Rydberg Atoms: Giants of the Atomic World
Article written by Dr F. Barry Dunning and Dr Thomas C. Killian
The creation of giant atoms whose size is comparable to that of a grain of sand might sound like the stuff of science fiction, but in fact such species exist in nature and can now be created in the laboratory using advanced laser systems. Such exotic atoms, in which one electron is placed in a highly-energetic state, are termed Rydberg atoms, after the Swedish spectroscopist J. R. Rydberg who first characterised their properties. As might be expected, such extreme atoms possess very unusual physical and chemical properties. Their study has provided many new insights into the properties of Rydberg atoms themselves, their interactions with other atoms and molecules, and phenomena that arise from their collective interactions. The extreme properties of Rydberg atoms now enable emerging technological applications in sensing and quantum computation.
Energetic Electrons
Atoms comprise a compact, positively-charged nucleus, surrounded by one or more negatively-charged electrons, which are bound to the nucleus by electrostatic forces. The size of the atom is determined by the volume occupied by the electrons. Under normal conditions, the electrons are held close to the nucleus, and a billion atoms placed side-by-side would extend only a few centimetres.
If an electron in the atom is given additional energy, it can jump into one of a series of allowed ‘excited states’, whose energies and orbital motions are defined by the principal quantum number, n. As the energy transfer and n increase, the electron is able to travel farther from the nucleus – the distance scaling as n2. Indeed, if the energy transfer is sufficient, the electron can escape, causing the atom to become ionised.
The largest atoms observed to date have n values of over 1200 and diameters greater than the width of a human hair. However, since the majority of the atomic volume is only occupied by a single electron, these so-called Rydberg atoms are transparent and not visible to the naked eye.
As n increases, the extra energy required to remove the electron from the atom, termed its binding energy, decreases rapidly, scaling as 1/n2. As a result, high-n atoms are not only very large but are also very fragile. Even a perfect vacuum can be a hostile environment for such atoms, because they can be destroyed by interactions with thermal radiation emitted by the walls of their enclosure at room temperature. However, their natural lifetimes are long. Whereas the lifetimes of low-lying excited states are typically measured in nanoseconds, those of very-high-n states can be milliseconds or longer, allowing plenty of time for their study.
Growth of Rydberg atom size with
principal quantum number n
Controlling Electron Motion
In very-high-n Rydberg atoms, the excited electron is so far from the rest of the atom (the ‘core ion’), that the electrostatic attraction it experiences from the nucleus is very weak, and its motion can be strongly perturbed or even dominated by weak external electric (and magnetic) fields.
Because the orbital period of the excited electron can approach a few tens of nanoseconds, the electron motion can be controlled with remarkable precision by applying external forces, or ‘kicks’, in the form of short-duration electric field pulses. Applying a carefully-tailored series of such pulses can be used to engineer the electron orbit and create ‘designer’ atoms.
This approach has been employed, for example, to localise the electron in a near-circular orbit about the nucleus, creating an atom that mimics the original Bohr model of the hydrogen atom, which pictured an electron in a classical circular orbit about the nucleus. Whereas this model has now been supplanted by more modern quantum theories, it still provides a convenient basis for discussing many Rydberg atom properties.
Similar control techniques have been used to create a model of the solar system within an atom by placing the electron in an orbit that mirrors the behaviour of Jupiter’s Trojan asteroids. These asteroids, named after figures in the Trojan Wars, orbit the sun at a similar radius some 60 degrees ahead of, and behind, the planet. In the future it appears possible to create a ‘planetary atom’ with two highly-excited orbiting electrons.

The image on the left shows Jupiter’s Trojan asteroids in their orbit, and the image on the right shows the electron probability density
distribution in a Rydberg atom that mimics their behaviour
Sensing Applications
Their sensitivity to external electric fields makes Rydberg atoms natural electric field sensors, especially for oscillating fields extending from radio-frequencies to the far infrared. Central to such applications is an effect termed electromagnetically-induced transparency, which involves a collection of atoms through which is directed a ‘probe’ laser beam tuned to a frequency that the atoms would normally strongly absorb, and a superposed ‘control’ laser beam tuned to a Rydberg state.
Under appropriate conditions, the presence of the control beam can result in the transmission of the probe beam through the otherwise opaque medium. Given that Rydberg atoms are extremely sensitive to external electric fields, such fields can cause marked changes in the probe beam transmission.
These changes have been exploited to detect radio-frequency fields over a wide frequency range that encompasses AM, FM, bluetooth, and Wi-Fi signals. Because atomic properties are intrinsically stable and are well characterised, the measurement technique is self-calibrating, permitting accurate measurements of absolute field strengths with widespread applications in, for example, communications, radar, and healthcare.
The ability to control the propagation of light through the medium afforded by electromagnetically-induced transparency has also been exploited to slow the speed of light in the medium and create ‘slow light’ with group velocities as low as a few kilometres per second.
Collisions and Molecule Formation
Because of their large physical size, in collisions with other atoms or molecules, Rydberg atoms behave not as a single atom, but rather as a pair of independent particles, namely the orbiting electron and the core ion. This behaviour is very different from that of atoms in the ground state or low-lying excited states, and results in a broad range of possible reactions, many unique to Rydberg atoms.
In collisions that are dominated by electron-target interactions, the electron behaves like a free particle, except that its average kinetic energy is determined by n and is very low. Indeed, Rydberg atoms provide a remarkable microscale laboratory in which to study a broad range of electron collision processes at effective collision temperatures under 1 Kelvin (−272°C), well below those accessible using any alternate techniques.
One novel outcome of collisions with targets that attach low-energy electrons to form negative ions is the creation of molecules that comprise an electrostatically-bound positive-negative ion pair that orbit at large separations. These giant neutral molecules, termed ‘heavy Rydberg’ molecules, possess many of the characteristics of Rydberg atoms, except that the electron is replaced by a much heavier negative ion.
A different giant molecular species is formed in Rydberg collisions with molecules that have large permanent electric dipole moments. The Rydberg electron is captured by the target molecule, becoming weakly bound in its dipole field and creating a ‘dipole-bound’ negative ion. The electron orbits at a large radius near the positive end of the dipole and much of its behaviour again parallels that of a Rydberg electron.
The scattering of the Rydberg electron from a neighbouring ground-state atom creates a novel chemical bond and allows the creation of ultralong-range Rydberg molecules with an internuclear separation comparable to the size of the Rydberg atom itself. The bond is very weak and such molecules are not stable at room temperature. Nevertheless, with the advent of laser cooling and trapping techniques, it is possible to create atomic gases with temperatures of 1 microkelvin or less, which permits the creation and study of such molecular species in the laboratory.
Initial work produced rubidium dimer molecules consisting of one Rydberg atom and one bound ground-state atom. Several different Rydberg dimer species have now been produced and their rich vibrational structure examined. So-called trilobite Rydberg dimers have also been realised that possess giant (permanent) electric dipole moments.
Since the size of a Rydberg molecule is determined by the size of the parent Rydberg atom, their formation depends on having atom pairs with the appropriate initial separation. Measurements of dimer formation as a function of n therefore provide a valuable probe of spatial correlations in quantum gases, allowing effects such as particle bunching in a gas of bosons and antibunching in a gas of fermions due to Pauli exclusion to be seen. The formation of more complex molecules with a Rydberg atom bound to two or three ground-state atoms has also been observed.
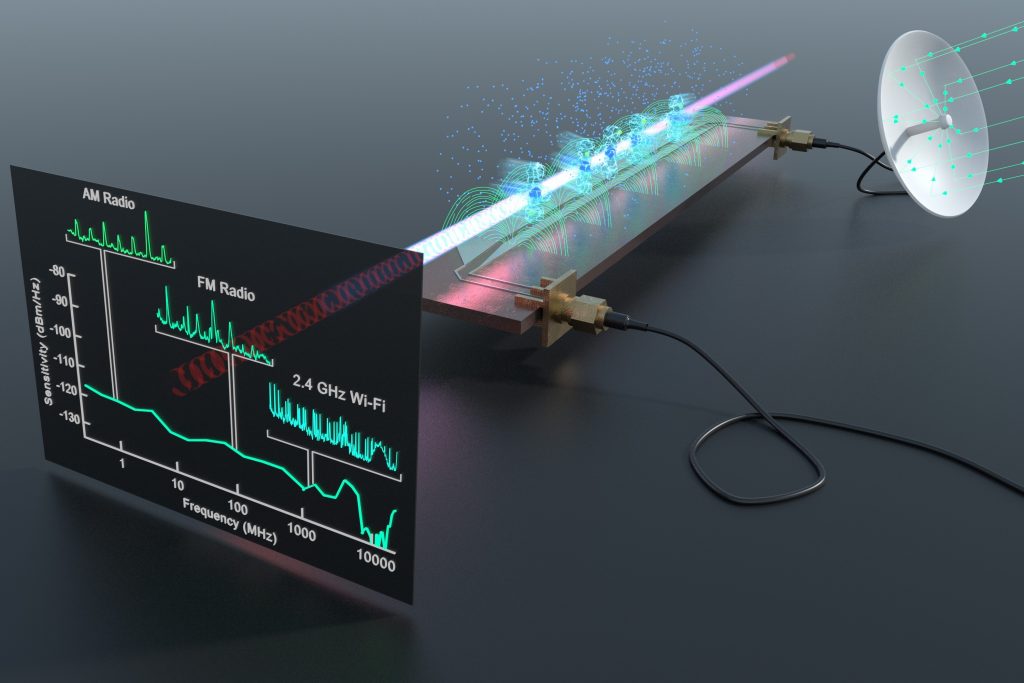
A Rydberg receiver and spectrum analyser above a microwave circuit can detect a wide range of radio signals,
including AM, FM, Wi-Fi and Bluetooth. CREDIT: US Army
Rydberg Atoms in Dense Gases
Measurements have been extended to Bose-Einstein condensates, in which the atom densities are so high that a Rydberg electron orbit can enclose tens to even hundreds of ground-state atoms. Those atoms within the Rydberg orbit form weak bonds and create a molecule composed of many ‘atoms within an atom’, the atoms being corralled by the orbiting electron which serves the role of a shepherd.
The many atoms contained within the molecule constitute a novel form of ‘quantum matter’ and their interactions can lead to novel collective responses including the formation of ‘quasi-particles’ similar to those that play important roles in determining the properties of many materials. Indeed, Rydberg molecules provide a valuable window into many of the important phenomena that emerge from many-body interactions in condensed-matter settings.
Quantum Research
Rydberg atoms interact strongly with each other even at large separations. The strength of their interactions is millions of times larger than those between ground-state atoms and is tuneable through the choice of the Rydberg states excited and their physical separations.
Rydberg-Rydberg interactions form the basis of many ongoing studies of quantum simulation and of quantum logic devices. Central to this work is the phenomenon of ‘dipole-blockade’, in which resonant excitation of one atom to a Rydberg state can shift the energy of neighbouring atoms within some critical ‘blockade’ radius preventing their subsequent excitation.
The use of focused laser beams, termed ‘optical tweezers’, to trap and hold atoms now permits atoms to be positioned with remarkable precision. This has enabled the realisation of fast and robust quantum gates using pairs of atoms positioned within the blockade radius. Such gates utilise conditional logic where the excitation of the second atom is governed by excitation of the first one. Quantum gates, or ‘qbits’, are critical to the operation of a quantum computer.
Tweezer arrays, as well as ‘optical lattices’ formed by intersecting laser beams, provide the opportunity to assemble one-, two-, and even three-dimensional ordered arrays of Rydberg atoms with precise control of their separations and geometry. Such arrays can be engineered to mimic the arrangement of atoms within a solid. They therefore provide a powerful quantum simulator with which to probe the physical processes that underpin quantum effects that give rise to high temperature superconductivity and magnetism.
Furthermore, by varying the atomic arrangements and interactions, a wide variety of ‘artificial’ structures not normally found in nature can be simulated and their properties explored. The effects of defects introduced by removing one, or more, atoms from the array at selected points can also be examined. Such studies promise an improved understanding of the behaviour of many condensed matter systems and will enable the design of new materials with enhanced electronic and magnetic properties.

Theoretical electron density pattern of a weakly bound Rydberg molecule, resembling a
trilobite. CREDIT: Chris Greene, Purdue University
A Future of Discovery
As is evident from the above examples, to study Rydberg atoms is to wander in a land of giants with exciting research opportunities appearing around every corner. They form a valuable bridge between the microscopic world described by quantum mechanics and the macroscopic world governed by Newton’s laws, and illuminate the transition from one realm to the other as the length scale increases.
New experimental capabilities continue to drive new avenues of research that exploit their novel characteristics, and going forward it is certain that they will remain the focus of much activity and provide many exciting discoveries.
Reference
https://doi.org/10.33548/SCIENTIA679
Meet the researchers

Dr F. Barry Dunning
Department of Physics and Astronomy
Rice University
Houston, TX
USA
Dr F. Barry Dunning is the Sam and Helen Worden Professor of Physics at Rice University in Houston, Texas. He received his BSc and PhD degrees from University College, London and moved to Rice University immediately on completion of an ICI Postdoctoral Fellowship. His research interests lie in experimental atomic, molecular, and optical physics. Dr Dunning was an early pioneer in the study of Rydberg atoms and his interest in such atoms and their applications continues to this day.
CONTACT
E: fbd@rice.edu
W: https://profiles.rice.edu/faculty/f-barry-dunning

Dr Thomas C. Killian
Department of Physics and Astronomy
Rice University
Houston, TX
USA
Dr Thomas C. Killian is the E. Dell Butcher Chair and Professor of Physics and Astronomy and the Dean of the Wiess School of Natural Sciences at Rice University in Houston, Texas. He earned an AB in Physics from Harvard University, attended Cambridge University on a Marshall Scholarship, and received his PhD at the Massachusetts Institute of Technology. Before appointment at Rice, he was a National Research Council Postdoctoral Fellow at the National Institute of Standards and Technology. His research interests include ultracold neutral plasmas, Rydberg atoms, and quantum degenerate atomic gases.
CONTACT
E: killian@rice.edu
W: https://profiles.rice.edu/faculty/thomas-c-killian
FUNDING
The research of the authors is funded by the National Science Foundation and the Air Force Office of Scientific Research.
FURTHER READING
JD Whalen, SK Kanungo, Y Lu, S Yoshida, J Burgdörfer, FB Dunning, TC Killian, Heteronuclear Rydberg molecules, Physical Review A, 2020, 101, 060701(R).
A Browaeys, T Lahaye, Many-body physics with individually-controlled Rydberg atoms, Nature Physics, 2020, 16, 132.
FB Dunning, S Buathong, Collisions of Rydberg atoms with neutral targets, International Reviews in Physical Chemistry, 2018, 37, 287.
M Saffman, Quantum computing with atomic qubits and Rydberg interactions: progress and challenges, Journal of Physics B: Atomic, Molecular and Optical Physics, 2016, 49 202001.
H Fan, S Kumar, J Sedlacek, H Kübler, S Karimkashi, and JP Shaffer, Atom based RF electric field sensing, Journal of Physics B: Atomic, Molecular and Optical Physics, 2015, 48, 202001.
Rydberg Physics, N Šibalić, CS Adams, IOP Publishing, 2018, DOI: 10.1088/978-0-7503-1635-4.
Rydberg Atoms, TF Gallagher, Cambridge University Press, Cambridge, 1994.
Rydberg States of Atoms and Molecules, edited by RF Stebbings and FB Dunning, Cambridge University Press, Cambridge, 1983.

Want to republish our articles?
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence
(CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
More articles you may like
Jella van de Laak | Could Tumour- Targeting Bacteria Enhance Cancer Immunotherapy?
Cancer immunotherapy has transformed treatment outcomes, yet many patients still fail to respond. At Maastricht University, Jella van de Laak, working with Professor Ludwig Dubois and Professor Jan Theys, is exploring how tumour-targeting bacteria could improve immune responses and expand treatment success.
Distinguished Professor Michael Zhdanov | Mapping Magma and Drilling for Oil: New Methods for Geophysical Modelling
Geophysicists use a variety of different methods to peer beneath the Earth’s surface. Seismic activity, gravitational fields , and magnetic fields each offer their own windows into the world underground, but, in isolation, are incapable of giving us the full picture. Combining data from distinct geophysical surveys, however, is its own challenge. For a number of years, a team of researchers led by Professor Michael Zhdanov has worked to develop a mathematical framework capable of generating detailed geophysical models from multiphysics data.
Through application to modelling magma chambers underneath Yellowstone and searching for oil deposits in the Barents Sea, they demonstrate that their approach can produce robust and accurate predictions
Professor Deanna Sellnow | How Strategic Communication Can Save Lives When Crisis Hits
Professor Deanna Sellnow from Clemson University argues that crisis and risk communication research must evolve beyond simply describing and deconstructing problems to actively empowering people protect themselves and their communities. In an era of increasing globalisation, she proposes that researchers adopt learner- and learning-centered approaches to ensure communication does not simply inform people about dangers, but also teaches them how to respond effectively (e.g., to reduce risks, mitigate harms, improve safety, save lives) and why.
Dr David Berry | Bridging the gap between Agriculture and Automation
As automation and controlled environments reshape modern farming, the need for technicians who understand both plant science and engineering is rapidly increasing. At Virginia Western Community College (VWCC) located in Roanoke, VA, Dr David Berry is leading a programme supported by the National Science Foundation (NSF) with the aim to merge agriculture with mechatronics, preparing students for the realities of emerging industries. A distinctive example of this approach is an automated hydroponics system designed by two industry-sponsored students, demonstrating how practical experience, interdisciplinary training and strong employer partnerships can create the skilled workforce required for the future of sustainable food production.




