Professor Matthew Bogyo – Improving Cancer Surgery Through Enzyme-activated Fluorescent Probes
Cancer is the second cause of death worldwide, despite continuous research efforts in the pursuit of better treatments. One of the most promising developments is that of cancer imaging, which aims to help clinicians visualize tumors within the body. Professor Matthew Bogyo and his team from Stanford University have developed fluorescent probes that can be injected into patients prior to cancer surgery. The probes emit fluorescence once in the tumor microenvironment, helping surgeons to distinguish cancer tissue from the surrounding healthy tissue to enable complete removal of the cancer and ultimately, improve patient outcomes.
Visualizing the Enemy
With a global death toll exceeding 9 million a year, cancer is the second leading cause of death worldwide. The World Health Organization estimates that globally, 1 in 6 deaths are due to cancer. This results in significant economic impact on healthcare budgets around the world, and of course, hugely detrimental impacts on the social and emotional wellbeing of patients and their families.
Thanks to advances in research, several treatment approaches are available in the effort to manage cancer, including chemotherapy, immunotherapy, and other forms of targeted drug treatments. Although any of these could serve as a primary cancer intervention, surgery is currently the most common for virtually all types of solid tumors.
The overall success of surgical intervention is dictated by the extent to which all cancer cells can be effectively removed from the affected organ while sparing as much of the surrounding healthy tissue as possible. Excess removal of tissue often leads to significant complications, while incomplete removal leads to increased recurrence rates (in the region of 30–65%) and the need for repeated surgeries (20–50% in breast cancers), causing considerable stress and anguish among patients.
Cancer imaging aims to help clinicians visualize tumors within the body, enabling both selective and sensitive detection of the tumor boundaries through the use of imaging contrast agents which have the potential to positively impact surgical treatment outcomes of many types of cancer.
Professor Matthew Bogyo and his colleagues at Stanford University have successfully developed and optimized the use of molecular probes that allow the real-time visualization of the tumor margins, enabling surgeons to achieve the complete removal of cancer tissue, thus minimizing the risks of cancer reoccurrence and metastasis.
A Growing Cancer Tumor Spreading
Enhancing the Selectivity of Contrast Agents
Despite the many advantages of fluorescence-guided surgeries (FGS), there are currently no Food and Drug Administration (FDA) approved optical contrast agents. The current challenge to this is achieving sufficient selectivity of contrast agents, as many of the established cancer targets are expressed in both healthy and tumor tissues.
To selectively target cancer cells, Professor Bogyo and his team have focused on developing contrast agents that produce fluorescence signals only within the tumor microenvironment. They achieved this by engineering smart probes that can only emit fluorescence when processed and cleaved by proteases that are highly expressed in tumor tissues.
Proteases are enzymes responsible for breaking down proteins into smaller molecules. Increased levels of one family of proteases, the cysteine cathepsins, can be found in tumors and are associated with growth and metastasis. This family of enzymes is also present in other pathologies such as inflammation and arthritis. Therefore, the contrast agents being developed in the Bogyo Lab could also be used for the detection and treatment of other diseases.
In 2005, Professor Bogyo and his team demonstrated for the first time that cathepsin-targeted fluorescent probes are effective in mouse models of cancer using non-invasive imaging methods. Then in 2015, they further reported on the development and optimization of a series of cathepsin-fluorogenic probes compatible with existing clinical instrumentation for use in FGS, demonstrating that they are suitable for the detection of diverse cancer types including breast, colon, and lung tumors using surgical robots.
Coming up with the optimal smart probes was not an easy task. Professor Bogyo and his team tested various molecular structures to facilitate the enzyme-assisted cleaving of the fluorophore. After numerous attempts, they developed a final, optimized probe, named 6QC-NIR, which was administered in a mouse model of breast cancer. The probe provided sufficient contrast to differentiate the tumor from normal surrounding tissues in less than 1 hour after administration. This facilitated the surgical removal of both primary and secondary tumors, which were buried deep within the tumor bed.
Having established the 6QC-NIR probe, Professor Bogyo’s team continued to look for ways to improve the contrast agents and their detection capabilities. In a paper published in 2017, Professor Bogyo’s team reported on the contrast capabilities of another fluorophore, indocyanine green (ICG), which they used in the development of the 6QC-ICG smart probe to better suit the capabilities of clinical imaging systems. Their studies in mice showed that the 6QC-ICG probe resulted in an overall brighter signal compared to the one generated by the previously developed 6QC-NIR probe.
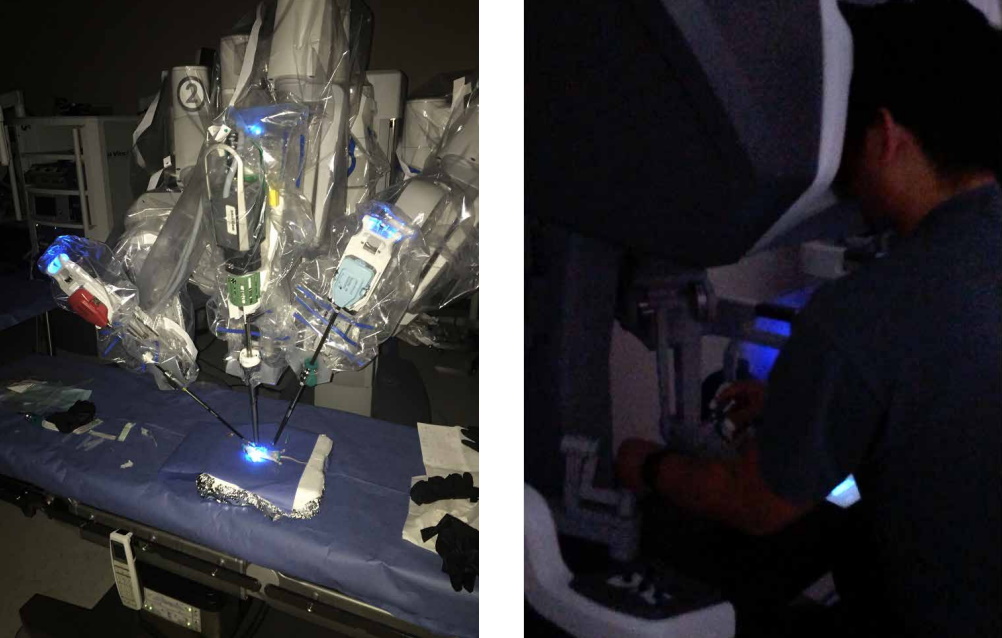
Optical Imaging During Surgery. Credit Matthew Bogyo.
AND-gate Contrast Agents
The single greatest challenge for the use of protease substrates as smart probes is optimizing their selectivity. The problem is there is a need to make sure the probes are only processed by the intended protease but unfortunately, many proteases are found both in healthy tissue and within the tumor microenvironment. This makes it challenging to generate a signal only in the tumor tissue and not in the surrounding normal healthy tissues.
To solve this problem, Professor Bogyo and his team further developed imaging probes using an ‘AND-gate’ strategy in which multiple protease activities must be present to activate the probe. The term ‘AND-gate’ is a Boolean logic element commonly used in computer science. It represents a system that requires at least two inputs to produce a single output – in this specific case, the generation of fluorescence.
AND-gate probes represent a significant advance because they produce an optical signal only when multiple reporters are processed within the tumor microenvironment, rather than a single proteolytic event by a single protease. This results in greatly increased contrast over healthy tissue when compared with single-parameter probes.
The first AND-gate probe, named DEATH-CAT-1, used substrates that are cleaved by cysteine cathepsins and caspases proteases that regulate cell death – thus the name DEATH-CAT. Although all tissues contain some level of active cathepsins, caspases are activated only during the late stage of apoptosis, a process of programmed cell death, which typically does not take place in healthy tissues. However, cell death is triggered within tumors as the result of events such as nutrient and oxygen starvation. This ‘two-step enzyme verification’ reduces background signals in the healthy normal tissues.
Professor Bogyo and his team changed the probe’s linker to resist cleavage by proteases other than the two primary target enzymes in the tumor microenvironment. They also replaced the ICG dye with FNIR dye to prevent aggregation and increase water solubility. The new DEATH-CAT-FNIR probe had fast activation, an overall high signal, good stability, and was able to locate cancer lesions with a diameter of less than 1 mm.
Overall, the data presented by the team confirm the value of the AND-gate approach, which significantly improves the specificity and sensitivity of optical probes. Critically, AND-gate probes will enable surgeons to visualize metastatic lesions in real time during FGS, improving patient outcomes and preventing removal of healthy tissue, while at the same time decreasing the risk of relapse due to incomplete removal of tumor tissue.
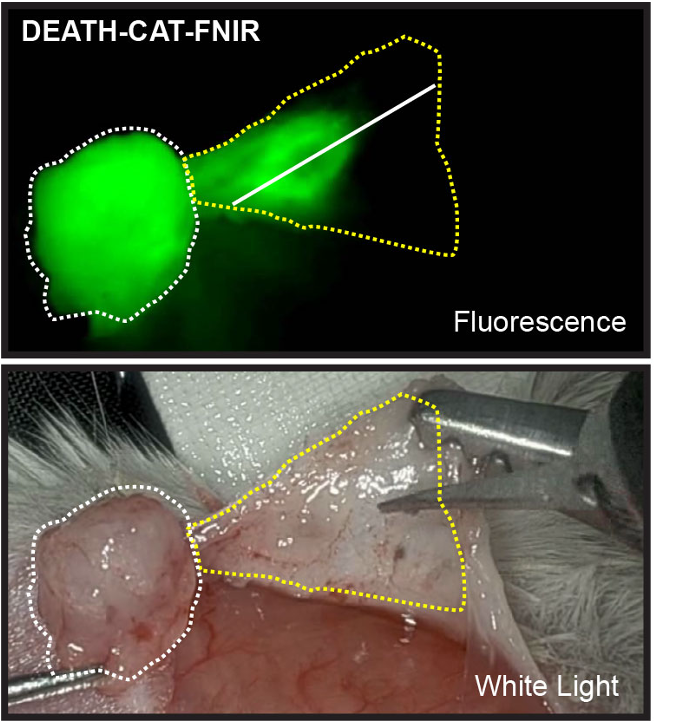
Highlighting Residual Tumor Cells After Resection.
Credit Matthew Bogyo.
Future Directions
Professor Bogyo and his colleagues continue to expand their work, fine-tuning probes to make them more widely applicable. In their latest research, they have shown that it is possible to accelerate and enhance the process of probe design by the direct screening of diverse substrate libraries obtained directly from diseased tissue extracts.
Professor Bogyo and his team have recently screened libraries of protease substrates directly in mouse breast cancer tissues to identify the optimal peptide sequences to incorporate into the design of next generation optical probes. This approach has resulted in new probes with increased overall signals in tumor tissues. These probes are not only brighter but are able to distinguish human breast cancer from adjacent mammary tissue. Critically, this suggests that it should be possible to use tissue from relevant animal models or even human biopsy tissue to build optimal smart-optical probes for use in specific types of human cancers.
In principle, this approach should work for the identification of optimal substrates for any enzyme that is active during given pathological processes. Therefore, this strategy could be applied to any diseases involving inflammation, such as fibrosis, arthritis, and osteoporosis, where contrast agents could benefit disease management. A similar strategy could also be used to target pathogen-derived enzymes for imaging infectious disease. In fact, the Bogyo Lab is currently developing new types of protease substrates that can be used for diagnostics and imaging of important human infections such as Mycobacterium tuberculosis.
The current cancer imaging probes are now entering human clinical trials with the help of several companies. Professor Bogyo aims to obtain FDA approval for the smart fluorescent probes so they can be authorized for common use in cancer surgery. By integrating the probes with surgical camera systems, cancer can now be visualized in real time during surgery at the flick of a switch, enabling clinicians to dramatically improve treatment outcomes.
SHARE
DOWNLOAD E-BOOK
REFERENCE
https://doi.org/10.33548/SCIENTIA753
MEET THE RESEARCHER

Professor Matthew Bogyo
Department of Pathology
University of Stanford
Stanford, CA
USA
Professor Matthew Bogyo received his PhD in Chemistry from Massachusetts Institute of Technology in 1997. He established his career as a Faculty Fellow at the University of California, San Francisco. Later, he directed the Chemical Proteomics Department with a focus on applying small molecule probes to the field of drug discovery. In July 2003, Professor Bogyo joined the Department of Pathology at Stanford University and in 2013 was promoted to professor. His laboratory works on the development of new chemical probe technologies that are applied to studies of proteases and their roles in complex biological pathways associated with human disease. During his career, Professor Bogyo has published over 250 primary research publications and took on the role of the President of the International Proteolysis Society. He has received numerous awards including the Searle Scholar Award and the Terman Fellowship. He is the co-founder of Akrotome Imaging, an innovative start-up company developing imaging contrast agents for the detection of surgical margins.
CONTACT
E: mbogyo@stanford.edu
W: https://profiles.stanford.edu/matthew-bogyo?tab=bio
KEY COLLABORATORS
Jonathan Sorger (Intuitive Surgical)
John Santini and Eric Bensen (Vergent Biosciences)
Go van Dam (Tracer)
FUNDING
National Institutes of Health
Intuitive Surgical
FURTHER READING
BM Babin, G Fernandez-Cuervo, J Sheng, et al., Chemiluminescent Protease Probe for Rapid, Sensitive, and Inexpensive Detection of Live Mycobacterium Tuberculosis, ACS Central Science, 2021, 7(5), 803–814.
JC Widen, M Tholen, JJ Yim, et al., AND-gate contrast agents for enhanced fluorescence-guided surgery, Nature Biomedical Engineering, 2021, 5, 264–277.
M Tholen, JJ Yim, K Groborz, et al., Design of optical imaging probes by screening of diverse substrate libraries directly in disease tissue extracts, Angewandte Chemie, 2020, 59, 1433–7851.
Yim JJ, Tholen M, Klaassen A, et al., Optimization of a Protease Activated Probe for Optical Surgical Navigation, Molecular Pharmaceutics, 2018, 15, 750–758.
LO Ofori, NP Withana, TR Prestwood, et al., Design of Protease Activated Optical Contrast Agents That Exploit a Latent Lysosomotropic Effect for Use in Fluorescence-Guided Surgery, ACS Chemical Biology, 2015, 10, 1977–1988.
G Blum, SR Mullins, K Keren, Dynamic imaging of protease activity with fluorescently quenched activity-based probes, Nature Chemical Biology, 2005, 1(4), 203–209.

REPUBLISH OUR ARTICLES
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence (CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
SUBSCRIBE NOW
Follow Us
MORE ARTICLES YOU MAY LIKE
Dr Hatim Hassan | Proteins identified in gut bacteria that reduce oxalate levels
New research has identified proteins from gut bacteria, called Sel1-like proteins, that have the potential to help the body get rid of excess oxalate, an organic substance linked to kidney stones, kidney disease, and other health problems. Sel1-like proteins help the cell in assembling large molecular complexes important for cell function. Dr Hatim Hassan from the Division of Nephrology and Hypertension, Mayo Clinic, Rochester, Minnesota, United States, is part of a team of scientists researching whether these proteins and their derived peptides could reduce blood and urinary oxalate levels to prevent and/ or treat hyperoxalemia (high blood oxalate), hyperoxaluria (high urine oxalate) and related disorders (including kidney stones).
Dr Norio Mitsuhashi | Measuring Respiratory Motion to Improve Precision in Lung Radiation Therapy
Dr Norio Mitsuhashi, former Professor of the Department of Radiation Oncology at Tokyo Women’s Medical University, leads revolutionary clinical research into optimising stereotactic body radiation therapy for lung cancer. Dr Mitsuhashi and his colleagues examine whether routinely available patient and tumour characteristics can predict respiratory tumour motion, a critical source of uncertainty in high precision radiotherapy. Their findings suggest that respiratory motion cannot be reliably inferred, and must instead be measured directly in every patient.
Professor Terry C. Hrubec | Clean is good – but is too clean better?
Quaternary ammonium compounds are a large class of compounds used as disinfectants in hospitals, restaurants, healthcare and animal care facilities, and are popular as household cleaners. With disease outbreaks increasing our fears about infections, the use of disinfectants has skyrocketed in recent years. Understandably, we all want to feel safe. However, as Professor Terry Hrubec from the Department of Biomedical Sciences of E. Via College of Osteopathic Medicine discovered, such products may be causing more harm than good.
Professor Abraham P. Lee | Delivering Cancer Immunotherapy with Acoustic-Electric Precision, AESOP’s Fact not Fable
Chimeric Antigen Receptor (CAR) T-cell therapy offers life-saving potential, particularly against blood cancers, but severe side effects such as cytokine release syndrome (CRS) limit its safety. These toxicities are linked to uncontrolled CAR expression levels on the T-cell surface. Led by Professor Abraham P. Lee, researchers at the University of California, Irvine, have developed an advanced microfluidic system, called the Acoustic-Electric Shear Orbiting Poration (AESOP) platform, to precisely control the dose of genetic material delivered into primary T cells. This innovation promises safer, more homogeneous, and highly effective cellular immunotherapies.