Dr Sanju A. Sanjaya | Bagyalakshmi Muthan – Creating Power Foods with Gene Technology
At least 820 million people suffer from hunger and malnutrition globally and human population growth is likely to exacerbate this problem in the future. It is becoming increasingly important to develop sustainable and efficient methods to meet food demands. To address this global issue, Dr Sanju A. Sanjaya and Bagyalakshmi Muthan from West Virginia State University and their colleagues from Michigan State University have developed genetic technologies to improve the nutritional and energy content of crops. Their technology could increase production and improve profitability and sustainability across a range of important crop plants.
The Sustainable Solution Agriculture Needs
The human population continues to expand rapidly, increasing the demand for food – both for ourselves and for pets and livestock. Agricultural intensification over the last few decades has allowed farmers to increase crop yields significantly and has led to the relatively low cost of food that we enjoy today. However, intensive agricultural practices are also associated with negative environmental consequences, such as habitat destruction, soil erosion, and loss of biodiversity. Many farmers are already struggling to maintain yields as these pressures grow, let alone increase them to meet future demands.
Solutions that are sustainable and environmentally friendly are urgently needed – particularly if the goal to reduce or eliminate global hunger and malnutrition is to be achieved. New tools in agriculture, ecosystem management and genetic technologies provide a suite of solutions that are sustainable, efficient, and cost-effective.
Recent advancements in genetic and molecular technologies have given scientists precise control over gene editing. Specific genes can be targeted for editing by using molecular tools with complementary DNA sequences. Genes can be altered, damaged genes can be removed, and new genes with beneficial effects can be added efficiently, and with few to zero errors. By harnessing the power of genetic technologies, scientists have created crops that are disease and pest resistant, tolerate harsh environmental conditions, and have enhanced growth rates and nutritional content.
Building on years of genetic and molecular research, Dr Sanju Sanjaya, Bagyalakshmi Muthan and their colleagues from West Virginia State University and Michigan State University have developed genetic tools to improve the nutritional and energy content of crops. They increased the energy content of their genetically edited plants by targeting the mechanisms that plants use to produce energy storage compounds, the carbohydrates and ‘lipids’ – or fats.
Healthy fats are important for heart health, cholesterol management, and overall health. Additionally, the enhanced energy content of Dr Sanjaya and his colleagues’ crops could help to stave off hunger and malnutrition in vulnerable communities, particularly if combined with technologies that provide other beneficial improvements. These crops could provide environmental and economic benefits by reducing the area required to produce the same amount of energy as a traditional crop variety.
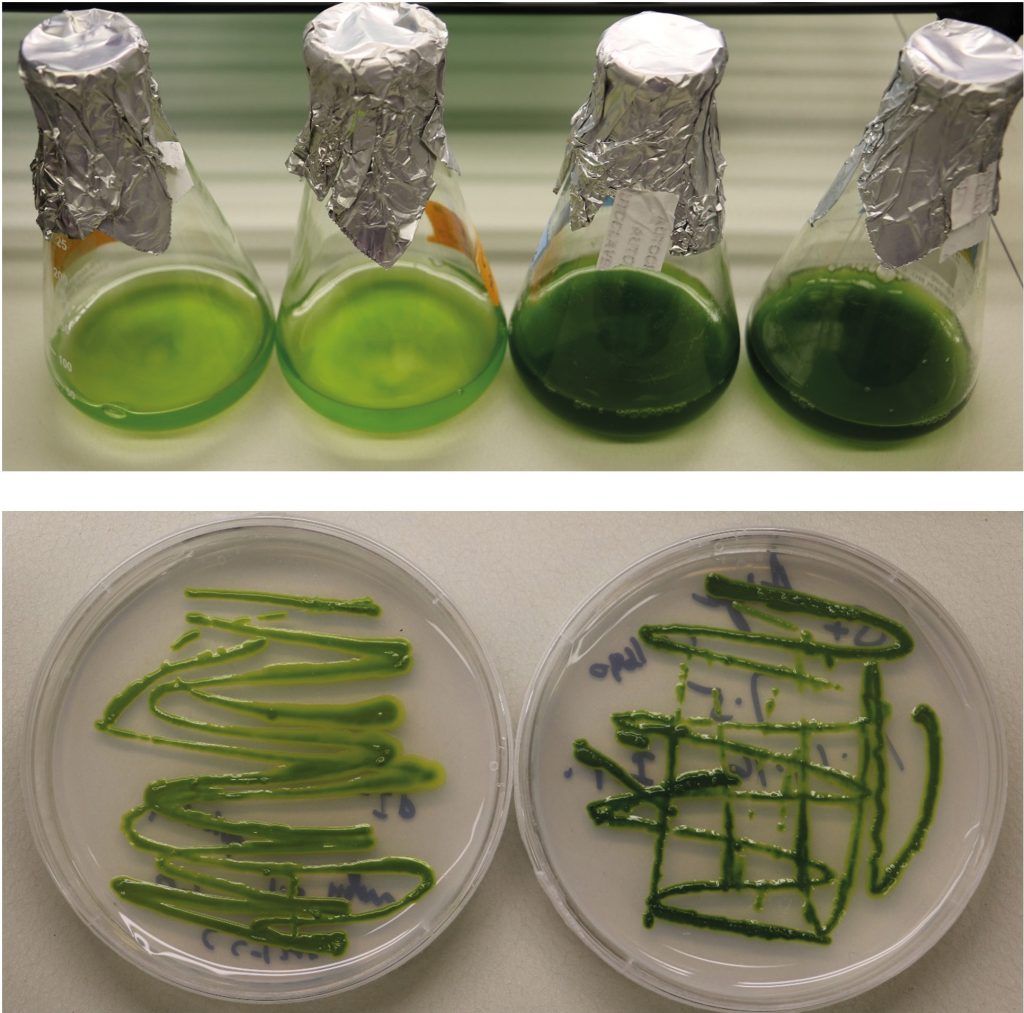
Green algae have the natural ability to synthesise and store Omega-3 fatty acids in their cells.
Increasing Lipid Production and Storage
Plants are the predominant ‘photosynthesising’ organisms on land – they capture carbon dioxide from the air and convert it into usable sugars and fats. In aquatic environments, photosynthesis is dominated by algae. From large seaweed attached to the ocean floor through to microscopic single-celled species – or ‘microalgae’ – drifting in the water column, algae can be found just about anywhere there is water, including ponds, rivers, lakes, and even under a dripping garden tap.
Mature plants direct most of the carbon they capture into the creation of starch – a large carbohydrate that can be broken down into simple sugars and converted to energy. Starch is stored throughout the plant’s ‘vegetative tissues’ – such as the stem, leaves, and roots – to provide an easily accessible energy source when photosynthesis is not possible. In contrast, lipids are more energy-dense than carbohydrates and take up less room for the equivalent amount of energy. The process of creating or breaking down lipids is more involved and time-consuming to achieve though, in both plants and animals. Therefore, plants tend to reserve the bulk of their lipid production and storage for places where space is at a premium – in their seeds. This is why nuts and other seeds are considered power foods – they fit a lot of energy into a small package.
Because plants can only capture a finite amount of carbon through photosynthesis, lipid and carbohydrate production are competing processes. Plants use complex metabolic pathways to partition carbon for storage compound production, which involve several enzymes and ‘coenzymes’ – the enzyme helpers. These pathways, and the associated enzymes, are remarkably similar in plants and algae, with the key difference being that microalgae preferentially store captured carbon as lipids. ‘Many of us think Omega-3 fatty acids are made in fish and consume more fish and fish oil,’ explains Dr Sanjaya. ‘In reality, fish consume algae and accumulate these specialised fatty acids in their fat tissues as a reservoir.’
Through their investigations, Dr Sanjaya and his colleagues identified a group of five novel enzymes in the microalgae Chlamydomonas reinhardtii that regulate storage compound pathways. They used molecular tools to determine which genes are responsible for the production of these enzymes and increasing the activity of the associated metabolic pathways. They then inserted these algal genes into the genome of a model plant species, Arabidopsis thaliana – a flowering species related to radishes, mustard and cabbage. The genetically engineered – or ‘transgenic’ – plants produced the same enzymes as observed in the algae. Plants producing one of these five enzymes stored more lipids in all their tissues – not just their seeds.
‘With increased concentrations of seed storage compounds, these crops will help meet the global population’s growing nutritional and fuel needs.’
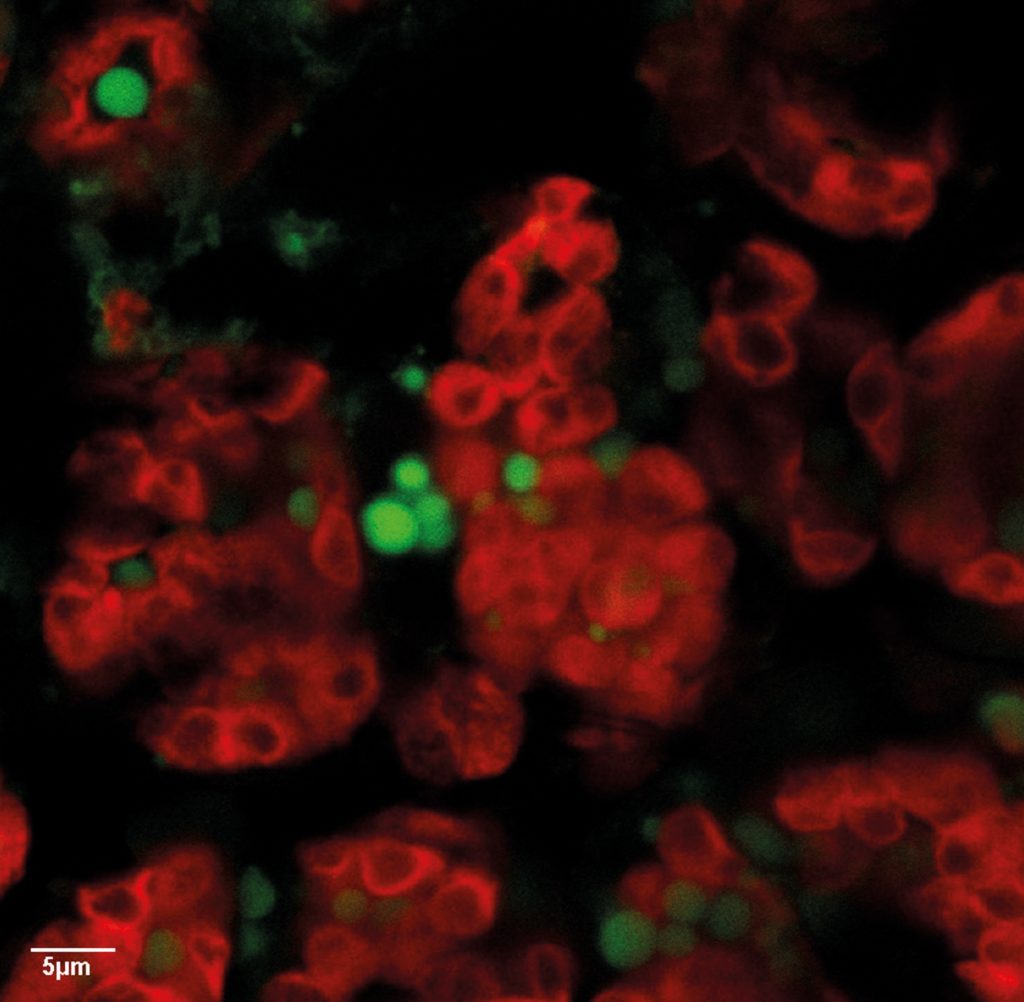
Expression of algal enzyme increases the production of oil droplets in plants.
Seed Oil Storage – Not Limited to Seeds
Tissue analysis revealed that the transgenic plants stored up to 6% more lipids in their vegetative tissues than the original plant lineage. So much, in fact, that globules of stored oil could be seen within the leaves under a microscope. The researchers also showed that this enhanced energy content had the expected dietary benefits. Moth caterpillars fed the transgenic plants gained more weight than caterpillars that consumed the same amount of the original plant lineage, weighing approximately a third more at the end of the feeding trial.
Analysis of the stored oil demonstrated that the lipid composition was similar to that observed in seed storage. In practice, this means that the entire plant could be used to harvest seed oils, greatly reducing the waste associated with seed oil production, and increasing efficiency and profitability.
‘Enhancing the genetic control of seed development and storage compound metabolism will lead to qualitative and quantitative improvement of oilseed crops,’ says Dr Sanjaya. ‘With increased concentrations of seed storage compounds, these crops will help meet the global population’s growing nutritional and fuel needs.’
Of the five microalgae enzymes Dr Sanjaya and his colleagues identified, only one has been characterised in the model plant species. The functions of the other four enzymes in plants are still unknown and could offer great potential in other plant species. The researchers are currently using genetic and metabolic engineering approaches to determine the enzymes’ functions in storage compound regulation in plants. Dr Sanjaya suggests that understanding their function could facilitate the development of genetic technologies that alter the production and storage of different types of lipids, which vary in size and composition. These could have advantages across different applications, either as a food source, or in the production of biodiesel.
The methods for augmenting plant genomes with algal genes have been developed by Dr Sanjaya and his colleagues over years of sustained effort. Their patented process includes the method for genetically editing plant cells or tissues, growing the plant cells or tissues into a whole plant, growing the plants from seed, and harvesting the fats or oils from the mature plant.

Camelina Sativa – a promising oilseed crop.
A Technology Full of Potential
Dr Sanjaya and his colleagues are now in the process of using their technique to improve the nutritional value of the oilseed crop camelina, which is also known as false flax or wild flax. Camelina is a robust crop able to flourish on poor-quality soils, such as sandy, rocky, or nutrient-poor soils, making it ideal for cultivation where other crops would struggle. ‘This energy-rich oilseed crop can grow on the marginal soil, such as reclaimed surface coal mine lands found in the USA’s Appalachian regions,’ says Dr Sanjaya. Energy-enhanced camelina could therefore provide nutritional benefits while also reserving prime agricultural land for other crops. In regions where poor soils are widespread, energy-enhanced camelina could provide a lifeline.
In the future, the technology could be used to increase the oil content across the vegetative tissues of a range of important crop species that produce and store oil in their seeds. Potentials include corn, soybean, potato, rice, alfalfa, and oilseed crops like sunflower or oilseed rape.
In addition to meeting food demands into the future, energy-enhanced crops are valuable for biodiesel production. Biodiesel offers a sustainable alternative to non-renewable fuels, such as petroleum. However, the crops used in biodiesel production include important food crops. Future reliance on biodiesel without implementing measures to avoid competition between food crop and biodiesel crop production could result in a rapid rise in food and fuel prices, increasing social inequality and hunger.
Energy-enhanced transgenic crops specifically designed for biodiesel production could provide a solution, by separating the cultivation for food and fuel. The higher lipid content of these crops also translates to higher biodiesel production per acre of farmland, which leaves more space for food production, development, and natural spaces.
With mounting environmental pressures and increasing demands from a growing human population, Dr Sanjaya’s gene technologies could provide the sustainable solutions we need for a better future.
Reference
https://doi.org/10.33548/SCIENTIA644
Meet the researchers

Dr Sanju A. Sanjaya
Energy and Environmental Science Institute
Department of Biology
Agricultural and Environmental Research Experimental Station
West Virginia State University
Institute, WV
USA
Dr Sanju Sanjaya earned his PhD in Applied Botany from the University of Mysore, India. Upon graduating, he conducted his postdoctoral research in India, Taiwan, and the USA. Before joining West Virginia State University, he held the position of Senior Research Associate at Michigan State University. Dr Sanjaya currently holds the position of Assistant Professor of Bioenergy and Environmental Biotechnology at West Virginia State University, and Director of the university’s Energy and Environmental Science Institute. His main research focus is in plant biotechnology, and designing plants and microalgae with enhanced properties, such as bioenergy and nutritional content, that improve sustainability, profitability, and production. Dr Sanjaya’s research has been published in numerous prestigious journals and he holds patents for several of his gene technology inventions.
CONTACT
E: sanjaya@wvstateu.edu
W: http://www.wvstateu.edu/Faculty/Dr-Sanjaya-(1).aspx

Bagyalakshmi Muthan
Douglass Land-Grant Institute
Agricultural and Environmental Research Experimental Station
Energy and Environmental Science Institute
West Virginia State University
Institute, WV
USA
Bagyalakshmi Muthan received her MPhil in Botany from the Bharathiar University Coimbatore, India, for a thesis entitled ‘Phytochemical and antimicrobial activities of tubers and seeds of Gloriosa superba L’. Upon graduating, she joined Agricultural Biotechnology Research Center at Academia Sinica in Taipei, Taiwan, where her work involved genetically engineering rice, orchids, and tomatoes, after which she held a Research Associate position at Michigan State University. Muthan currently holds the position of Research Associate at West Virginia State University, Gus R. Douglass Land-Grant Institute, and Agricultural and Environmental Research Experimental Station. Her primary focus is in conducting independent research in plant biochemistry, lipids, statistics, bioinformatics, metabolic engineering and biofuels, Agrobacterium-mediated transformation of crops, and biotechnology.
CONTACT
E: bmuthan@wvstateu.edu
KEY COLLABORATORS
Dr Christoph Benning, Michigan State University
Dr Edgar Cahoon, University of Nebraska-Lincoln
Dr Timothy Durrett, Kansas State University
Dr Dylan Kosma, University of Nevada
Dr Abraham Koo, University of Missouri
Dr Changcheng Xu, Brookhaven National Laboratory
Dr Christian Tobias, USDA-ARS
FUNDING
United States Department of Energy
United States Air Force Office of Scientific Research
United States Department of Agriculture, 1890 Capacity Building Grant
United States Department of Agriculture, Evens-Allen Grant
National Science Foundation Research Initiation Award Grant
FURTHER READING
SA Sanjaya, R Miller, TP Durrett, DK Kosma, TA Lydic, B Muthan, AJK Koo, YV Bukhman, GE Reid, GA Howe, J Ohlrogge, C Benning, Altered Lipid Composition and Enhanced Nutritional Value of Arabidopsis Leaves following Introduction of an Algal Diacylglycerol Acyltransferase 2, The Plant Cell, 2013, 25, 677–693.
SA Sanjaya, TP Durrett, SE Weise, C Benning, Increasing the energy density of vegetative tissues by diverting carbon from starch to oil biosynthesis in transgenic Arabidopsis, Plant Biotechnology Journal, 2011, 9, 874–883.
C Benning, SA Sanjaya, R Miller, Increased calorific and nutritional content of plant biomass, Issued US Patent Number 10,392,629, August 2019.
C Benning, SA Sanjaya, Genetically engineered plants with increased vegetative oil content, Issued US Patent Number 10,280,432, May 2019.

Want to republish our articles?
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence
(CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
More articles you may like
Grandmothers: Innovation Through Tradition
Grandmother Project – Change through Culture (GMP) is an organisation dedicated to documenting the role of grandmothers and demonstrating the effectiveness of grandmother-inclusive strategies in improving the health and well-being of women, children, and adolescents. GMP’s groundbreaking work challenges conventional wisdom to transform community-based interventions in Africa and beyond, harnessing a powerful but often overlooked resource: the wisdom and influence of grandmothers.
Dr Robert Larkin | Cultivating Change to Improve Soil Health and Increase Potato Yield
Environmental quality and food production are facing the pressing challenges of climate change and global population growth. Dr Robert Larkin from the United States Department of Agriculture-Agricultural Research Service (USDA-ARS) and a team of plant scientists developed and tested a range of crop management systems to help overcome these compounding challenges. Their work is improving soil health and increasing the yield of potato crops, contributing to the future food security of nations.
Professor Giorgio Buttazzo | Artificial Intelligence and a Crossroads for Humanity
Where do we stand with artificial intelligence? Might machines take over our jobs? Can machines become conscious? Might we be harmed by robots? What is the future of humanity? Professor Giorgio Buttazzo of Scuola Superiore Sant’Anna is an expert in artificial intelligence and neural networks. In a recent publication, he provides considered insights into some of the most pressing questions surrounding artificial intelligence and humanity.
Dr Ralf Adam | New Technologies Shaping the Future of Oral Hygiene
Understanding the efficiency of various toothbrush technologies is essential for achieving optimal oral health. Dr Ralf Adam, who leads a dedicated team at Procter & Gamble in Germany, is keen to investigate the complexities of these technologies. His team have provided new insights into the best toothbrush types for plaque removal and the maintenance of gum health. By highlighting the importance of informed oral care decisions and ongoing investigations, this vital research works towards ensuring everyone can achieve a brighter, healthier smile.




