Dr Scott Gerber – A Novel Combination Therapy for Pancreatic Ductal Adenocarcinoma
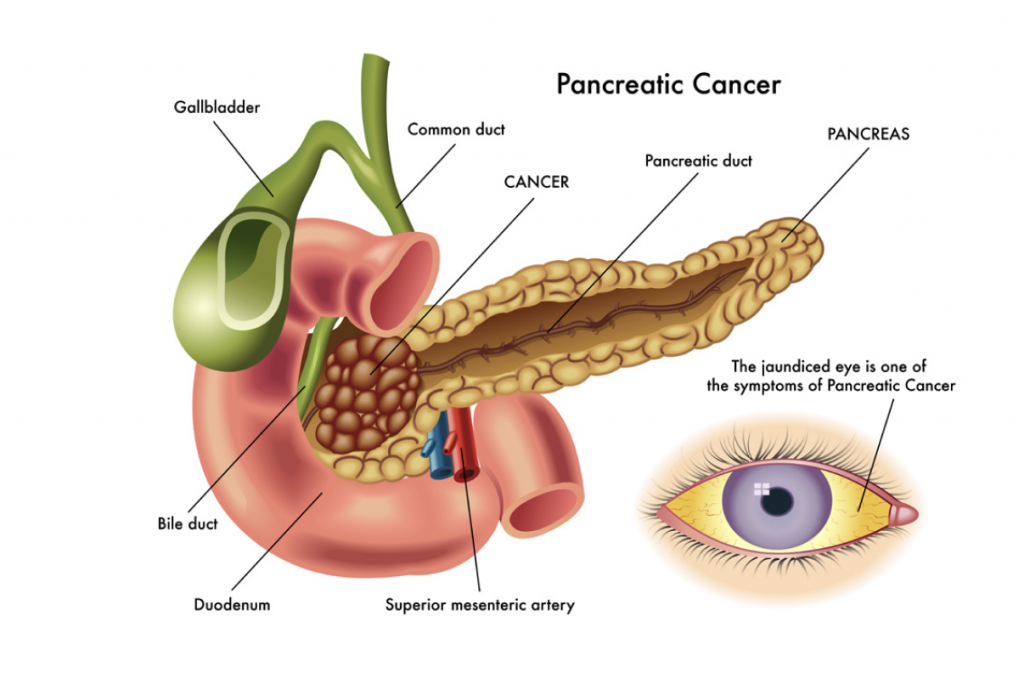
Pancreatic ductal adenocarcinoma (PDA) is an aggressive type of cancer. It is relatively common and is one of the leading causes of cancer mortality. Unfortunately, it is often detected only in the late stage of the disease and fails to respond to pre-surgical approaches, such as chemotherapy or radiotherapy, that are needed to shrink the tumour mass before surgical removal. Dr Scott Gerber at the University of Rochester Medical Center, USA, is working with colleagues to develop a novel combined therapy to overcome this issue and increase the survival of PDA patients.
Why is Survival of Pancreatic Ductal Adenocarcinoma Patients So Poor?
The prognosis for pancreatic ductal adenocarcinoma (PDA) patients is very poor. The only cure for this cancer is the surgical removal of the tumour but by the time that patients are diagnosed, the tumour tends to be very advanced. Consequently, only a minority of patients – between 10 and 20% – are eligible for surgery or respond to initial treatment, also called neoadjuvant therapy, to shrink their tumour such that the patient is appropriate for surgery.
There are currently several potential options for shrinking the mass of tumours. However, in PDA, chemotherapy or combined chemotherapy and radiotherapy regimes have proven to be largely ineffective in reducing the size of the advanced tumours.
An Alternative to Conventional Radiotherapy
Recently, stereotactic body radiation therapy (SBRT), a method which uses radiation to target the tumour in an extremely precise fashion, is now showing some promise as a neoadjuvant to shrink the tumour before surgical removal.
Typically, conventional radiotherapy consists of small doses of radiation delivered over 4–6 weeks. This method is somewhat ineffective in treating PDA and often results in damage to normal tissue. However, the precision targeting of SBRT allows for higher doses of radiation to be administered over days, rather than weeks, and results in much higher levels of tumour cell destruction, while the damage to local, healthy tissues is minimised.
It is now widely accepted that the immune system mediates many of the anti-tumour effects of radiotherapy. Importantly, it has been shown that SBRT does a better job stimulating the immune system to fight cancer when compared to conventional radiotherapy. This is an important aspect of the treatment as it enables a stronger immune attack on the tumour cells, which have been exposed by the SBRT treatment. In addition, SBRT induces processes within the tumour that encourage the release of chemoattractants, which are chemical messengers that encourage vital immune cells to infiltrate the remaining tumour.
Stereotactic Body Radiation Therapy to Treat PDA
Dr Scott Gerber at the University of Rochester Medical Center, USA, and his team recently completed a clinical trial examining how well SBRT activated the immune system in PDA. On comparing untreated to SBRT-treated tumours, they found that CD8 T-cells, an immune cell type important to initiating tumour cell death, had infiltrated into the middle of only the SBRT-treated tumours. However, it is important to note that the treatment did not alter the numbers of CD68+ immune system cells in the tumour, which are key to the tumour’s survival as these cells suppress the immune system response. Overall, SBRT did initiate an anti-tumour immune response, but could not sustain this response long enough to eliminate the tumours. Therefore, the group looked toward immunotherapy to enhance SBRT and bolster the immune attack on the tumour.
The Problems with Immunotherapy in PDA
Immunotherapy for cancer treatment, in general, is a relatively novel approach, and to date, there have been only limited benefits, especially for patients with PDA. There are several different modes of immune therapies that may be used in PDA tumour treatment. However, depending on the precise strategy used, most are ineffective, can be relatively toxic to the patient and, in other cases, can lead to the tumour becoming resistant to treatments. This is largely due to a unique tumour microenvironment where cancerous PDA cells hijack certain immune cells and cause them to promote tumour progression rather than inhibit it.
This phenomenon also makes it difficult for immunotherapies, which rely on stimulating immune cells against the tumour, to work well in this particular malignancy. In PDA, many of the anti-tumour mechanisms are inactive resulting in the survival of the tumour. To overcome these hurdles, Dr Gerber’s team made use of a potent anti-tumour cytokine, called Interleukin-12, to ramp up the immune system.
Targeted Delivery of Interleukin 12
Interleukin 12 (IL-12) is a pleiotropic cytokine, a small, protein chemical messenger that can affect many different cell types. In early tests, IL-12 appeared to have significant potential as an anti-tumour cytokine that operates by promoting activation of T-cells. However, early trials generated poor results and showed high levels of toxicity to patients when it was administered systematically. More recently, researchers have focused on delivering doses of IL-12 directly into the tumour, anticipating that targeted delivery will provide the benefits of IL-12 therapy whilst minimising the toxicity issues unveiled in the early testing.
Dr Gerber and his colleagues have adopted the novel approach of encapsulating immune-modulatory IL-12 in microspheres, which are small particles. The microspheres serve a dual function. First, they protect the IL-12 from destruction by enzymes in the body, and second, they allow a slow, sustained release of IL-12 into the tumour.
‘We feel this combination therapy could give pancreatic cancer patients much needed “hope” in their fight against the disease.’
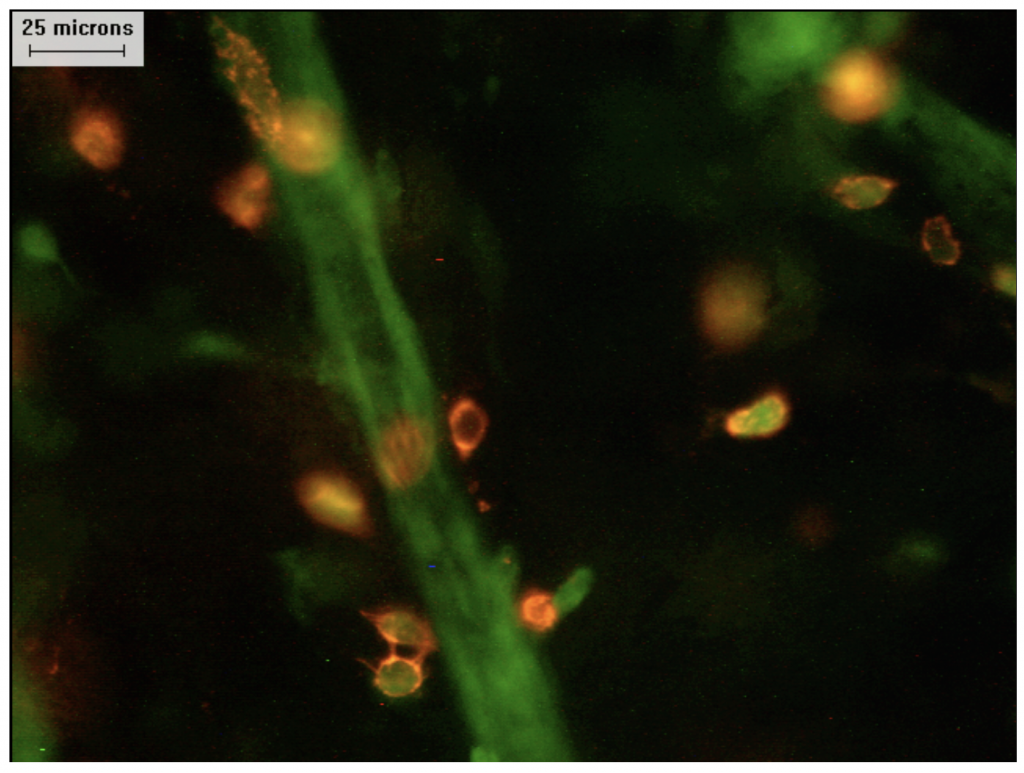
Image of anti-tumour CD8+ T cells (orange) infiltrating the tumour microenvironment (black) with tumour blood vasculature (green) Credit: Scott Gerber.
The Development of a Combination Therapy
Dr Gerber and his colleagues tested their critical prediction that the immune system effects derived by SBRT can be further enhanced by providing IL-12 directly to the tumour. In their tests, IL-12 loaded microspheres were injected directly into tumours in an animal model of pancreatic cancer, in which the IL-12 was then slowly released over a period of up to two weeks.
The team dosed the animal models with a quantity and schedule of radiation similar to the clinical SBRT schedule, using a small animal radiation research platform. In their test animals, the team demonstrated a substantial destruction in tumour mass following SBRT-like treatment.
Using animals in which tumours had been transplanted into the pancreas, the team found that following the combination therapy (SBRT + IL-12 microspheres) the tumours were undetectable at 20 days post-implantation, and this remained the case as far out as 120 days. This combined approach was superior to treatment with either IL-12 microspheres or SBRT alone. Additionally, assessment of the post-treatment tumours at day 11 of the study showed that the combination treatment resulted in the tumour being overwhelmed by immune cell infiltration.
The team tested the individual and combination therapies on a range of animal models, and overall, they found combination treatment was vastly superior to individual therapy modes. In one case, SBRT treatment resulted in 20% of test subjects having increased survival at 120 days of the study. However, with the combination therapy, 100% of the studied animals survived to this point. This is an unprecedented finding, in that the combination therapy fully eradicated the hard-to-treat PDA tumour. Of great significance is the fact that this combined therapy not only eradicated the tumour mass from a transplanted model which is known to be sensitive to radiotherapy but showed the same effect in a model which is known to be insensitive to radiotherapy.
The Immune Response to Combination Therapy
It is generally accepted that many of the functions of Il-12 are mediated through stimulation of other cytokines, and specifically, IFN-δ (interferon-gamma). IFN-δ is an important cytokine for immune responses, and a key function of this cytokine is its ability to induce a range of actions, which are vital in anti-tumour processes.
When Dr Gerber and his team analysed the cytokine profiles of tumours treated with combination therapy, they found that the expression of both Il-12 and IFN-δ was increased compared to individual treatments. These increases occurred on the day after treatment and were sustained for 24 and 48 hours, respectively. The team demonstrated the requirement for IFN-δ to induce tumour death by testing a model which was unable to produce this important cytokine, and they found that tumours in these mice did were not eradicated in the same way as in the mice which were capable of producing IFN-δ.
On assessing the cell types found within the treated tumours, the team noted that the tumours exposed to the combination treatment showed significantly higher levels of CD45+ cells, which are IFN-δ positive, and CD4 and CD8 T-cells, which can target the tumour directly or change the tumour microenvironment.
As IFN-δ is a cytokine which promotes an inflammatory response from other immune system cells, Dr Gerber and colleagues investigated the effect of the increased levels of IFN-δ on the other immune cell types within the tumour. It is known that immunosuppressive cells are increased in response to radiotherapy in PDAs, and the team predicted that the combination approach could overcome this suppression. They found that tumour-associated myeloid cells, which can be increased by SBRT, could be modified by IL-12-microsphere treatments. For example, the team demonstrated that tumour-associated myeloid cells which are immune suppressive following radiotherapy are reprogrammed following exposure to Il-12 and actually then assist in the destruction of the tumour.
Building on Pre-clinical Success
The pre-clinical work conducted by Dr Gerber and colleagues shows great promise in providing treatment for PDA, for which currently, the vast majority of patients do not survive past five-years. As Dr Gerber notes, ‘One of the key findings to our work was that the combination therapy was not only effective against the primary tumour, but was also able to destroy established metastases. This is important, as metastatic disease is what eventually kills most cancer patients.’
Looking to the future, Dr Gerber further shares, ‘We feel this combination therapy could give pancreatic cancer patients much needed “hope” in their fight against the disease.’ Now, the team are setting up clinical trials in humans to assess the effects of this potentially revolutionary treatment and to ascertain the benefits to locally-advanced and metastatic PDA patients.
Reference
https://doi.org/10.33548/SCIENTIA549
Meet the researcher

Scott Andrew Gerber, PhD
Associate Professor
University of Rochester Medical Center
Rochester, NY
USA
Dr Scott Gerber is an Associate Professor in the Department of Surgery at the University of Rochester Medical Center with over two decades of experience. After completing his PhD in Immunology at the University of Rochester, New York, in 2005, Dr Gerber undertook a three-year postdoctoral position at Yale University, before returning to University of Rochester. The majority of Dr Gerber’s research has been focused on cancer therapies, and his major interest is in overcoming tumour-induced immune suppression and enhancing the efficacy of radiotherapy treatment. His team has recently identified a highly promising combination of therapies, using a targeted radiotherapy treatment and intra-tumour injection with immune-modulating microspheres that, if successful in human trials, will result in a vast improvement in treating or extending survival, which is currently dismally low, of pancreatic cancer patients.
CONTACT
E: scott_gerber@urmc.rochester.edu
W: https://www.urmc.rochester.edu/labs/gerber.aspx
W: https://www.urmc.rochester.edu/labs/tumor-immunology.aspx
KEY COLLABORATORS
Dr David C. Linehan, MD
Dr Edith M. Lord, PhD
Dr Nejat K. Egilmez, PhD
Dr Bradley N. Mills, PhD
FUNDING
National Institutes of Health (R01CA230277)
FURTHER READING
J Ye, BN Mills, T Zhao, et al, Assessing the Magnitude of Immunogenic Cell Death Following Chemotherapy and Irradiation Reveals a New Strategy to Treat Pancreatic Cancer, Cancer Immunology Research, 2020, 8, 94–107.
HJ Han, JD Murphy, S Qin, et al, Microspheres Encapsulating Immunotherapy Agents Target the Tumor-Draining Lymph Node in Pancreatic Ductal Adenocarcinoma, Immunological Investigations, 2020 (online ahead of print). Doi:10.1080/08820139.2020.1765795.
BN Mills, KA Connolly, J Ye, et al, Stereotactic Body Radiation and Interleukin-12 Combination Therapy Eradicates Pancreatic Tumors by Repolarizing the Immune Microenvironment, Cell Reports, 2019, 29, 406–421.

Want to republish our articles?
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence
(CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
More articles you may like
Grandmothers: Innovation Through Tradition
Grandmother Project – Change through Culture (GMP) is an organisation dedicated to documenting the role of grandmothers and demonstrating the effectiveness of grandmother-inclusive strategies in improving the health and well-being of women, children, and adolescents. GMP’s groundbreaking work challenges conventional wisdom to transform community-based interventions in Africa and beyond, harnessing a powerful but often overlooked resource: the wisdom and influence of grandmothers.
Dr Robert Larkin | Cultivating Change to Improve Soil Health and Increase Potato Yield
Environmental quality and food production are facing the pressing challenges of climate change and global population growth. Dr Robert Larkin from the United States Department of Agriculture-Agricultural Research Service (USDA-ARS) and a team of plant scientists developed and tested a range of crop management systems to help overcome these compounding challenges. Their work is improving soil health and increasing the yield of potato crops, contributing to the future food security of nations.
Professor Giorgio Buttazzo | Artificial Intelligence and a Crossroads for Humanity
Where do we stand with artificial intelligence? Might machines take over our jobs? Can machines become conscious? Might we be harmed by robots? What is the future of humanity? Professor Giorgio Buttazzo of Scuola Superiore Sant’Anna is an expert in artificial intelligence and neural networks. In a recent publication, he provides considered insights into some of the most pressing questions surrounding artificial intelligence and humanity.
Dr Ralf Adam | New Technologies Shaping the Future of Oral Hygiene
Understanding the efficiency of various toothbrush technologies is essential for achieving optimal oral health. Dr Ralf Adam, who leads a dedicated team at Procter & Gamble in Germany, is keen to investigate the complexities of these technologies. His team have provided new insights into the best toothbrush types for plaque removal and the maintenance of gum health. By highlighting the importance of informed oral care decisions and ongoing investigations, this vital research works towards ensuring everyone can achieve a brighter, healthier smile.




