Dr Peter Bretscher – A Substantiated Framework for the Prevention and Treatment of Immune System-Related Diseases
Dr Bretscher has articulated his views, as expressed here, in a fuller manner that is accessible to the layperson and the interested immunologist, in a document called Spiral Immunology. This consists of a 1-, a 10- and a 20-pager, designed to be read successively. The full text is available on his website.
Immunity is generated normally against invaders, such as viruses and cancer cells, but not against parts of the body to which the immune system belongs. In 1970, Dr Peter Bretscher and Dr Melvin Cohn proposed a theory to account for how this is achieved. Importantly, immune responses against invaders can take one of two main forms, and Dr Bretscher (currently at the University of Saskatchewan) also proposed an explanation for how the choice of immunity is made. These two proposals are supported by diverse findings. Here, we outline and justify these proposals and explain how they lead to strategies to prevent and treat diverse diseases.
The Clonal Selection Theory
Animals can make an astronomical number of chemically distinct antibodies, produced by the body’s immune system when foreign substances (known as foreign or non-self-antigens) are detected. But how can the body produce so many diverse antibodies? The Clonal Selection Theory, formulated in the 1950s and 1960s, addressed this and a second question: How does the body normally manage to respond against foreign but not against self-antigens? Unfortunately, this attribute of self-nonself discrimination can occasionally fail, leading to immunity against self-antigens. This occurs, for example, in autoimmune diabetes.
According to the Clonal Selection Theory, one cell can produce only one chemically unique antibody. The cell bears this antibody on its surface as a receptor for antigen. As the body can produce so many different antibodies, the cell with the potential to produce an antibody specific for a particular antigen (an antibody precursor cell) is scarce indeed. According to Clonal Selection Theory, an antigen can select this cell by binding its antibody receptor, causing it to multiply over days to form a clone of cells, some of which secrete copious amounts of antibody. This theory explains why it takes days to produce significant antibody and how scarce cells, through cellular multiplication, can produce substantial amounts of antibody.
These antibody precursor cells belong to a class of white blood cells called lymphocytes, on the basis of their microscopic characteristics. Antibody precursor cells are generated in a particular organ. If they interact with an antigen, as generated, they die; if not, they give rise to mature antibody precursor cells. Most self-antigens are present in this organ. The lymphocytes specific for the self-antigens present in the organ are thus obliterated. This process is called central tolerance, resulting in a population of lymphocytes primarily specific for foreign antigens.

A Model for the Activation/Inactivation of Mature Lymphocytes
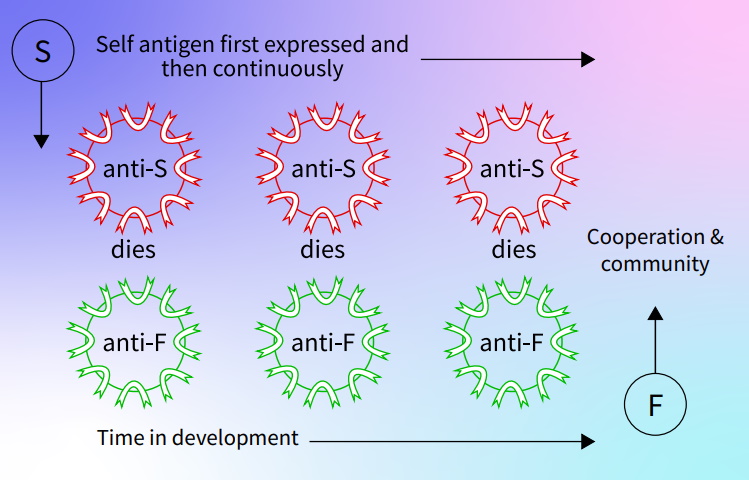
Lymphocytes are the cells of the body that can generate immune responses. While most anti-self lymphocytes are eliminated by central tolerance, some self-antigens, such as insulin, are not sufficiently present in the organ where lymphocytes are generated, to obliterate their lymphocytes.
Mature lymphocytes, specific for peripheral antigens such as insulin, a target self-antigen in autoimmune diabetes, also exist. Immunologists realised in the 1960s that antigens can interact with mature lymphocytes in two ways, one leading to their activation and immunity, the other to their inactivation, i.e., foreign and peripheral self-antigens would, respectively, activate and inactivate their corresponding lymphocytes in the best of circumstances. What determines ‘To Be or Not To Be?’
Dr Peter Bretscher (currently at the University of Saskatchewan) and Dr Cohn proposed in 1970 how these two ways of interaction differ: an antigen inactivates a single lymphocyte, whereas its activation requires its antigen-mediated interaction with other, ‘helper’ lymphocytes. These proposals explained many observations. In addition, they explain how peripheral antigens, such as insulin, normally inactivate their mature lymphocytes. Insulin, being a peripheral self-antigen, is present when the first insulin-specific lymphocyte is generated. This loner lymphocyte is therefore inactivated. Further insulin-specific lymphocytes (see Figure above) are obliterated as generated, either one or a few at a time. Lymphocytes specific for a foreign antigen accumulate in its absence. When this antigen impinges upon the body, it mediates the lymphocyte interactions required to generate an immune response against the foreign antigen.
These ideas led to the Two Signal Model of Lymphocyte Activation. The interaction of an antigen with a lymphocyte’s receptors generates signal 1, leading to its inactivation, unless helper lymphocytes recognise the antigen and deliver signal 2, leading to its activation.
The Two Signal Model also explains how autoimmunity arises. For example, infection by bacteria known as group A streptococci can result in autoimmunity against heart tissue. This is because the bacteria are foreign but also contain some structures similar to a peripheral heart antigen. Thus, scarce lymphocytes specific for these similar structures can be activated by the bacteria but would normally be inactivated by the heart tissue!
If To Be, What To Be?
There are three types of lymphocytes. B cells, generated in the bone marrow, multiply when activated by an antigen and some of their progeny secrete antibody. The CD4 T and CD8 T cells are generated in the thymus gland. The CD8 T cells, when activated, multiply and produce cytotoxic T lymphocytes that bind to the surface of cancer and virally infected cells, destroying them, thereby protecting against cancer and viral infections. The cover illustration shows cytotoxic T cells killing a cancer cell.
The CD4 T cells are the helper lymphocytes. When activated, they help the activation of B cells and CD8 T cells; in their absence, the antigen inactivates these cells, in accord with The Two Signal Model. Thus, answering the question of ‘What To Be?’ requires us to understand what different circumstances result in the activation of B cells to produce antibody, or CD8 T cells to express cell-mediated immunity.
The role of the absence/presence of CD4 T cells in determining whether an antigen inactivates or activates B cells and CD8 T cells has been broadly accepted by the immunological community for the last three decades. Dr Bretscher argues strongly that single CD4 T cells are inactivated by antigens, and that their activation requires CD4 T cell cooperation, on both conceptual and observational grounds. An alternative, currently more popular idea is that the signal 2 for the activation of a CD4 T cell is not a result of CD4 T cell collaboration, but rather the engagement of pattern recognition receptors with pathogen-associated molecular patterns (PAMPs), representing an evolutionarily much older form of defence than the immune system. As explained below, these different ideas on the activation/inactivation of CD4 T cells are central to considering the question of ‘If To Be, What To Be?’
The conditions of immunisation leading to strong cytotoxic T lymphocytes, an expression of cell-mediated immunity, and to antibody responses, are different. This raises two important questions: What is the biological advantage of such differential regulation and what is its mechanistic basis?
The Threshold Hypothesis
Immunologists realised in the 1980s that the activation of CD4 T cells can give rise to two main types of T helper cells, Th1 and Th2 cells. Th1 cells facilitate the generation of cytotoxic T lymphocytes, i.e., of cell-mediated immunity, whereas Th2 cells facilitate B cells to produce antibodies. Thus, the question, ‘If To Be, What to Be?’, can be translated into ‘What determines whether Th1 or Th2 cells are generated?’
Dr Bretscher proposed in 1974 that Th1 and Th2 cells are respectively generated when antigen mediates tentative and robust CD4 T cell interactions. This ‘Threshold Hypothesis’ accounts for how various variables of immunisation affect the Th1/Th2 nature of the ensuing response.
Minimally foreign antigens are only able to induce naïve CD4 T cells to generate Th1 cells, as there are only a few CD4 T cells specific for such antigens; even with an optimal amount of antigen to support CD4 T cell collaboration only tentative CD4 T cell collaboration can occur. An optimal level of more foreign antigen, for which there are more CD4 T cells, supports robust CD4 T cell collaboration and so the generation of Th2 cells, whereas a lower level of the same antigen only supports tentative CD4 T cell collaboration and so Th1 cells.
Moreover, responses often evolve with time from a Th1 towards a Th2 mode. This too can be explained by Dr Bretscher’s account. The CD4 T cells multiply when an antigen impacts the immune system and so, as long as the antigen level is sustained, the strength of CD4 T cell collaboration increases, accounting for this evolution.
One example shows the biological importance of the generalisation that minimally foreign antigens should only induce cell-mediated immunity. Cancers are minimally foreign, being derived from and only slightly different from their parental self-cells, and are only susceptible to cell-mediated attack. This naturally raises the question of how they sometimes escape the immune response by deviating it into an antibody mode?
A widely held hypothesis, an alternative to the Threshold Hypothesis, is that PAMPs are required not only to activate CD4 T cells, but the nature of the particular PAMPs associated with an antigen determines the Th1/Th2 phenotype of the response. Dr Bretscher discusses in Spiral Immunology many reasons why this is implausible. One reason is that this theory does not explain the evolution of the response from a Th1 towards a Th2 mode by either PAMP-containing and PAMP-free foreign antigens, as in the former case the PAMPs do not change during the course of the response and, in the latter case, there are no PAMPs.

The Threshold Hypothesis and Medical Strategies for Prevention and Treatment
Vaccination procedures currently in use increase the rapidity of the antibody response upon infection. They are effective against pathogens contained by antibodies but ineffective against invaders only contained by cell-mediated immunity, such as cancers and the pathogens responsible for AIDS and tuberculosis. Critically, work in Dr Bretscher’s laboratory has developed ways of protecting against and treating infections caused by pathogens best and uniquely contained by cell-mediated immunity.
One such parasite causes human cutaneous leishmaniasis. Resistance and disease, respectively, correlate with Th1 and Th2 or mixed Th1/Th2 responses. Infection of mice results in a similar pattern as in humans. Infection of ‘susceptible’ mice with one million parasites quickly results in a Th2 response and progressive disease, and a sustained Th1 response and containment of the parasite in ‘resistant’ mice. Dr Bretscher’s laboratory has shown that infection of susceptible mice with 300 parasites results in a stable Th1 response and, in time, a Th1 imprint. Reinfection with a million parasites, sometime after this first infection, results in a Th1 response and resistance. The same strategy works in mice with tumours and mycobacteria, responsible for tuberculosis and leprosy. These studies underpin the low dose vaccination strategy.
Visceral leishmaniasis, which is fatal if untreated, is caused by a parasite related to the one that causes cutaneous leishmaniasis. Visceral leishmaniasis is also only contained by a Th1 response. The mixed Th1/Th2 response of patients is modulated to have a Th1 phenotype on a short treatment with drugs that kill the parasite. Dr Bretscher suggests this change occurs because the reduced antigen load following parasite killing modulates the immunity to have a Th1 phenotype.
Less than 1% of individuals infected with HIV contain the virus without treatment. These ‘elite controllers’ generate a stable cytotoxic T lymphocyte, Th1 response and produce little antibody. Most HIV-infected and untreated individuals will, sometime after infection, produce antibody as the response acquires a Th2 component, and then experience the successive stages of AIDS.
Dr Bretscher proposes that the drop in viral load, caused by anti-retroviral therapy, modulates the immune response from a non-protective, mixed Th1/Th2 response at the initiation of therapy, to a protective, Th1 mode, similarly as the administration of anti-parasite drugs results in the cure of visceral leishmaniasis. A simple assay, involving an examination of the nature of the anti-HIV antibodies, can be employed to assess when the response is of a predominant Th1 phenotype, and when anti-retroviral therapy should be stopped. Given the presence of protective immunity, viral rebound will not occur, as most often happens upon cessation of treatment at random times.
The work of Dr Bretscher is an inspiring example of the linkage between basic science and medical innovation. It is clear that Dr Bretscher has presented foundational ideas that are on course to facilitate the development of improved strategies to prevent and treat diverse diseases.
Reference
https://doi.org/10.33548/SCIENTIA743
Meet the researcher

Professor Peter Bretscher
Department of Biochemistry, Microbiology and Immunology
University of Saskatchewan
Saskatoon
Canada
Dr Peter Bretscher, Professor at the University of Saskatchewan, Canada, studied physics at Cambridge, England, as an undergraduate, and obtained his PhD in protein X-ray crystallography in 1968, at the Cambridge Laboratory of Molecular Biology. Here, he became fascinated by immunology and discussed his early ideas with Francis Crick. In 1970, Bretscher and Cohn proposed the Two-Signal Model of lymphocyte activation, a theory made to explain how the immune system fights foreign invaders but not constituents of the body to which it belongs. In 1974, he proposed how the immune system chooses one of the two major classes of immunity with which to attack an invader. This theory is pertinent to understanding how immune responses against diverse antigens, including cancers and infectious agents, are regulated, and so to achieving vaccination against AIDS, tuberculosis and cancers. In 2017, Professor Bretscher published the short book, ‘The Foundations of Immunology and Their Pertinence to Medicine’. It provides an overview of contemporary immunology and its pertinence to strategies of prevention and treatment of diverse immune-system related diseases. It is written to provoke immunologists but be accessible to non-specialists.
CONTACT
E: peter.bretscher@usask.ca
W: https://medicine.usask.ca/profiles/biochemistry-microbiology-immunology/peter-bretscher.php
FURTHER READING
P Bretscher, G Al-Yassin, Can interruption/withdrawal of anti-retroviral therapy provide personalized immunotherapy against HIV-1? Scandinavian Journal of Immunology, 2020, 92(6), e12934.
P Bretscher, On Analyzing How the Th1/Th2 Phenotype of an Immune Response Is Determined: Classical Observations Must Not Be Ignored, Frontiers in Immunology, 2019, 10, 1234.
GA Al-Yassin, PA Bretscher, Does T Cell Activation Require a Quorum of Lymphocytes? The Journal Immunology, 2018, 201(10), 2855–2861.
P Bretscher, G Wei, JN Menon, H Bielefeldt-Ohmann, Establishment of stable, cell-mediated immunity that makes “susceptible” mice resistant to Leishmania major, Science, 1992, 257(5069), 539-542.
J Salk, P Bretscher, P Salk, et al., A strategy for prophylactic vaccination against HIV, Science, 1993, 260(5112), 1270–1272.
P Bretscher, M Cohn, A Theory of Self-Nonself Discrimination, Science, 1970, 169(3950), 1042–1049.
Want to republish our articles?
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence
(CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
More articles you may like
Grandmothers: Innovation Through Tradition
Grandmother Project – Change through Culture (GMP) is an organisation dedicated to documenting the role of grandmothers and demonstrating the effectiveness of grandmother-inclusive strategies in improving the health and well-being of women, children, and adolescents. GMP’s groundbreaking work challenges conventional wisdom to transform community-based interventions in Africa and beyond, harnessing a powerful but often overlooked resource: the wisdom and influence of grandmothers.
Dr Robert Larkin | Cultivating Change to Improve Soil Health and Increase Potato Yield
Environmental quality and food production are facing the pressing challenges of climate change and global population growth. Dr Robert Larkin from the United States Department of Agriculture-Agricultural Research Service (USDA-ARS) and a team of plant scientists developed and tested a range of crop management systems to help overcome these compounding challenges. Their work is improving soil health and increasing the yield of potato crops, contributing to the future food security of nations.
Professor Giorgio Buttazzo | Artificial Intelligence and a Crossroads for Humanity
Where do we stand with artificial intelligence? Might machines take over our jobs? Can machines become conscious? Might we be harmed by robots? What is the future of humanity? Professor Giorgio Buttazzo of Scuola Superiore Sant’Anna is an expert in artificial intelligence and neural networks. In a recent publication, he provides considered insights into some of the most pressing questions surrounding artificial intelligence and humanity.
Dr Ralf Adam | New Technologies Shaping the Future of Oral Hygiene
Understanding the efficiency of various toothbrush technologies is essential for achieving optimal oral health. Dr Ralf Adam, who leads a dedicated team at Procter & Gamble in Germany, is keen to investigate the complexities of these technologies. His team have provided new insights into the best toothbrush types for plaque removal and the maintenance of gum health. By highlighting the importance of informed oral care decisions and ongoing investigations, this vital research works towards ensuring everyone can achieve a brighter, healthier smile.




