Dr Zi-Bing Jin – Repairing the Cornea: A New View on Novel Therapies
Damage and disease of the cornea are some of the leading causes of sight loss. This can often be remedied is through a transplant with a healthy cornea from a donor. However, donors are few and far between, so innovative solutions are required. Dr Zi-Bing Jin from the Beijing Institute of Ophthalmology in China is working on this with some exciting results. Utilising specific small molecules to control cell differentiation, he has discovered a new method for creating the necessary cells for corneal transplant, without the need of a cornea donor.
The Cornea: A Window to the World
Our eyes are complex organs that help us to decipher the world. Vision is created when light bounces off objects and enters our eye through its protective window, called the cornea. The main purpose of the cornea is to focus the light entering the eye by refracting (bending) it onto the retina at the very back of the eyeball. Pupils control how much light enters by expanding or contracting tiny muscles that are constantly working. Images reach the retina upside down because of the curved nature of the eyeball, but this is processed by the brain later on. On the surface of the retina, millions of photoreceptor cells detect the qualities of the light entering. Cone cells allow us to see in colour and detail and are responsible for daylight vision, but rod cells are not sensitive to colour so are only utilised for night vision. All of this information is sent through the optic nerve to the brain, where what we see is compiled and given meaning.
Unusually for the human body, the tissue of the cornea does not contain any blood vessels for nutrient supply but is entirely made up of proteins and cells. This is because it requires complete transparency for clear vision, so nutrients must be distributed in a different way. Oxygen is supplied to the cornea dissolved in tears and then it diffuses throughout the structure to maintain its health. In a similar manner, nutrients are also transported by tear fluid and diffuse from the outside of the cornea, through a special fluid called the aqueous humour, and eventually reach the inside.
The cornea is comprised of five different layers, the outermost being a layer of cells called the epithelium that protects the eye and absorbs nutrients and oxygen. The innermost layer is called the corneal endothelium and unlike the epithelium that can heal itself relatively quickly, the endothelium cannot regenerate. Instead of producing new cells to replace dead ones, endothelial cells stretch to fill the gaps. However, this makes the layer much less dense and less able to regulate the fluid that is present in the cornea. Therefore, damage to the cornea can create excess fluid build-up, which makes it less transparent and causes eyesight to be impaired.
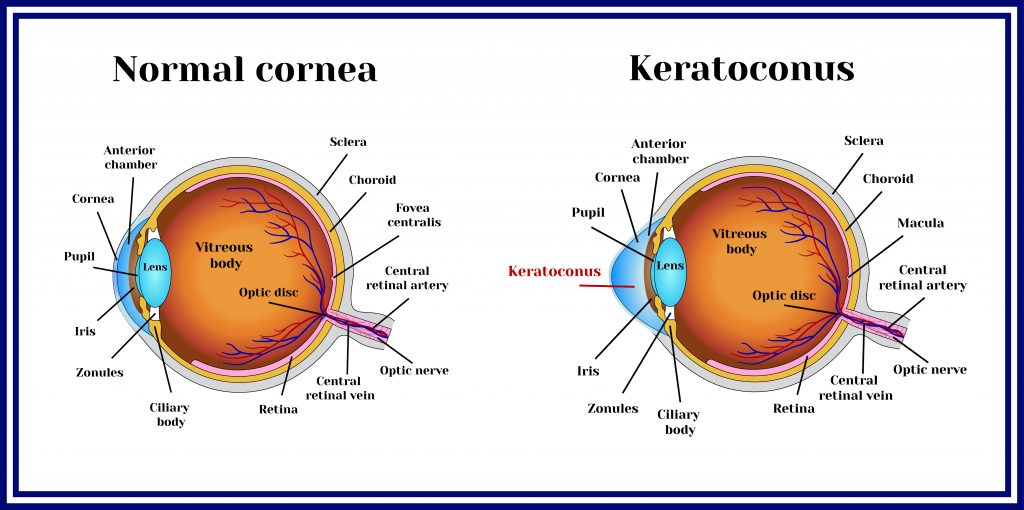
Unfortunately, this is common in old age and in certain diseases. In fact, corneal disorders are the number one cause of blindness globally and they can come in many forms. For example, corneal abrasion is the loss of the epithelium due to trauma and keratitis is caused by the inflammation of the cornea. Keratoconus is a degenerative disease uncommonly prevalent in young adults that causes the cornea to thin and become more coned in shape. These each come with their own complications and difficulties, but dedicated research is targeted at combatting them in their own way.
Reprogramming Cells for Corneal Transplantation
Thanks to innovative modern medicine, there are some solutions for those suffering from corneal disorders. Replacing a damaged cornea with a healthy one from a deceased donor through corneal transplantation is the most common therapy. It is thought around half of these procedures are to replace the endothelium of the cornea. Nevertheless, a huge shortage of donated corneas means that fewer than 1.5% of patients who require a corneal transplant are able to receive them from a donor.
However, progress is being made in this field and corneal endothelial cells have been grown from healthy donor tissues and successfully transplanted into models. But this method is still in its early stages and it is difficult to grow a sufficient number of cells for the procedure. Through a method called direct lineage reprogramming, Dr Zi-Bing Jin from the Beijing Institute of Ophthalmology in China is helping to push forward new and exciting techniques for corneal regeneration.
The lineage of a cell is like its family history – what types of cells it differentiated (split and specialised) from to get to its current form and function. In the case of corneal endothelial cells, they originate from neural crest cells, which are able to differentiate into lots of different types of cells in the eye as well as important cells of the nervous system, like glia and neurons.
During the process of lineage reprogramming, the normal route of cell differentiation is altered so that desired and useful cells can be generated from a cell that would not ordinarily produce them. For corneal endothelial cells, this means that elusive donor neural crest cells aren’t necessary and other terminally differentiated cells can be utilised. Terminally differentiated refers to cells that have left the cycle of replication and are carrying out their specialised function. If successful, lineage reprogramming could mean that more cell replacement therapies are possible for people with damaged and diseased corneas.
Transcription Factors and Small Molecules
Dr Jin’s work focuses on targeting transcription factors. These are proteins that help to control which genes are turned on and off and therefore, can determine what type of cell is produced. Lineage-specific transcription factors have successfully been used to convert terminally differentiated cells into another cell type in test-tube experiments (in vitro). In tests on tiny organisms (in vivo), an abundance of lineage transcription factors has also been shown to create these conversions.
One way that these transcription factors can be controlled is by using small molecules, which are exactly what they sound like! They are molecules around 1 nanometer in size which aid the regulation of many biological processes; many drugs are classified as small molecules. Recently, they have been used to influence transcription factors to convert connective tissue cells called fibroblasts into other functional cells.
In a novel study, Dr Jin and his colleagues have further developed this method to define specific small molecules that may be applied to therapeutics in the future.
Exciting Results from Diseased Corneal Experiments
Initially, the team screened a multitude of small molecules and eventually found a cocktail that could generate chemically induced neural crest cells from mice fibroblasts, through lineage reprogramming. They found that these chemically induced cells had many of the same features as naturally produced neural crest cells, including which genes they used for protein production (gene expression), their capacity for self-renewal and their differentiation potential. Importantly, they retained the ability to differentiate into corneal endothelial cells. To encourage this second differentiation, Dr Jin had to uncover another mixture of small molecules for the task, which they were successful in finding.
The subsequent chemically induced corneal endothelial cells were also remarkably similar to naturally differentiated ones. Their gene expression, characteristics and function were all alike to primary corneal endothelial cells. This two-step lineage reprogramming strategy is especially exciting as Dr Jin and the team went on to demonstrate it working in a real-world situation.
After reprogramming cells and growing the tissue of chemically induced corneal endothelial cells, they transplanted it into an animal model. They used rabbits that had corneal endothelial disease, which had resulted in bullous keratopathy. Whereas a healthy endothelium layer prevents excessive fluid absorption in the cornea by pumping out liquid, a damaged or diseased one carries out this role ineffectively. As a result, fluid-filled blisters, called bullae, build up on the surface (epithelium) of the cornea and cause the swelling and often pain associated with bullous keratopathy.
Interestingly, the diseased rabbits who were given transplants from these chemically induced cells experienced an almost complete reversal of their corneal opacity. They eventually exhibited clear tissue on their cornea, meaning their sight was restored and the transplant was successful.

Implications for Real-world Application
The results produced by Dr Jin are an exciting step towards improved therapies for damaged corneas and sight loss. He believes that his work could provide a real alternative to relying on donor corneal tissues for transplantation. Fibroblasts could be a new cell source for regeneration and engineering of corneal endothelia and maybe even other tissues that originate from neural crest cells. The small molecules that Dr Jin and his team have identified as integral to this strategy will be incredibly useful for future research on this subject.
The research of Dr Jin and his colleagues at the Beijing Institute of Ophthalmology could go on to make a significant difference in the lives of real people as it is developed further to integrate into therapeutics. By introducing methods to repair corneas, countless people could have their eyesight and quality of life restored.
Reference
https://doi.org/10.33548/SCIENTIA711
Meet the researcher

Dr Zi-Bing Jin
Beijing Institute of Ophthalmology
Beijing
China
Dr Zi-Bing Jin is the director of Beijing Institute of Ophthalmology, Full Professor of Capital Medical University (CMU) and Chief Physician at Beijing Tongren Hospital, CMU. Dr Jin focuses on stem cell translational medicine in retinal health and the genetic mechanisms of ocular diseases. His team is dedicated to elucidating the disease mechanisms of inherited retinal degeneration and childhood ocular disorders, translating laboratory technology to improve bedside outcomes, and solving key problems around retinal degeneration. He also focuses on cell reprogramming and transdifferentiation, and this work has led to an exciting new understanding of the mechanisms behind corneal regeneration.
CONTACT
Post: Dr Zi-Bing Jin, Beijing Institute of Ophthalmology, Beijing Tongren Eye Center, Beijing Tongren Hospital, Capital Medical University, Beijing Ophthalmology and Visual Sciences Key Laboratory, 100730 Beijing, China
E: jinzibing@foxmail.com
KEY COLLABORATORS
Beijing Advanced Innovation Center for Big Data-Based Precision Medicine
FURTHER READING
SH Pan, N Zhao, X Feng, et al., Conversion of Mouse Embryonic Fibroblasts into Neural Crest Cells and Functional Corneal Endothelia by Defined Small Molecules, Science Advances, 2021, 4(7), eabg5749.

Want to republish our articles?
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence
(CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
More articles you may like
Grandmothers: Innovation Through Tradition
Grandmother Project – Change through Culture (GMP) is an organisation dedicated to documenting the role of grandmothers and demonstrating the effectiveness of grandmother-inclusive strategies in improving the health and well-being of women, children, and adolescents. GMP’s groundbreaking work challenges conventional wisdom to transform community-based interventions in Africa and beyond, harnessing a powerful but often overlooked resource: the wisdom and influence of grandmothers.
Dr Robert Larkin | Cultivating Change to Improve Soil Health and Increase Potato Yield
Environmental quality and food production are facing the pressing challenges of climate change and global population growth. Dr Robert Larkin from the United States Department of Agriculture-Agricultural Research Service (USDA-ARS) and a team of plant scientists developed and tested a range of crop management systems to help overcome these compounding challenges. Their work is improving soil health and increasing the yield of potato crops, contributing to the future food security of nations.
Professor Giorgio Buttazzo | Artificial Intelligence and a Crossroads for Humanity
Where do we stand with artificial intelligence? Might machines take over our jobs? Can machines become conscious? Might we be harmed by robots? What is the future of humanity? Professor Giorgio Buttazzo of Scuola Superiore Sant’Anna is an expert in artificial intelligence and neural networks. In a recent publication, he provides considered insights into some of the most pressing questions surrounding artificial intelligence and humanity.
Dr Ralf Adam | New Technologies Shaping the Future of Oral Hygiene
Understanding the efficiency of various toothbrush technologies is essential for achieving optimal oral health. Dr Ralf Adam, who leads a dedicated team at Procter & Gamble in Germany, is keen to investigate the complexities of these technologies. His team have provided new insights into the best toothbrush types for plaque removal and the maintenance of gum health. By highlighting the importance of informed oral care decisions and ongoing investigations, this vital research works towards ensuring everyone can achieve a brighter, healthier smile.




