/faculty/index.php/g275/p8968
FUNDING
R01AR051380, RO1-CA165836, RO1-ES02811, and P30-AR069589 National Institutes of Health grant P30-AR069589
FURTHER READING
Q Zheng, B Capell, V Parekh, et al., Whole exome and transcriptome analysis of UV-exposed epidermis and carcinoma in situ reveals early drivers of carcinogenesis, Journal of Investigative Dermatology, 2020, 141(2), 295–307.
V Lee, MD Gober, H Bashir, et al., Voriconazole enhances UV-induced DNA damage by inhibiting catalase and promoting oxidative stress, Experimental Dermatology, 2020, 29(1), 29–38.
X Yang, AEM Daifallah, S Shankar, et al., Topical kinase inhibitors induce regression of cutaneous squamous cell carcinoma, Experimental Dermatology, 2019, 28(5), 609–613.
L Zhao, W Li, C Marshall, et al., Srcasm inhibits Fyn-induced cutaneous carcinogenesis with modulation of Notch 1 and p53, Cancer Research, 2009, 69, 9439–9447.
E Ayli, W Li, T Brown, et al., Activation of Src-family tyrosine kinases in epidermal hyperproliferative disorders, Journal of Cutaneous Pathology, 2008, 35, 273–7.

Want to republish our articles?
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence
(CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
More articles you may like
Grandmothers: Innovation Through Tradition
Grandmother Project – Change through Culture (GMP) is an organisation dedicated to documenting the role of grandmothers and demonstrating the effectiveness of grandmother-inclusive strategies in improving the health and well-being of women, children, and adolescents. GMP’s groundbreaking work challenges conventional wisdom to transform community-based interventions in Africa and beyond, harnessing a powerful but often overlooked resource: the wisdom and influence of grandmothers.
Dr Robert Larkin | Cultivating Change to Improve Soil Health and Increase Potato Yield
Environmental quality and food production are facing the pressing challenges of climate change and global population growth. Dr Robert Larkin from the United States Department of Agriculture-Agricultural Research Service (USDA-ARS) and a team of plant scientists developed and tested a range of crop management systems to help overcome these compounding challenges. Their work is improving soil health and increasing the yield of potato crops, contributing to the future food security of nations.
Professor Giorgio Buttazzo | Artificial Intelligence and a Crossroads for Humanity
Where do we stand with artificial intelligence? Might machines take over our jobs? Can machines become conscious? Might we be harmed by robots? What is the future of humanity? Professor Giorgio Buttazzo of Scuola Superiore Sant’Anna is an expert in artificial intelligence and neural networks. In a recent publication, he provides considered insights into some of the most pressing questions surrounding artificial intelligence and humanity.
Dr Ralf Adam | New Technologies Shaping the Future of Oral Hygiene
Understanding the efficiency of various toothbrush technologies is essential for achieving optimal oral health. Dr Ralf Adam, who leads a dedicated team at Procter & Gamble in Germany, is keen to investigate the complexities of these technologies. His team have provided new insights into the best toothbrush types for plaque removal and the maintenance of gum health. By highlighting the importance of informed oral care decisions and ongoing investigations, this vital research works towards ensuring everyone can achieve a brighter, healthier smile.




