The Thinnest of Materials May Hold a Cure for Osteoarthritis
Since the turn of the century, a myriad of exciting applications for graphene have emerged. Amongst the most exciting might be its use as a scaffold for promoting tissue growth in the treatment of various medical conditions, including osteoarthritis. Researchers at Boise State University in the USA and Carl von Ossietzky University of Oldenburg in Germany are gaining a greater understanding of the interactions between graphene and cells, towards the development of implantable graphene-based devices that can rebuild damaged tissue.
Crippling Osteoarthritis
Osteoarthritis is a debilitating musculoskeletal disorder of the bones, muscles and connective tissues that results from the breakdown of cartilage tissue in joints. The resulting pain that accompanies osteoarthritis can be crippling – reducing the quality of life for those afflicted with the disease.
Affecting almost 250 million people worldwide, osteoarthritis is the most common form of arthritis, and can result in significant disability. In the US, the condition affects 50% of the population over the age of 65. Given that between 2015 and 2050, the proportion of the world’s population over 65 years of age will almost double, the World Health Organization has categorised osteoarthritis as a priority disease in urgent need of new therapies. Currently, there is no cure for osteoarthritis; instead, most therapies simply alleviate symptoms.
Tissue Engineering
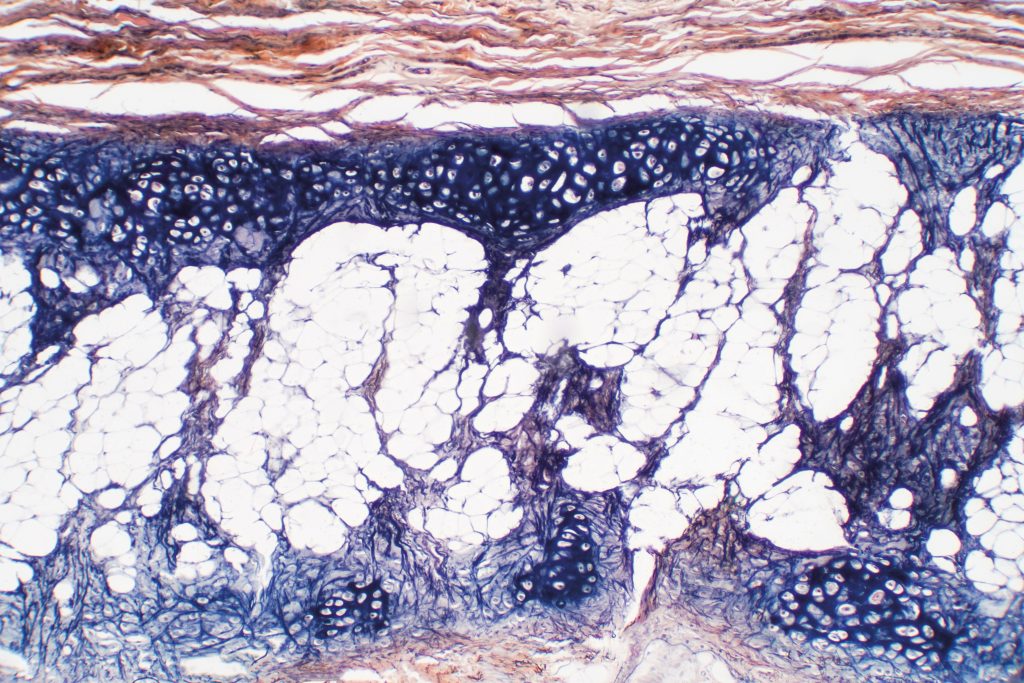
Applying principles from engineering and life sciences, tissue engineering involves the development of biological substitutes that can repair damaged tissue. This exciting field often involves the use of materials called ‘bioscaffolds’, which can be seeded with cells and implanted into the body to regenerate an area of damaged or lost tissue. Stem cells are special cells that have the ability to develop into many different types of tissue, such as cartilage. Combining stem cells and bioscaffolds represents a promising method to replace the cartilage lost due to osteoarthritis.
A well designed bioscaffold can encourage stem cells to develop, or ‘differentiate’, into the type of tissue required for the desired application. In the case of osteoarthritis treatment, a bioscaffold should provide a conducive environment to promote the development of cartilage-producing cells, and ultimately, functional musculoskeletal tissue.
There are various different environmental factors that determine the type of tissue that stem cells will develop into. Therefore, scientists are trying to understand the effects of these chemical, physical and mechanical factors on cells, so that they can be tuned to create an ideal environment that encourages the formation of a particular tissue type.
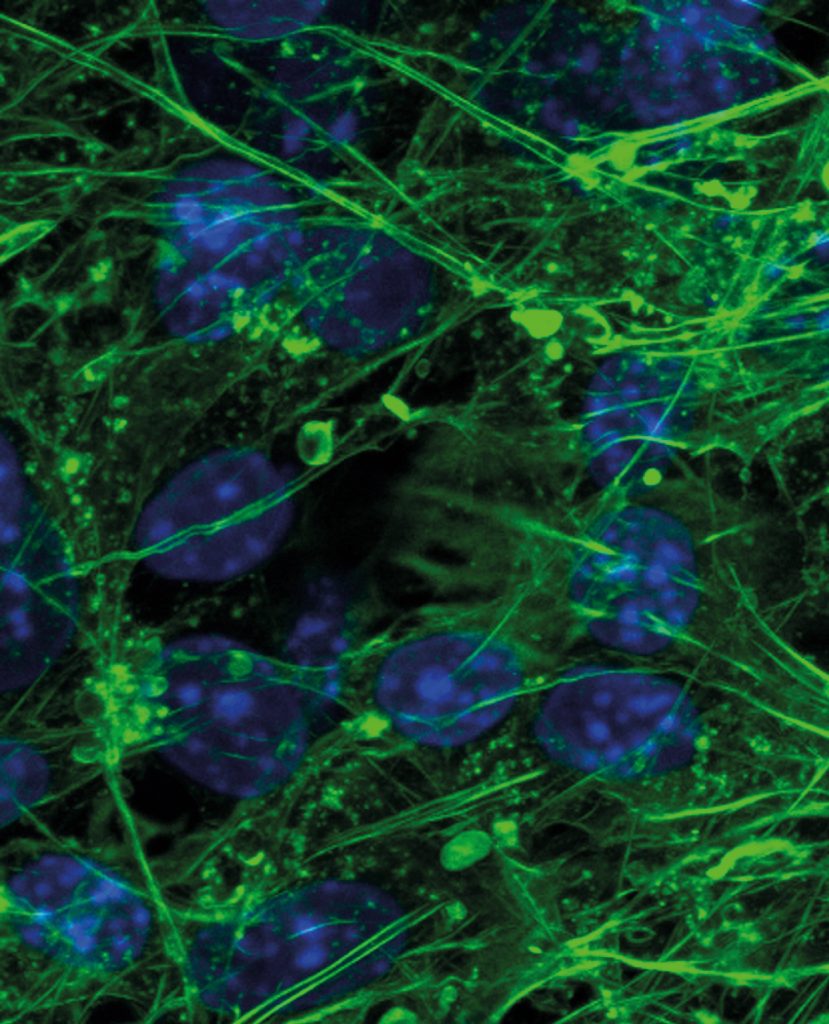
The Potential of Graphene
Graphene is a single layer of carbon atoms arranged in a two-dimensional hexagonal crystal structure, which has exceptional strength and conductive properties. These characteristics have led to its use in many different fields including electronics and energy, but its application in tissue engineering has yet to be fully exploited.
With the potential to communicate electrically and mechanically with adhered stem cells, graphene represents a unique bioscaffold material. Under the right conditions, graphene has been shown to encourage stem cells to differentiate into cartilage tissue. Therefore, it could play a vital role in regenerating damaged or lost cartilage tissue in osteoarthritis patients.
Dr David Estrada and Dr Julia Oxford at Boise State University, USA, and Dr Solov’yov at Carl von Ossietzky University in Oldenburg, Germany are studying the influence of this material on stem cells under a broad range of conditions, towards the development of optimised bioscaffolds for regenerating damaged tissue, including cartilage.
Uncovering the fundamental interactions between graphene and stem cell biology requires interdisciplinary knowledge and expertise, which is why these three researchers have combined forces. With Dr Solov’yov’s interest in biological processes, Dr Estrada’s extensive experience in graphene research and Dr Oxford’s track record in advancing stem cell biology, the team is perfectly equipped to make great advances in this area.
Graphene Foam
Recently, the researchers have become particularly interested in a porous, three-dimensional form of graphene known as graphene foam. Its flexibility and conductive properties make it suitable for helping nerve cells transmit information, while its three-dimensional porous nature means that many cells can be seeded throughout the material’s structure.
The team has been investigating whether it is possible to adhere a patient’s own stem cells to a graphene foam scaffold, and then insert this material into the body to repair and regenerate cartilage tissue. Graphene foam’s exceptional flexibility and strength make it ideal for this application, because cartilage in joints must regularly dissipate high-impact forces.
Before the research team’s investigations, little had been reported about the mechanical properties of graphene foam with adhered cells under dynamic compressive forces. Likewise, the use of graphene foam as a three-dimensional scaffold needed examination, so the researchers began by assessing stem cell attachment, and subsequent cell differentiation under different conditions. After first demonstrating that graphene foam could be used to grow muscle tissue, they built bioscaffolds by mixing graphene foam with animal cells and tested their mechanical properties.
After measuring the elastic and viscous characteristics of these bioscaffolds under deformation, the researchers were able to determine the cellular contribution to the mechanical strength and elastic properties of the composites, as well as concluding that cartilage-producing cells were viable on graphene foam.
Combining the Micro with the Nano
Another aspect of three-dimensional graphene foam scaffolds is their potential to combine micro-scale material and nano-scale molecular properties. In particular, the team evaluated the impact of these combinations on cell attachment and cell morphology.
The researchers achieved this by coating graphene foam with a protein called fibronectin before growing stem cells within the scaffold. They found that the fibronectin coating increased the elastic strength of the scaffold, predominantly through interactions between the graphene and the protein, which also alters the cellular response to graphene foam. The team found that their modified scaffold provided an environment conducive to creating early-stage cartilage formation.
Selecting appropriate molecular modifications for scaffold materials can be key for ultimately growing a particular tissue type, such as cartilage, as this is regulated by a combination of the molecular and material properties of the underlying scaffold. In other words, the nature of the proteins used to modify scaffolds must be considered carefully.
Improving Fundamental Understanding
The research to date has been encouraging, but a more fundamental understanding of the chemical and physical interactions between graphene and stem cells that promote tissue development is still needed, in order to optimise a potential graphene foam scaffold for osteoarthritis patients.
Towards this aim, Dr Oxford, Dr Estrada and Solov’yov are embarking on a major project funded by the US National Science Foundation, entitled ‘Graphene as a Bioscaffold for Musculoskeletal Tissue Engineering’. Through this project, the team aims to modulate the electrical, thermal and mechanical properties of graphene foam in just the right way to guide and differentiate stem cells into musculoskeletal tissue that will rebuild cartilage.
First of all, the researchers plan to explore the effects of graphene’s electrical voltage on the growth of stem cells, and their differentiation into musculoskeletal tissue. By heating graphene to different temperatures, they also plan to study how temperature influences stem cell behaviour. The third aspect of graphene that they plan to investigate is its mechanical stiffness, which can be modulated during the manufacturing process. Once these studies have been conducted, the team hopes to achieve a complete dataset that will reveal the effects of graphene’s physical properties on the behaviour and differentiation of stem cells.
The researchers are also planning to investigate different methods of fabricating graphene, to obtain variations in the material’s structure. Preliminary data have shown that structural changes significantly impact graphene’s performance as a bioscaffold in terms of cell growth and differentiation. The team plans to utilise various fabrication methods, including inkjet printing, chemical vapor deposition, and sublimation of silicon from silicon carbide wafers, to create a range of different graphene films with varying structures and properties. They aim to pay particular attention to tuning the surface roughness of the material, as this property is known to affect cell behaviour.
As the abovementioned preliminary experiments will utilise two-dimensional graphene, the next part of the team’s project will extend their results to three-dimensional graphene foam, where softness, porosity and pore shape are related factors that are known to play key roles in helping cells to adhere to bioscaffolds. Like their work with two-dimensional graphene, they aim to modulate the various characteristics of graphene foam to identify an optimum set of properties that will encourage the growth of stem cells and their differentiation into cartilage tissue.
Disruptive Devices
Until now, symptomatic relief – or in extreme cases, complete joint replacement – has been the only viable therapeutic option for osteoarthritis sufferers. Repairing damaged cartilage has always been challenging in regenerative medicine, due in part, to the limited ability for cartilage to heal. Implantable graphene-based biomedical devices may hold the key to revolutionising tissue engineering as a viable therapy for those suffering from osteoarthritis. There can be little doubt that Ds Estrada, Dr Oxford and Dr Solov’yov sit at the vanguard of this future breakthrough.
Reference
https://doi.org/10.33548/SCIENTIA586
Meet the researchers

Professor David Estrada
Micron School of Materials Science and Engineering
Boise State University
Boise, ID
USA
Having gained a PhD in 2013 from University of Illinois at Urbana-Champaign, Professor David Estrada subsequently went on to hold several Associate Professorships within Boise State University, including at the Micron School of Materials Science and Engineering and Center for Advanced Energy Studies. Currently, he is also Advanced Manufacturing Deputy Director of Academic Research at Idaho National Laboratory. Professor Estrada’s research output has realised the filing of more than 6 patent applications on topics ranging from nanoparticle inks to printed graphene-based biosensors.
CONTACT
E: daveestrada@boisestate.edu
W: https://www.boisestate.edu/coen-materials/faculty-and-staff/david-estrada-ph-d/

Professor Julia Thom Oxford
Biological Sciences and Biomolecular Research Center
Boise State University
Boise, ID
USA
Professor Julia Thom Oxford gained her PhD in Biochemistry and Biophysics at Washington State University in 1986. She is currently Distinguished Professor in the Department of Biological Sciences of Boise State University, where she holds the Stueckle Endowed Chair, as well as being Director of the Center of Biomedical Research Excellence in Matrix Biology and Director of the Biomolecular Research Center. Over the course of her successful research career, Professor Oxford has been awarded in excess of $40 million in grant funding. In addition to her tissue engineering research, she is also passionate about cultivating the next generation of STEM professionals, supervising numerous student research projects and teaching an array of different biology and chemistry courses.
CONTACT
E: joxford@boisestate.edu
W: https://www.boisestate.edu/biology/faculty-and-staff/faculty/julia-oxford/
W: https://www.boisestate.edu/brc

Professor Ilia Solov’yov
Department of Physics
Carl von Ossietzky University
Oldenburg
Germany
Professor Solov’yov graduated with a PhD in Physics from Johann Wolfgang Goethe University, Germany, in 2008. After conducting postdoctoral research at University of Illinois at Urbana-Champaign in the US, he worked as an Associate Professor at the University of Southern Denmark between 2014 and 2019. He currently works as Professor of Physics at Carl von Ossietzky University in Germany, where his research interests cover a broad range of areas relating to biomolecules and smart inorganic materials. He has published over 120 research articles, and is currently an Academic Editor for the journal Scientific Reports, which is published by the Nature Publishing Group.
CONTACT
E: ilia.solovyov@uni-oldenburg.de
W: https://www.quantbiolab.com
KEY COLLABORATORS
Camilla Coletti of the Italian Institute of Technology conducted work on epitaxial graphene
FUNDING
National Institutes of Health Center of Biomedical Research Excellence in Matrix Biology P20GM109095
Idaho IDeA Network for Biomedical Research Excellence P20GM103408
National Science Foundation CAREER award #1848516, Graphene as a Bioscaffold for Musculoskeletal Tissue Engineering
National Science Foundation #0619793 Confocal Microscopy
National Science Foundation #0923535 Mass Spectrometry
Lori and Duane Stueckle Endowed Chair in Biology (
The MJ Murdock Charitable Trust


Want to republish our articles?
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence
(CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
More articles you may like
Prof Candis M. Morello – Prof Jan D. Hirsch | Recent innovations in pharmacy education
A pioneering research team from the Skaggs School of Pharmacy and Pharmaceutical Sciences, University of California, San Diego, United States, has been instrumental in developing innovative techniques for teaching pharmacy students. The Next Generation of Pharmacist Educators (NextGen-RxEd) programme is a new method of training the next generation of pharmacist educators and academics. To help pharmacists and pharmacy students visualise the complex issues experienced by their patients, the team led by Professors Candis Morello and Jan Hirsch developed an innovative educational tool, called the Medication Therapy Management (MTM) Spider Web.
Professor Michael Yarus | How RNA Started the Conversation That Built Life
The genetic code stores the instructions for building proteins, yet how it first arose remains unclear. It likely did not appear fully formed, but instead emerged step by step from simple chemical interactions. In this study, a team led by Professor Michael Yarus at the University of Colorado Boulder shows how early ribonucleic acid (RNA) molecules could bind specific amino acids, creating the first coding relationships. These early interactions are then refined through evolutionary processes such as duplication and merging of partial codes. By combining the experimental data with computer simulations, the work provides a testable pathway from prebiotic chemistry to the modern genetic code.
Identifying Nutritional Risk in Early Childhood: Insights from NutriSTEP®
Early childhood is a critical period for growth and development, yet many young children face nutritionrelated risks that can go unnoticed. Professor Janis Randall Simpson and colleagues have developed NutriSTEP®, validated and reliable screening tools that help identify potential nutritional concerns in toddlers and preschoolers. Their large-scale analysis of Canadian data reveals patterns in diet, behaviour, and food access that could help guide early interventions and support healthy development.
Professor Tian Yu Cao | Twistor Theory: A New Framework for Quantum Gravity
At Boston University, Professor Tian Yu Cao is rethinking the foundations of modern physics. His work builds on twistor theory which demonstrates that spacetime is secondarily derived from twistor constructions, but goes further to highlight the most important implication of the Penrose transform in that the primary physical agents can only be mathematically described by elements of cohomology with the defining feature having roots in spin. This view of primary agents combines with Cao’s other major claim that quantum behaviour itself may arise from the physical property of spin leads to a new consistent framework of quantum gravity in which long-standing puzzles in black holes (evaporations) and cosmology (transitions between cycles of cosmos) can be adequately addressed, with the crucial help from the on-going development of operator product expansion formular defined on twistor space.




