Dr Y. Peng Loh – Discovery of Neurotrophic Factor-α1 Reveals New Treatment Strategies for Stress-induced Neurodegenerative Diseases and Depression
Stress produces numerous negative effects on the human body. Lying deep within the brain, one particularly sensitive area is the hippocampus, where chronic exposure to stress hormones can lead to the degeneration and death of neurons. Thankfully, the brain holds defence mechanisms that block some of these negative effects. Deciphering these mechanisms with the aim of better treating neurodegenerative diseases and depression is Dr Y. Peng Loh from the Eunice Kennedy Shriver National Institute of Child Health and Human Development in the USA.
Stress on the Brain
Stress is a feeling familiar to us all, and results in the body releasing a cascade of hormones that cause physiological changes. These changes are important survival mechanisms; a quickened heart rate and reduced gastrointestinal activity help prepare the body for a fight-or-flight response. However, the modern age brings with it chronic stress, something the human body is not fully prepared for.
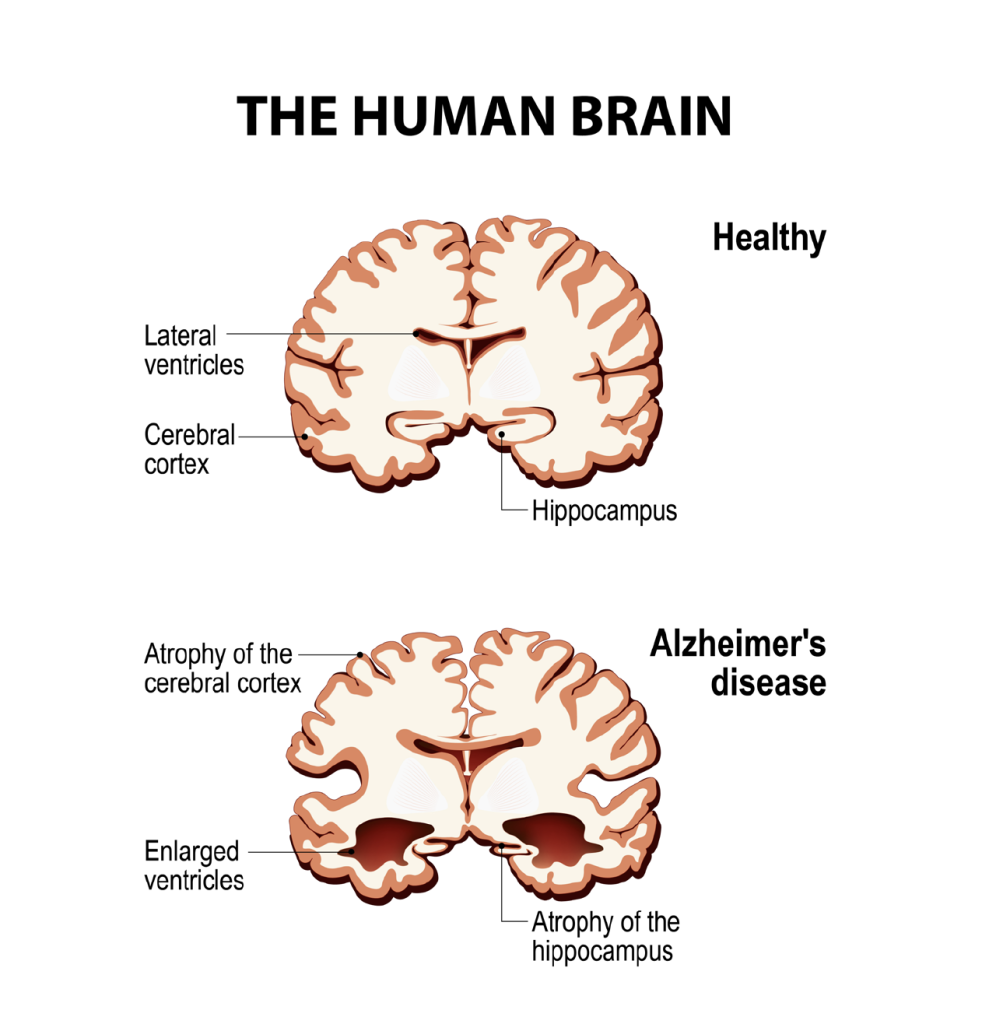
Continual activation of the stress response can lead to a number of health problems, including high blood pressure, heart attacks, and alterations to the brain that contribute to mental health issues. Fortunately, protection mechanisms are in place to prevent some damage, including damage to the stress-sensitive brain, although these are not always effective. One specific area of the brain, the hippocampus, is particularly at risk of the destructive effects of the stress hormones called glucocorticoids.
The hippocampus is a complex structure deep within the brain and it plays a major role in emotion, learning and memory, including spatial memory. As a result, when Alzheimer’s disease and other forms of dementia develop, the damage is often first seen in this area. This manifests as short-term memory loss and disorientation that worsens over time. A shrunken hippocampus is sometimes observed in patients with untreated depression, and it is implicated in this disease as well.
Investigating how the brain protects itself from stress-induced damage and how this knowledge can be used to develop treatments for dementia and depression is the goal of Dr Y. Peng Loh and her team from the Section on Cellular Neurobiology at the Eunice Kennedy Shriver National Institute of Child Health and Human Development within the National Institutes of Health, USA.
The Role of Neurotrophic Factor-α1
The early focus of Dr Loh’s work was an enzyme called carboxypeptidase E. Enzymes are proteins that catalyse biochemical reactions. Importantly, this enzyme is involved in the production of amino acid chains called neuropeptides which regulate brain activity and also the production of peptide hormones, which are involved in the body’s many different responses to its environment.
Peptide hormones are also important in helping neuron cells develop and connect with surrounding neurons for brain activity. These types of molecules are known as trophic factors or trophins. They are secreted outside the cell and bind to a cell receptor which then transduces a signal directing the cell to modify its function. In 2013, Dr Loh discovered that carboxypeptidase E is also a trophic factor that works independently of its enzyme function, leading her to coin the new name of neurotrophic factor-α1 (or NF-α1 for short). NF-α1 has been found to have profound effects in protecting hippocampal cells from oxidative stress-induced cell death. Not only has Dr Loh found that NF-α1 is important for neurodevelopment, but it is also implicated in cancer development, found in significant quantities in tumours accompanied by strong evidence it promotes tumour growth.
Clearly, the role of NF-α1 in the brain is varied and complex, and the full range of its functions is not yet well understood. Dr Loh is shining a light on a previously unknown task of this important enzyme – that of a trophin, working to prevent neuronal degeneration and death when the brain is faced with severe stress.
More Important Than Previously Thought
Trophic factors are proteins that promote cell growth and viability. For example, brain-derived neurotrophic factor (BDNF) is protective against stress and when it is reduced, neurodegeneration is observed, as seen in post-mortem examinations of Alzheimer’s patients. As such, BDNF is needed for the survival, proliferation and protection of neurons in the brain.
However, Dr Loh found that the known trophic factors did not account for the full neuroprotection of CA3 pyramidal neurons in the highly stress-sensitive hippocampus. Intrigued by this, she turned her attention to NF-α1, which is produced in high quantities by the CA3 neurons. In these groundbreaking experiments, Dr Loh used mice that were engineered to be unable to produce NF-α1.
At three weeks old, the mice were subjected to social and physical stress. Even though the mice had sufficient BDNF, this stress led to complete CA3 neuron degeneration. According to Dr Loh, this shows that NF-α1 is essential for neuroprotection during stress, as when it is missing, neurodegeneration clearly occurs. Even more importantly, she showed for the first time that NF-α1 is more critical to neuroprotection than BDNF.
In the same study, Dr Loh studied a different set of mice that were engineered not to make normal NF-α1 but a form that could not act as an enzyme for its already known functions. After stress, these mice showed no neurodegeneration and showed normal learning and memory, unlike the mice with no NF-α1 at all. This means that the protective function and activities of NF-α1 are separate from its functions as an enzyme. Protection against stress-induced degeneration protects against cognitive decline which can present as depression. Dr Loh has further solidified NF-α1 as an important protective factor by elucidating how it works on a molecular level.
The Molecular Mechanisms of NF-α1
When investigating a biological pathway such that of NF-α1 in protecting neurons, it is important to understand the minute details if the right molecules and reactions are to be targeted when the research is eventually used to innovate new therapies. For the NF-α1 pathway, such therapies may in the future include new gene therapy approaches, antidepressants or even drugs to treat Alzheimer’s disease or depression.
Dr Loh also looked into how NF-α1 works to prevent stress from destroying the cells of the hippocampus. She did this using hippocampus cells from mice and found that glucocorticoids released during stress increase NF-α1 production. NF-α1 binds specifically and strongly to these types of cells (or more precisely, to receptors on their surface). Recently, she identified the receptor to which NF-α1 binds – HTR1E, a serotonin receptor in human neurons.
When human neurons grown in Petri dishes were engineered to not express the HTR1E receptors, added NF-α1 was unable to protect these cells from dying when subjected to induced oxidative and neurotoxic stress. Once NF-α1 has bound to the receptors, a pathway called the ERK pathway is activated and the cell is signalled to increase energy metabolism. It also increases the amount of mitochondrial pro-survival protein, BCL2, both of which boost the neuron’s ability to survive.
Another mechanism that Dr Loh and her colleagues investigated in a different study is the effect of NF-α1 on a signalling protein called fibroblast growth factor 2 (FGF2). This protein is involved in the survival and replication of fibroblasts, the cells that create structure in connective tissue and aid in wound healing. This study also used mice, some of which were subjected to short-term chronic stress and others to long-term chronic stress.
The hippocampus of the mice that experienced short-term stress showed high levels of NF-α1 which was accompanied by high levels of FGF2. They hypothesised that as a direct result of these elevated levels, these mice had high neurogenesis and exhibited no depressive-like behaviours. However, the mice with long-term stress had low levels of NF-α1, FGF2 and neurogenesis and consequently, they showed clear signs of depressive behaviour. This shows that NF-α1 is protective, but prolonged stress can still cause damage to the brain.
Other mice that had been engineered to not produce NF-α1 had decreased FGF2 and neurogenesis within their hippocampus whilst also showing depressive-like behaviours. This supported the idea that NF-α1 regulates the abundance of FGF2 in the hippocampus and so, depression. In addition, once these mice were treated with FGF2, their depression symptoms were reversed.
Additional studies have demonstrated that when hippocampal neuron cells are grown in Petri dishes and treated with NF-α1, this directly upregulates FGF2 expression (more of it is made). Therefore, Dr Loh explains, when mice are subjected to short-term stress, the hippocampus makes more NF-α1 and becomes more active and has a key part to play in preventing depression through upregulating FGF2 for neurogenesis.

The Potential for New Antidepressants
To understand these findings even further, Dr Loh studied a drug that is known to have antidepressant qualities for both mice and humans – rosiglitazone (Rosi). Interestingly, the drug increased the activity of both NF-α1 and FGF2 in the hippocampus, resulting in increased neurogenesis. This provides exciting insight into the potential use of Rosi, or other drugs that act on this pathway, as antidepressants.
Dr Loh believes that NF-α1 and the neurogenesis pathways associated with it are the future for promising new therapies for depression. The proven effectiveness of targeting these and mitochondrial metabolic pathways to prevent neuron death and degeneration is a real indicator of their promise. But with that in mind, she notes that additional NF-α1 -activated signalling pathways may be involved in neurogenesis and neuroprotection should also be investigated.
Dr Loh is continuing to drive progress in her field through exploring different potential therapeutic targets and innovating novel therapies for depression and neurodegenerative diseases such as Alzheimer’s disease.
SHARE
DOWNLOAD E-BOOK
LISTEN TO THE AUDIO
REFERENCE
https://doi.org/10.33548/SCIENTIA787
MEET THE RESEARCHER

Dr Y. Peng Loh
Section on Cellular Neurobiology
Eunice Kennedy Shriver National Institute of Child Health and Human Development
National Institutes of Health
USA

Dr Vinay Sharma
Dr Lan (Ashley) Xiao

Dr Xuyu Yang
Dr Y. Peng Loh achieved her BSc in Biochemistry from University College Dublin in Ireland, followed by her PhD in Molecular Biology from the University of Pennsylvania in the USA. She carried out postdoctoral work both at the National Institutes of Health in the USA and the Max Planck Institute in Germany, and has received multiple honours and awards for her work. Currently, Dr Loh serves as the Senior Investigator and Head of the Section on Cellular Neurobiology Eunice Kennedy Shriver National Institute of Child Health and Human Development at the National Institutes of Health. Her research has led to exciting developments in our understanding of stress-induced neurodegeneration and depression, and the neuroprotective and anti-depressant mechanisms mediated by the new neurotrophic factor discovered in her laboratory, now named NF-α1.
CONTACT
W: https://www.nichd.nih.gov/research/atNICHD/Investigators/loh/research
KEY COLLABORATORS
Prof Yong Cheng, Minzu University of China, China
Prof Beata Lecka-Czernik, University of Toledo, USA
Prof William Goddard III, California Institute of Technology, USA
Prof Daniel Saiz-Sanchez, University of Castilla-La Mancha, Spain
Dr Loh thanks her team of staff, postdoctoral fellows and students, past and present, for their hard work and contribution to the studies described in this article.
FUNDING
Eunice Kennedy Shriver National Institute of Child Health and Human Development
National Institutes of Health
American Diabetes Association
Margaret Early Medical Research Trust
Spanish Ministry of Economy and Competitiveness-FEDER
Autonomous Government of Castilla-La Mancha/FEDER
FURTHER READING
VK Sharma, X Yang, S-K Kim, et al., Novel interaction between neurotrophic factor-α1/carboxypeptidase E and serotonin receptor, 5-HTR1E, protects human neurons against oxidative/neuroexcitotoxic stress via β-arrestin/ERK signaling, Cellular and Molecular Life Sciences, 2021. DOI: https://doi.org/10.1007/s00018-021-04021-3
E Bosch, M Hebebrand, B Popp, BDV Syndrome: an Emerging Syndrome With Profound Obesity and Neurodevelopmental Delay Resembling Prader-Willi Syndrome, The Journal of Clinical Endocrinology & Metabolism, 2021, dgab592. DOI: https://doi.org/10.1210/clinem/dgab592
L Xiao, VK Sharma, L Toulabi, et al, Neurotrophic factor-α1, a novel tropin is critical for the prevention of stress-induced hippocampal CA3 cell death and cognitive dysfunction in mice: comparison to BDNF, Translational Psychiatry, 2021, 11, 24. DOI: https://doi.org/10.1038/s41398-020-01112-w
A Chougule, V Kolli, S Baroi, et al., Nonenzymatic and Trophic Activities of Carboxypeptidase E Regulate Bone Mass and Bioenergetics of Skeletal Stem Cells in Mice, JMBR plus, 2020, 4(9), 10392. DOI: https://doi.org/10.1002/jbm4.10392
L Xiao, X Yang, YP Loh, Neurotrophic, Gene Regulation, and Cognitive Functions of Carboxypeptidase E-neurotrophic Factor-α1 and Its Variants, Frontiers in Neuroscience, 2019, 13, 243. DOI: https://doi.org/10.3389/fnins.2019.00243
L Ji, HT Wu, XY Qin, R Lan, Dissecting carboxypeptidase E: properties, functions and pathological roles in disease, Endocrine Connections, 2017, 6(4), R18–R38. DOI: https://doi.org/10.1530/EC-17-0020
Y Cheng, NX Cawley, T Yanik, et al., A human carboxypeptidase E/NF-α1 gene mutation in an Alzheimer’s disease patient leads to dementia and depression in mice, Translational Psychiatry, 2016, 6(12), e973. DOI: https://doi.org/10.1038/tp.2016.237
Y Cheng, RM Rodriguiz, SRK Murthy, et al., Neurotrophic factor-α1 prevents stress-induced depression through enhancement of neurogenesis and is activated by rosiglitazone, Molecular Psychiatry, 2015, 20(6), 744–754. DOI: https://doi.org/10.1038/mp.2014.136
SRK Murthy, E Thouennon, W-S Li, et al., Carboxypeptidase E Protects Hippocampal Neurons During Stress in Male Mice by Up-regulating Prosurvival BCL2 Protein Expression, Endocrinology, 2013, 154(9), 3284–3293. DOI: https://doi.org/10.1210/en.2013-1118
REPUBLISH OUR ARTICLES
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence (CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
SUBSCRIBE NOW
Follow Us
MORE ARTICLES YOU MAY LIKE
Dr Ralf Adam | New Technologies Shaping the Future of Oral Hygiene
Understanding the efficiency of various toothbrush technologies is essential for achieving optimal oral health. Dr Ralf Adam, who leads a dedicated team at Procter & Gamble in Germany, is keen to investigate the complexities of these technologies. His team have provided new insights into the best toothbrush types for plaque removal and the maintenance of gum health. By highlighting the importance of informed oral care decisions and ongoing investigations, this vital research works towards ensuring everyone can achieve a brighter, healthier smile.
Professor Alan Templeton | When Fire Sparks Ecological Opportunity and Habitat Restoration
How far would you be willing to go to save an endangered species? Would you consider burning part of a forest as a solution? As unconventional as it may sound, conservationists sometimes resort to such measures to restore lost habitats. One remarkable example is the efforts to save eastern collared lizards – and indeed the entire biological community in which they live – in the Ozarks, spearheaded by American geneticist and statistician Professor Alan Templeton of Washington University in St Louis, USA.
Dr Toby Phesse | Revealing the Mysteries of Wnt Signalling: Novel Approaches to Beating Cancer
Cancer remains a leading cause of mortality worldwide, and the need for new, more effective treatments remains an urgent challenge. Dr Toby Phesse from Cardiff University in the UK focuses on the role of the Wnt receptor found on the surface of cells and its involvement with cell communication and cancer growth, bringing fresh hopes for new therapeutic options.
Dr Vijay Reddy | The Virus World Database: An Invaluable Resource for Public Health and Healthcare
Severe viral disease presents an ongoing challenge to the health of humankind. While unparalleled developments in science and technology are improving our understanding of such viruses, this information needs to be readily accessible to researchers to ensure continued progress in public health and healthcare. Dr Vijay Reddy and his colleagues at the Hormel Institute (University of Minnesota) developed the Virus World database, an invaluable resource that details the genome, structure, and host of practically every discovered virus to date.