Professor Masahito Oh-e – Intriguing Molecular Discoveries in Display Materials
It is a widely-accepted scientific fact that the motions of molecules increase as their temperature rises. However, Dr Masahito Oh-e at the National Tsing Hua University in Taiwan has recently made a counterintuitive discovery in an organic semiconductor called ‘CADN’. Within a thin film of this material, his team has found that the motion of one part of the CADN molecule increases, while another part becomes more ordered as the temperature increases and approaches the material’s phase transition. This research is scientifically intriguing, but also has profound implications for improving display technologies based on organic semiconductor materials.
Indispensable Display Technology
Human society has become increasingly dependent upon display technologies, which are an indispensable part of our smartphones, televisions, tablets and computer monitors. During the COVID-19 pandemic, such technologies have played an even more prominent role in our daily lives, allowing us to work, socialise, learn and attend cultural events, all from the safety of our homes.
Thankfully, these technologies have improved dramatically in recent years, in part due to the variety of materials that can be used to produce displays. Liquid crystals are one such group of materials, which have properties between those of a liquid and those of a solid crystal. These materials form the basis of liquid crystal displays (LCDs), which are commonly used in all kinds of devices.
Dr Masahito Oh-e at the National Tsing Hua University in Taiwan is one of the inventors of in-plane switching LCDs. This technology involves arranging and switching the orientation of molecules within liquid crystals, in order to achieve vibrant colour from all viewing angles. Dr Oh-e also contributed to developing ultra-broad-angle LCD screens, which are now an industry standard, being used in numerous common devices including iPhones. His vast experience in this area has allowed him to deeply investigate the properties of these materials, to find ways of making them even more efficient and high-performing.

Exploring Molecular Orientation
Dr Oh-e is particularly interested in studying the orientation of molecules within liquid crystals. LCD technology relies on a layer of molecules in a liquid crystal state, which are sandwiched between two substrates with electrodes. These molecules manipulate the light that passes through them, which is then controlled in a way that produces an image on a screen.
Energy efficiency, and the quality of image produced, are both influenced by the way in which molecules arrange themselves. Therefore, it is important to understand how these molecules arrange themselves and what affects their orientation.
OLEDs: An Exciting Alternative
Although liquid crystals remain the most commonly used material in current display technologies, organic light-emitting-diodes (OLEDs) are another technology that can produce vibrant, flexible and inexpensive displays. OLEDs are composed of films of an organic semiconductor – special materials comprising mainly carbon and hydrogen atoms, whose behaviour lies somewhere in between that of an electrical conductor and that of an insulator.
When an electrical current is passed through an OLED, it emits light of a certain colour. By making up a matrix of these components, scientists have developed high-quality displays that produce vibrant images. OLED technology has been making its way into many of our devices over the years, including televisions, smartphones, tablets and laptops.
Because of their low cost and flexibility, it’s easy to see why research into organic semiconductors is so important. Therefore, scientists across the globe are studying their properties in detail, to identify ways that they can be optimised for various device applications. Alongside their research into liquid crystals, Dr Oh-e and his team have also been studying molecular orientation in organic semiconductors for over a decade.
Much like with liquid crystals, the properties of organic semiconductor films can be heavily influenced by the way in which their constituent molecules are oriented. Differences in molecular orientation can have an impact on the behaviour of these films, and the properties of the resulting electronic device. For instance, molecular orientation in OLED films can affect device lifespan and energy efficiency, so it is important that scientists understand what controls orientation, and how it can be tuned. However, these films can be made up of molecules with complex shapes, making their orientation difficult to control.
Layers of organic semiconducting material
A Cross-Shaped Semiconductor Molecule
Dr Oh-e and his team employ a uniquely shaped organic molecule called CADN in order to investigate molecular orientation and alignment in semiconductor films. CADN is a cross-shaped molecule, with different electronic properties across its different branches. Its unique shape makes it a good candidate for studying orientation, as it is relatively easy to determine which way it is facing.
In a recent study, the team first experimented by preparing films of CADN, using a technique called vacuum vapor deposition, which is practically adopted in the manufacturing process of OLEDs. Vapour deposition involves preparing a surface made from a silicon wafer and applying a thin coating of the organic material in question, while under a vacuum. The benefit of this technique is that it allows for the creation of evenly coated films with a defined thickness, while creating a stable non-crystalline state.
Once the team had produced CADN films with a thickness of 100 nanometres ( just one ten-thousandth of a millimetre), they set to work analysing them. Using a technique called variable-angle spectroscopic ellipsometry, Dr Oh-e and his team were able to monitor how the molecules were orienting themselves with respect to the surface.
The team took measurements as they varied the temperature from room temperature up the material’s phase transition temperature, beyond which the material becomes a so-called ‘supercooled liquid’. This means that the film was still below its melting point but was able to behave more like a liquid.
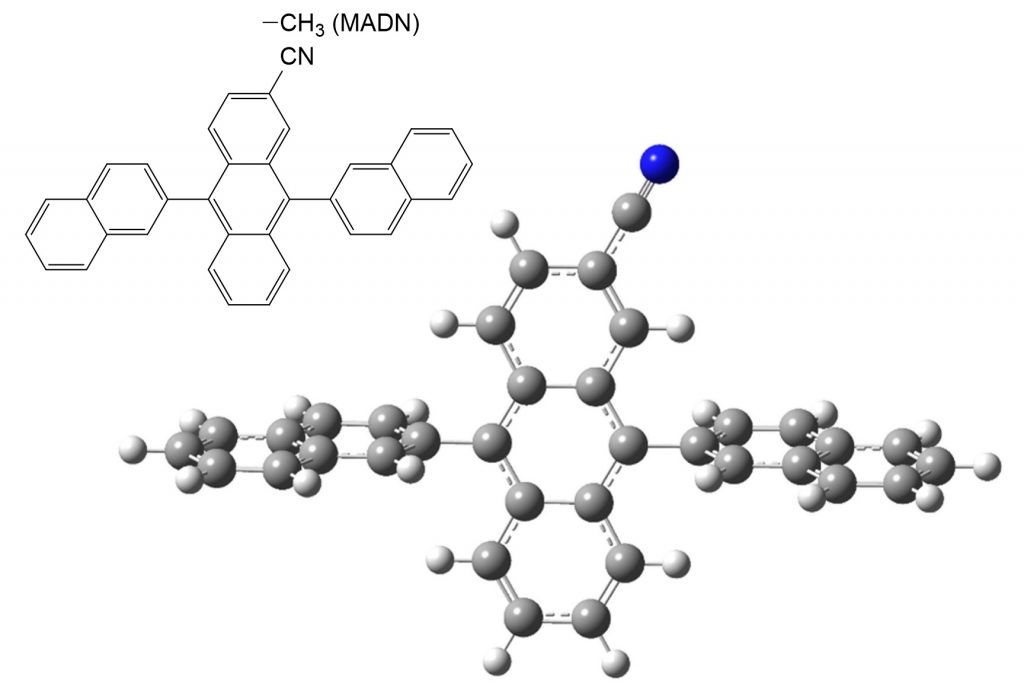
The cross-shaped CADN molecule.
An Interesting Discovery
Upon analysing the data they had collected, the team found that the molecules were aligned to one another throughout the film. As cyano groups in the molecules pull electrons towards them, causing each molecule to be electrically polarised, the team measured a large polarisation effect in the bulk material.
The cross-shaped CADN molecule can be perceived as consisting of two intersecting bars. Dr Oh-e and his colleagues found that one bar of the molecule preferentially aligned parallel to the surface of the silicon wafer, while the other bar was oriented close to the so-called ‘magic angle’ (about 54.7°) from the surface. As the temperature rose, the motion of the latter bar increased, causing its orientation to become even more random. This was unsurprising, as when the temperature of a material increases, the motions of its constituent molecules also increase.
However, the team was surprised to discover that the bar that was preferentially aligned parallel to the surface behaved in an unusual manner when the temperature increased. Although the rising temperature first caused the orientation of this bar to become disordered, as the temperature continued to rise and approached the phase transition, its parallel alignment to the silicon surface became even more orderly.
This discovery is highly counterintuitive, as one would expect that the increased heat energy would cause all parts of the molecule to move more freely, leading to random orientations. This surprising finding was very carefully observed while measuring the sample and analysing the data. Interestingly, such behaviour has not been observed in other organic semiconductor materials.
‘We observed that part of the molecule becomes more randomised while another becomes more constrained as the temperature increases,’ says Dr Oh-e. ‘This observation of molecular orientation is significantly striking and interesting.’
This observation was supported through the use of another technique, called second-harmonic generation analysis. Using this method, the team was able to gather even more data on how the orientation of the CADN molecules changes while the temperature increases, based on the different properties of each bar of the cross-shaped molecule. The data collected here consistently supported the previous conclusions, coming together to make a strong case for their observation.
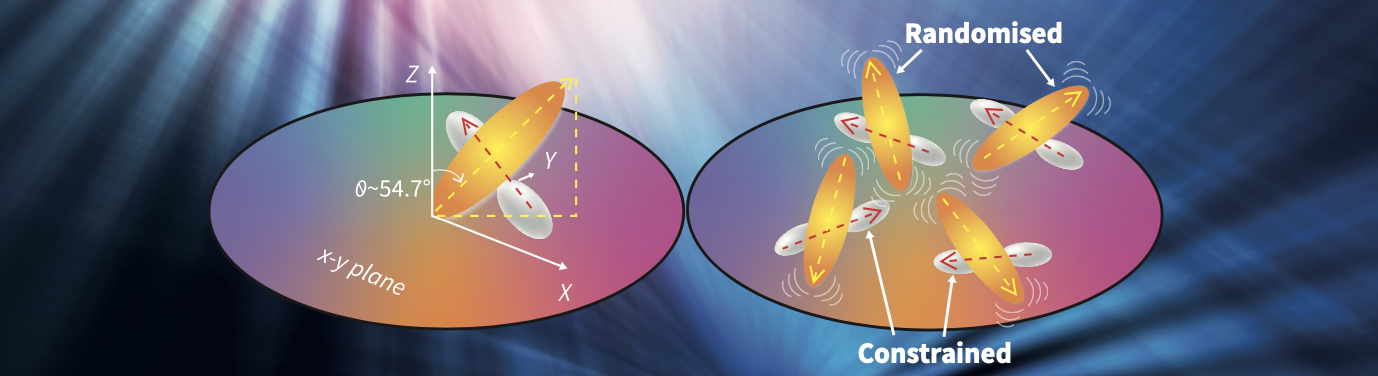
Orientation of a CADN molecule at room temperature (left), and changes in the molecular orientation of CADN
with increasing temperature (right).
The Bigger Picture
Through this study, the team gained important insights into the behaviour of molecules that comprise organic semiconducting films, wherein one part of a molecule is disordered while another becomes well-ordered with increasing temperature. ‘To have a solid picture, we need further efforts to collect more data including other molecules by independently probing two different moieties of a molecule,’ says Dr Oh-e.
They have also provided insights into how molecular alignment of CADN films can be manipulated, through simply heating the material to near its phase transition temperature. This was not the first time either; a previous study by Dr Oh-e used a similar molecule to CADN, known as ‘MADN’. The results of the two studies were consistent with one another, further supporting the case for how these uniquely shaped molecules prefer to orient themselves with respect to the silicon surface they are attached to.
This information is vital to understand when it comes to designing and fabricating organic semiconductors for any application. It is known that molecular alignment in a thin film semiconductor can affect its conductivity, ability to produce and absorb light, and efficiency. In Dr Oh-e’s MADN study, he showed that when one bar of each molecule was well aligned with the silicon surface, the conductivity of the film was improved. This is also likely the case with CADN, as this molecule is very similar in structure to MADN.
On top of this, understanding how to control molecular orientation in organic semiconductors and liquid crystals alike opens a whole world of potential applications, as it means that they will be more customisable than ever before.
Dr Oh-e and his team have conducted research that will change the way that we look at both liquid crystals and organic semiconductors, and will hopefully go on to improve the quality and variety of applications that we use them for.
Reference
https://doi.org/10.33548/SCIENTIA690
Meet the researcher

Professor Masahito Oh-e
Institute of Photonics Technologies
Department of Electrical Engineering
National Tsing Hua University
Hsinchu City
Taiwan
Dr Masahito Oh-e earned his master’s degree from the Tokyo Institute of Technology, after which he worked as a research scientist at the Hitachi Research Laboratory of Hitachi, Ltd. He then went on to gain his PhD from the Tokyo Institute of Technology in 1998, before becoming a visiting research fellow at the University of California, Berkeley. Continuing to work with Hitachi, Dr Oh-e worked with the Japanese government on several projects involving nano-structured liquid crystals between 2002 and 2008, before being offered a position as the department director and chief technical research fellow of the Sharp Labs of Tokyo. As of 2016, he has worked as a professor at the Institute of Photonics Technologies at the National Tsing Hua University, Taiwan. Here, his research focuses on fundamentally exploring multidisciplinary photonics and materials science, and addressing current technical challenges including optoelectronic applications.
CONTACT
E: oh-e@ee.nthu.edu.tw
W: http://www.ee.nthu.edu.tw/oh-e/
FUNDING
Ministry of Science and Technology (MOST), Taiwan
MOST and Profound Material Technology Co., Ltd., Taiwan
KEY COLLABORATORS
Mr Hidenori Ogata, Institute of Photonics Technologies, National Tsing Hua University
Dr Fumito Araoka, RIKEN Center for Emergent Matter Science
FURTHER READING
M Oh-e, A Nagasawa, Advancing the a Posteriori Quest for Deep-Blue Phosphorescence: Quantifying Excitation-Induced Metal-to-Ligand Charge Transfer as a Guiding Indicator, Organometallics, 2020, 39, 3951. https://doi.org /10.1021/acs.organomet.0c00506
M Oh-e, A Nagasawa, Interactions Between Hydrated Cerium(III) Cations and Carboxylates in an Aqueous Solution: Anomalously Strong Complex Formation with Diglycolate, Suggesting a Chelate Effect, ACS Omega, 2020, 5, 31880. https://doi.org /10.1021/acsomega.0c04724
M Oh-e, H Ogata, F Araoka, Randomization and Constraint of Molecular Alignment and Orientation: Temperature-Dependent Anisotropy and Phase Transition in Vapor-Deposited Thin Films of an Organic Cross-Shaped Molecule, ACS Omega, 2019, 4, 39.https://doi.org /10.1021/acsomega.8b02560
F Araoka, M Isoda, D Miyajima, I Seo, M Oh-e, T Aida, H Takezoe, Polar Dynamics at a Functional Group Level: InfraredVisible Sum-Frequency Generation Study on Polar Columnar Liquid Crystals, Advanced Electronic Materials, 2017, 3, 1600503. https://doi.org /10.1002/aelm.201600503
DR Evans, HS Kwak, DJ Giesen, A Goldberg, MD Halls, M Oh-e, Estimation of charge carrier mobility in amorphous organic materials using percolation corrected random-walk model, Organic Electronics, 2016, 29, 50. http://dx.doi.org /10.1016%2Fj.orgel.2015.11.021
M Oh-e, H Ogata, Y Fujita, M Koden, Anisotropy in amorphous films of cross-shaped molecules with an accompanying effect on carrier mobility: Ellipsometric and sum-frequency vibrational spectroscopic studies, Applied Physics Letters, 2013, 102, 101905. https://doi.org /10.1063/1.4792746
Oh-e, H Yokoyama, Sum-Frequency Vibrational Spectroscopy of a Helically Structured Conjugated Polymer, Physical Review Letters, 2004, 93, 267402. https://doi.org /10.1103/PhysRevLett.93.267402
Oh-e, H Yokoyama, Mapping molecular conformation and orientation of polyimide surfaces for homeotropic liquid crystal alignment by nonlinear optical spectroscopy, Physical Review E, 2004, 69, 051705. https://doi.org /10.1103/PhysRevE.69.051705

Want to republish our articles?
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence
(CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
More articles you may like
Alex Spezowka | Responsible Research Writing in the Age of AI: From Detection to Transparency
Artificial intelligence is often discussed as a future challenge for research, yet it is already shaping how many papers are written. Drawing on emerging evidence and recent analysis, work led by Alex Spezowka highlights a key shift in thinking: rather than trying to detect AI use, the focus is moving towards how it can be used responsibly. This has important implications for how research is produced, evaluated, and trusted.
Laura Daniela Muñoz Duarte – Jo Philips | Rethinking Hydrogen Use in Acetogenic Bacteria: Kinetics, Thresholds, and Biotechnological Potential
Acetogenic bacteria can convert carbon dioxide and hydrogen into useful products, making them promising tools for sustainable biotechnology. Research led by Laura Muñoz and Jo Philips reveals that hydrogen consumption follows a simpler kinetic pattern than previously assumed, while also uncovering major differences between species in how efficiently they operate at low hydrogen levels. Together, these findings provide a clearer framework for selecting and optimising acetogens for industrial applications.
Dr Lynn George | A New Link Between Elongator Dysfunction and TDP-43 Loss in ALS
At Montana State University Billings, Dr Lynn George and her team are investigating the cellular mechanisms underlying amyotrophic lateral sclerosis (ALS). Using a new mouse model, their research reveals that disruption of the Elongator complex leads to nucleolar stress and the loss of the protein TDP-43 from motor neurons. These findings connect two major hallmarks of ALS and offer new insight into how the disease develops.
Dr David Hansen – Abhishek Juneja | Coming of Age: Active Balancing in Adolescence
Adolescence, the transitional stage between childhood and adulthood, comes with challenges as well opportunities for growth and learning.




