Dr Richard B. Brandon | Dr Thomas D. Yager – A Fully Integrated Diagnostic Test to Discriminate Sepsis from Infection-negative Systemic Inflammation
Sepsis is a serious medical condition that manifests with a dysregulated immune response to an infection in the bloodstream, and is a major cause of morbidity and mortality worldwide. Many non-infectious conditions can lead to a state of hyper-inflammation known as the systemic inflammatory response syndrome (SIRS), which clinically can look very similar to sepsis. A lack of analytical tools that allow for the early detection of sepsis makes discrimination between sepsis and SIRS a very complex but critical diagnostic task. Dr Richard B. Brandon, Dr Thomas D. Yager and their colleagues at Immunexpress have developed a molecular diagnostic platform capable of detecting genetic biomarkers diagnostic for sepsis in about an hour. Early sepsis diagnosis can potentially lead to better patient management, more efficient use of resources, and a more sensible approach to the use of antibiotics.
Differentiating Between Sepsis and Sterile Inflammation
During severe sepsis, the immune reaction becomes so dysregulated that multi-organ failure and death can result. It is important to note that not all patients presenting with a hyper-inflammatory response are septic. Some patients who have acute dehydration, intestinal or end organ ischemia, autoimmune disorders, adrenal insufficiency or haematological malignancy can also present with SIRS, a state of inflammation characterised by fever, increased heart rate and other clinical signs that are commonly associated with sepsis.
Although important, differentiating between sepsis and the non-infectious SIRS condition is a very challenging diagnostic task because patients present with similar clinical signs. The diagnostic tools currently available to most ICU physicians are less than adequate, leaving clinical staff with no choice but treat all patients suspected to have sepsis with antibiotics. However, the overuse of antibiotics can lead to side effects due to microbiome disruption, as well as the development of antibiotic-resistant infections among the most vulnerable patients. In any case, antibiotic therapy will not be effective in the treatment of viral sepsis.
Immunexpress is a molecular diagnostic company, founded in Australia and based in Seattle, Washington, USA, with the mission of improving the outcomes for patients suspected of sepsis. The Immunexpress team of researchers have discovered specific immune system biomarkers, now implemented in SeptiCyte® technology that allows clinicians to diagnose sepsis accurately and rapidly, differentiating it from SIRS in critically ill patients.
The technology, which led initially to the development of a kit named SeptiCyte LAB, was recently transitioned to a next generation test, SeptiCyte RAPID, which is entirely self-contained in a cartridge that runs on the Biocartis Idylla™ system. Both tests rely on the same gene expression assay, but the newer version has a turnaround time for results of about an hour.
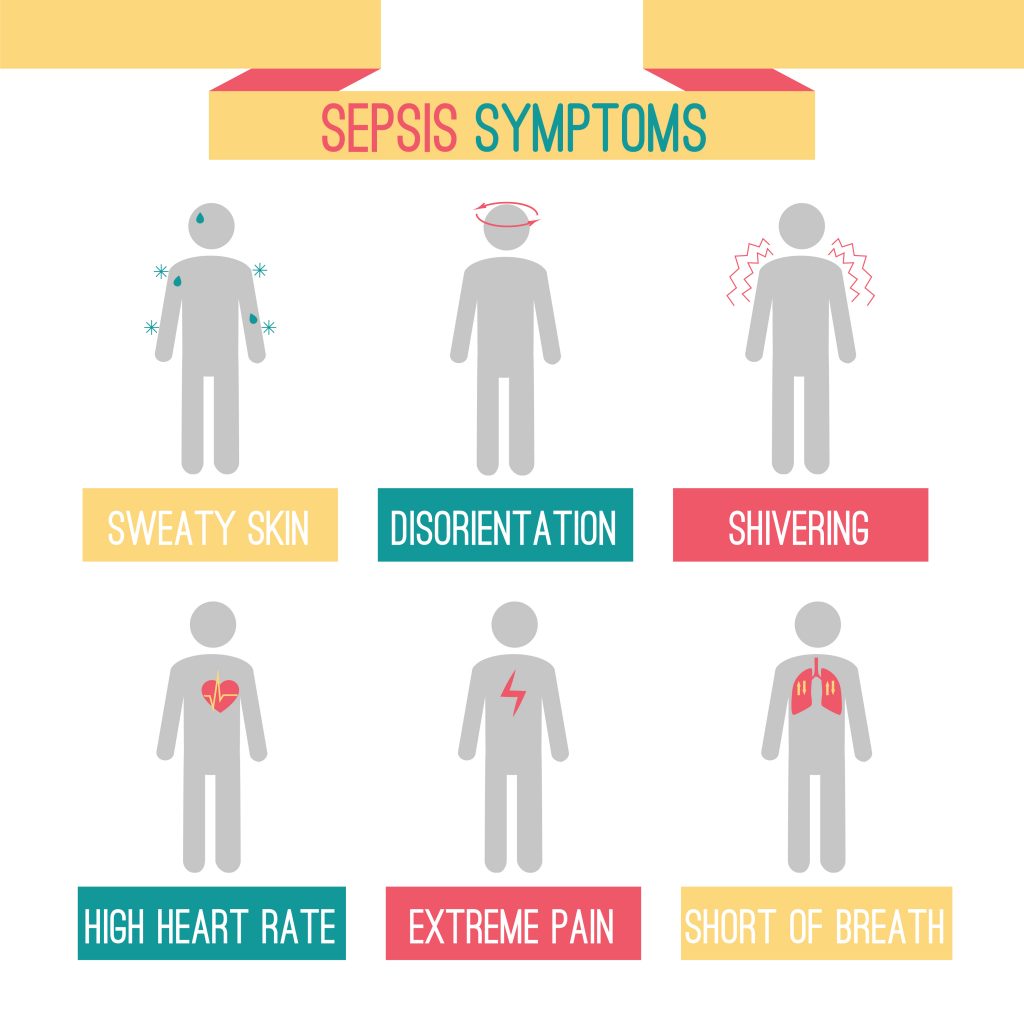
Importance of a Rapid Diagnosis of Sepsis
The timing of the diagnosis of sepsis can make the difference between survival and death. Physicians may or may not spot early sepsis signs in the initial stages of the disease, before multi-organ failure begins to occur and when treatment would be most effective. These early clinical signs can often be absent or indistinguishable from those of SIRS. The earlier sepsis is diagnosed, the greater are the chances of a favourable prognosis following therapeutic intervention.
Unfortunately, most of the current microbiology procedures adopted in the diagnosis of sepsis are affected by a large proportion of false positives (the wrong indication that a disease is present) or false negatives (the wrong indication that a disease is absent). In the absence of accurate and reliable diagnostic tools, physicians often rely on their clinical experience and intuition to make an early diagnosis of sepsis.
The variability in clinical presentations and in the training and experience of physicians make the diagnosis of sepsis particularly complex, especially before serious organ damage is evident. The team at Immunexpress quantified physician agreement in diagnosing SIRS or sepsis in a study published in 2019. They concluded in this work that there is a poor degree of consensus between different physicians in the differential diagnosis of sepsis. The least agreement was reached among physicians treating ICU patients presenting with clinical signs of respiratory illness.
Normally, sepsis is diagnosed through blood tests and cultures to confirm the presence of bacterial, viral or fungal infection along with other parameters, such as imbalanced electrolytes, reduced oxygen levels and increased levels of blood clotting factors. Unfortunately, blood tests that focus on confirming the presence of a pathogen take time to yield clinically actionable results, with most of these tests producing results in an about 24 to 48 hours.
A different approach to confirming infection, that potentially could yield results in about 1 hour, would be to look at molecular biomarkers that are up- or down-regulated when the immune system is responding to a pathogen. The human immune system responds specifically to an invading pathogen and expresses a variety of genes that produce detectable genetic signatures. These signatures could allow clinicians to discriminate between patients who are affected by sepsis from those who aren’t.
The researchers at Immunexpress Inc. developed and validated a gene expression assay method based on the blood expression levels of four genes involved in the host response to infection. Their SeptiCyte LAB assay is able to detect the presence of sepsis biomarkers more rapidly – in approximately 6 hours – and more accurately than methods that rely on the direct identification of pathogens.
The SeptiCyte LAB test was cleared for market by the Food and Drug Administration (FDA) in the USA, on the basis of clinical trials involving 447 patients. The objective of the studies was to measure the performance of the assay method in discriminating between sepsis and SIRS in adults in critical care. Compared with other diagnostic methods, SeptiCyte LAB scored better at discriminating between cases of sepsis from cases of SIRS and the diagnostic power was not affected by the severity of disease. The method was confirmed to be both sensitive and specific in confirming the presence of infection, as it decreased the occurrence of false negative and false positive diagnoses compared to other diagnostic methods.
Development of a Near-patient Diagnostic Platform
Although cleared for market by the FDA, SeptiCyte LAB was never commercialised, due to its relatively long turnaround time (around 6 hours) in providing diagnostic evidence of sepsis. Its traditional workflow setup meant that testing had to be conducted in a centralised laboratory by highly experienced technicians.
To address these issues, the researchers at Immunexpress developed SeptiCyte RAPID, a sepsis diagnostic tool that relies on the measurement of two peripheral-blood gene-expression biomarkers using the same quantitative reverse transcription polymerase chain reaction (RT-qPCR) which highly correlates with SeptiCyte LAB clinical findings, but with a much easier workflow. Further clinical trials confirmed that SeptiCyte RAPID accurately differentiates between non-infectious systemic inflammation and sepsis.
The RAPID test is easy to administer and fully automated. The test requires minimal sample handling and can be performed using whole blood. The near-patient, cartridge-based RAPID test is fully integrated, from sample extraction, to sample analysis by RT-qPCR, and finally to the automated generation of a report. Since the technique does not rely on pathogen cultures, no incubation time is needed for the samples. In addition, samples are barcode-traceable. The RAPID platform provides results in around an hour, only requiring 2.5 ml of whole blood drawn into a PAXgene tube. The Idylla cartridge uses about 1/10 of the initial blood draw and only 2 minutes of set up time by a technician is required.
Although easier to use, the RAPID test correlates well with its manual counterpart in terms of accuracy of results. The technology can diagnose sepsis up to 48 hours prior to conventional microbiology-based methods, enabling physicians to choose the most appropriate treatment.
A further SeptiCyte product is being optimised that differentiates between bacterial and viral sepsis. This will be an important development, as viral sepsis does not respond to the antibiotic treatments routinely given to all patients suspected of presenting with sepsis. Early and accurate sepsis diagnosis in trauma and burns patients, and post-surgical patients suspected of infection, will lead to better patient management and outcomes, more efficient use of hospital resources, and more sensible use of antibiotics.
COVID-19 and the Management of Sepsis
Severe COVID-19 infections, like other viral infections, can lead to the sustained dysregulation of the host immune response that defines sepsis. Severe cases of COVID-19 can manifest with a wide range of clinical effects including breathing difficulties, hyperinflammation, confusion, accelerated heart rate, and even shock, multi-organ failure and death. Escalating clinical signs and symptoms that point toward multi-organ failure, taken together with a positive COVID-19 test result, confirms the presence of sepsis in severely ill patients.
The current COVID-19 crisis has highlighted the need for technologies that can discriminate between cases of mild infection from those that may develop sepsis. Sepsis is among the most common factors associated with COVID-19 mortality. The early diagnosis of bacterial and viral sepsis will assist in triaging COVID-19 patients, ensuring rapid sepsis management and saving lives.
Exciting Prospects for SeptiCyte RAPID
Early diagnosis of sepsis would lead to more appropriate treatment choices, improved prognosis, fewer days in intensive care and hospital, more efficient use of hospital resources and more appropriate use of antibiotic treatment, only when confirmed to be necessary.
Dr Brandon, Dr Yager and their colleagues at Immunexpress have successfully discovered, validated and patent protected Septicyte LAB and SeptiCyte RAPID. The latter is a fully-integrated, rapid, near-patient, gene expression diagnostic method that provides accurate results in 1 hour turnaround time to differentiate sepsis from SIRS. The team is now validating SeptiCyte RAPID in real-time studies across multiple sites in the USA and Europe and plans to launch the technology and get full FDA clearance by Q1 2021.
Reference
https://doi.org/10.33548/SCIENTIA602
Meet the researchers

Dr Richard Bruce Brandon, PhD
Chief Scientific Officer
Immunexpress
Seattle, WA
USA
Dr Richard Bruce Brandon is a co-founder of Immunexpress and is currently the Chief Scientific Officer. Dr Brandon has a PhD in biochemistry and molecular genetics from the University of Queensland, Australia. His interest and knowledge of infectious disease, immunology, molecular genetics and veterinary clinical diagnostic procedures helped him develop the original concept behind the creation of Immunexpress, which was established in Australia in 2006 and has now expanded with offices in Seattle, USA.

Dr Thomas Dean Yager, PhD
Director of Research and Development
Immunexpress
Seattle, WA
USA
Dr Thomas Dean Yager obtained his PhD in Biochemistry and Biophysics from Oregon State University. In 2014, after several successful years as a consultant and lead scientist in the field of molecular diagnostics for various companies in Canada and the USA, he became Director of Research and Development at Immunexpress, where he had a fundamental role in developing the SeptiCyte RAPID cartridge test for discriminating between sepsis and SIRS.
CONTACT
E: clinical@septicyte.com
Twitter: @Immunexpress
KEY COLLABORATORS
Professor Jerry Zimmerman, Seattle Children’s Hospital, Seattle WA
Professor Mahdad Noursadeghi, University College London, London UK
Professor Tom van der Pol, Amsterdam Medical Center, Amsterdam, Netherlands
FURTHER READING
BK Lopansri, RR Miller, JP Burke, et al., Physician agreement on the diagnosis of sepsis in the intensive care unit: estimation of concordance and analysis of underlying factors in a multi-center cohort, Journal of Intensive Care, 2019, 7, 13.
RR Miller, BK Lopansri, JP Burke, et al., Validation of a host response assay, SeptiCyte LAB, for discriminating sepsis from systemic inflammatory response syndrome in the ICU, American Journal of Respiratory and Critical Care Medicine, 2018, 198, 903–913.
JJ Zimmerman, E Sullivan, TD Yager, et al., Diagnostic Accuracy of a Host Gene Expression Signature That Discriminates Clinical Severe Sepsis Syndrome and Infection-Negative Systemic Inflammation Among Critically Ill Children, Critical Care Medicine, 2017, 45, e418-e425.
L McHugh, TA Seldon, RA Brandon RA, et al., A Molecular Host Response Assay to Discriminate Between Sepsis and Infection-Negative Systemic Inflammation in Critically Ill Patients: Discovery and Validation in Independent Cohorts, PLoS Medicine, 2015, 12, e1001916.

Want to republish our articles?
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence
(CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
More articles you may like
Prof Candis M. Morello – Prof Jan D. Hirsch | Recent innovations in pharmacy education
A pioneering research team from the Skaggs School of Pharmacy and Pharmaceutical Sciences, University of California, San Diego, United States, has been instrumental in developing innovative techniques for teaching pharmacy students. The Next Generation of Pharmacist Educators (NextGen-RxEd) programme is a new method of training the next generation of pharmacist educators and academics. To help pharmacists and pharmacy students visualise the complex issues experienced by their patients, the team led by Professors Candis Morello and Jan Hirsch developed an innovative educational tool, called the Medication Therapy Management (MTM) Spider Web.
Professor Michael Yarus | How RNA Started the Conversation That Built Life
The genetic code stores the instructions for building proteins, yet how it first arose remains unclear. It likely did not appear fully formed, but instead emerged step by step from simple chemical interactions. In this study, a team led by Professor Michael Yarus at the University of Colorado Boulder shows how early ribonucleic acid (RNA) molecules could bind specific amino acids, creating the first coding relationships. These early interactions are then refined through evolutionary processes such as duplication and merging of partial codes. By combining the experimental data with computer simulations, the work provides a testable pathway from prebiotic chemistry to the modern genetic code.
Identifying Nutritional Risk in Early Childhood: Insights from NutriSTEP®
Early childhood is a critical period for growth and development, yet many young children face nutritionrelated risks that can go unnoticed. Professor Janis Randall Simpson and colleagues have developed NutriSTEP®, validated and reliable screening tools that help identify potential nutritional concerns in toddlers and preschoolers. Their large-scale analysis of Canadian data reveals patterns in diet, behaviour, and food access that could help guide early interventions and support healthy development.
Professor Tian Yu Cao | Twistor Theory: A New Framework for Quantum Gravity
At Boston University, Professor Tian Yu Cao is rethinking the foundations of modern physics. His work builds on twistor theory which demonstrates that spacetime is secondarily derived from twistor constructions, but goes further to highlight the most important implication of the Penrose transform in that the primary physical agents can only be mathematically described by elements of cohomology with the defining feature having roots in spin. This view of primary agents combines with Cao’s other major claim that quantum behaviour itself may arise from the physical property of spin leads to a new consistent framework of quantum gravity in which long-standing puzzles in black holes (evaporations) and cosmology (transitions between cycles of cosmos) can be adequately addressed, with the crucial help from the on-going development of operator product expansion formular defined on twistor space.




