Dr Daisuke Minakata – Sunshine and Organic Molecules in Water
Organic molecules dissolved in rivers, lakes, seas and oceans are essential to plant and animal life. Some of these molecules are also degraded and enter a complex cycle of carbon, nitrogen and sulphur containing compounds. Surprisingly, scientists currently have a limited understanding of the fate of these molecules. Dr Daisuke Minakata and his colleagues from Michigan Technological University are involved in an ambitious programme to overcome this critical knowledge gap.
Organic Molecules in Water
Organic molecules are complex carbon containing structures that are indispensable for the existence of life. ‘Organic material’ is an umbrella term that includes a very wide range of compounds and amino acids – the building blocks of proteins – represent some of the most precious compounds necessary for living organisms. Given the importance of these molecules, it is hardly surprising that NASA is looking for amino acids in the search for evidence of life elsewhere in the universe.
Amino acids and other organic compounds enter natural bodies of water from multiple sources. Bacteria, algae, plants and animals living in the water itself release significant amounts of organic molecules. In addition, agricultural, human and animal waste products also enter waterways in large quantities, contributing to their amino acid and other organic material content.
A Poorly Understood Cycle
The fate of these compounds present in fresh and saltwater environments is poorly understood. Some organic molecules, including amino acids, are taken up and reused by organisms living in the water, but a significant proportion is broken down through chemical and physical processes and these smaller degradation products enter a complex cycle of nitrogen and sulphur containing molecules. The photochemical process is one important degradation pathway … and this is where things get complicated.
Theoretically, the energy from sunshine could degrade certain amino acids, making them unavailable for living creatures. However, there is evidence that dissolved organic molecules generate very short-lived reactive intermediates under sunlight irradiation and these reactive intermediates degrade the amino acids – the photosensitising effect of dissolved organic molecules.
Dr Daisuke Minakata from Michigan Technological University was prompted to develop a theory to explain the loss of critical amino acids from water. He assumed that other organic molecules absorb the energy carried by the rays of sunshine and those reactive intermediates transform amino acids into small pieces. According to his theory, such light-activated compounds could act as catalysts, inducing the indirect breakdown of critical amino acids.
A Need to Simplify Things
As Dr Minakata explains, testing this theory was difficult for two reasons. Firstly, dissolved organic molecules form very poorly defined, large complexes that show considerable variability at different locations. Using such complex mixtures in experiments would make the analysis of the results extremely difficult. The other issue was that are more than 20 different amino acids in nature, and these are very different chemically. Finding the degradation products of all of these would be a Herculean task. Thus, to conduct the experiments, the research team needed to simplify things.
Despite the complexity of the dissolved organic material, scientists had identified the three main mechanisms explaining its catalytic behaviour. Furthermore, they were able to identify smaller single compounds that could mimic this action. Using these mimics in the experiments promised to simplify the analysis of the results.
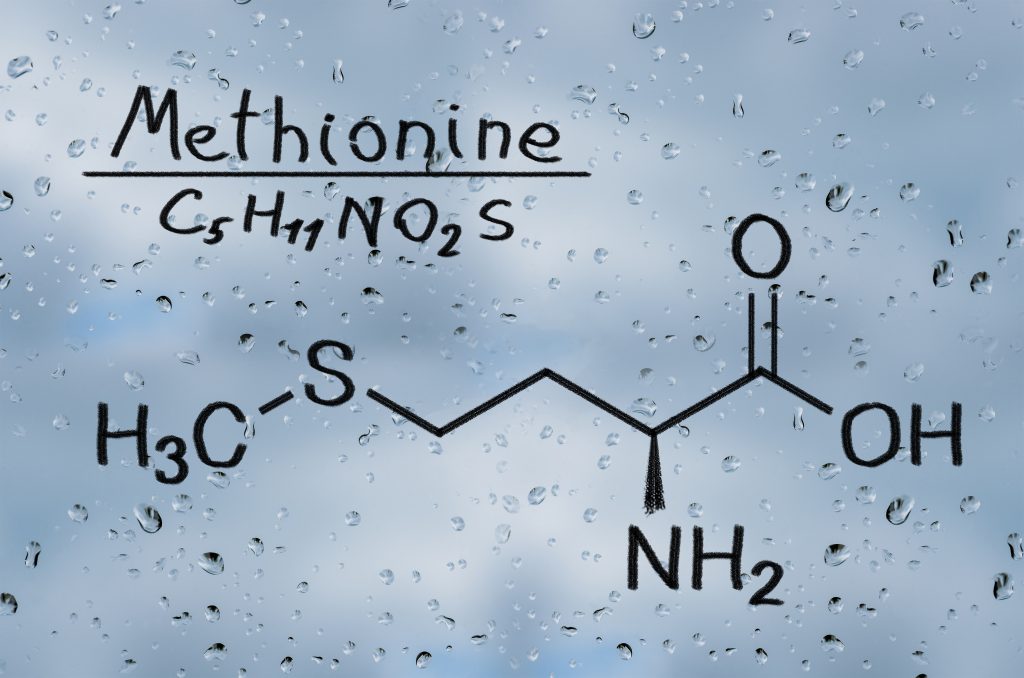
The next decision Dr Minakata and his colleagues made was that they would initially focus their attention on the study of a single amino acid, methionine, that is typically depleted in water under natural conditions. Methionine is one of only five photochemically degradable amino acids and it contains sulfur-atom, a unique element that contributes to the global cycling of sulfur. Sulfur cycling is not as well understood as carbon and nitrogen cycling.
An Elaborate Experimental Setup
Next, the scientists needed to build the appropriate experimental setup. Firstly, the effects of sunlight were replicated using a high-energy xenon arc light source. The water, containing the mixture of methionine and the tested catalytic mimics, needed to be placed under this light in a container that allowed all the wavelength through. However, illuminating a relatively small volume of solution in the specialised quartz containers resulted in an additional problem: the water became very hot. Thus, scientists had no way of establishing which degradation products were produced by light exposure, and which ones resulted from the heat. Understanding the basic science needed a controlled environment in the laboratory as the first step. This meant they needed to build a cooling system that allowed the team to keep the solution at the desired temperature.
A technique called mass spectrometry was used to detect individual degradation products. After irradiating the samples with the artificial sun, the molecules were exposed to a strong electric field that made them fly along a vacuum tube. Since under these conditions smaller molecules accelerate faster while larger ones move more slowly, measuring the time it takes for a molecule to reach the detector allows its mass to be determined. This allowed the scientists to establish what its chemical structure was likely to be. This may sound complicated – and it certainly was. The group had to resort to the use of some of the most cutting-edge equipment to make these measurements.

The Results Start Coming In
The initial screening experiments identified common methionine degradation products, and a range of compounds that were produced in smaller quantities during light exposure in the presence of mimics acting as catalysts. Knowing the composition of these degradation products allowed the scientists to work out the possible chemical reactions resulting in their emergence. Once the reaction pathways were identified in this relatively simple experimental system, the scientists started to aim higher. Instead of using chemical mimics acting as catalysts, they started to use a more complex naturally occurring dissolved organic molecule mixture, derived from either the Suwannee River or an Elliot Soil sample, typically used as standard organic molecules.
While these mixtures have the same technical name, humic acid, this is a misleading scientific term, as humic acid represents a very complex mixture of decomposed biological material, that is different at the two sites. Not unexpectedly, the degradation of methionine produced in the presence of the two distinct humic acid samples showed marked differences. Firstly, the light-dependent degradation of methionine was faster, particularly in the presence of the Elliot Soil humic acid. The proportion of produced degradation products also showed considerable differences, both between the two humic acid samples, and compared to the results obtained using simple chemical catalytic mimics. However, the results from simple mimics advance scientific understanding of the roles of different sites in complex mixtures of dissolved organic compounds.
Computational Chemistry to the Rescue
As Dr Minakata tells us, testing the mechanisms causing the degradation of the amino acid caused by such complex chemical mixtures is impossible using conventional experimental approaches. In the analysis of such complex reactions, scientists are increasingly relying on the use of computational chemistry.
By the start of the 21st century, scientists had accumulated enough experimental information to enable them to model chemical reactions using computers. By modelling details such as the shape of molecules, their known interactions, the energy needed to introduce changes, or the amount of energy being released during chemical reactions, scientists can predict chemical interactions, including those that have not been studied under experimental conditions.
After entering their experimental data into computer models, Dr Minikata and his colleagues were able to take advantage of this approach to fully understand the fate of methionine in natural water. To a superficial observer, this may sound like a lot of effort to study the fate of a single amino acid. However, deciphering what happens to this vital building block provides is the first step in understanding chemical processes in nature that allow life to flourish in natural bodies of water. Unquestionably, such an understanding is fundamentally important to humanity.
SHARE
DOWNLOAD E-BOOK
LISTEN TO THE AUDIO
REFERENCE
https://doi.org/10.33548/SCIENTIA882
MEET THE RESEARCHER

Dr Daisuke Minakata
Department of Civil, Environmental and Geospatial Engineering
Michigan Technological University
Houghton, MI
USA
Dr Daisuke Minakata obtained his BSc degree at the Environmental Engineering Department of Kyoto University in 2002. In 2005, he completed his MSc degree at the same institution. After moving to the USA, Dr Minakata studied at the Department of Environmental Engineering of the Georgia Institute of Technology, gaining his PhD in 2010. After graduation, he started working as a Research Engineer at the Brook Byers Institute for Sustainable Systems at the Georgia Institute of Technology. In 2013, he was appointed as an Assistant Professor in the Department of Civil and Environmental Engineering at the Michigan Technological University, where he became an Associate Professor in 2019. He also spent a year as a visiting scholar in Europe, at the Swiss Federal Institute of Aquatic Science and Technology in Dübendorf. Over his career to date, Dr Minakata has mentored over 30 graduate students, published more than 50 peer-reviewed papers, and given more than 25 invited and keynote talks and 70 conference presentations. Dr Minakata has been involved in projects exceeding $7M of which his own fraction is greater than $1.3M. His contributions to environmental science have been recognised with several prestigious awards.
CONTACT
FUNDING
National Science Foundation
Michigan Space Grant Consortium
FURTHER READING
B Barrios, D Minakata, Aqueous-phase single-electron transfer calculations for carbonate radicals using the validated Marcus theory, Environmental Science & Technology Letters, 2023, 10, 204–209. DOI: https://doi.org/10.1021/acs.estlett.2c00913
B Barrios, B Mohrhardt, P Doskey, D Minakata, Mechanistic insight into the reactivities of aqueous-phase singlet oxygen with organic compounds, Environmental Science & Technology, 2021, 55(12), 8054–8067. DOI: https://doi.org/10.1021/acs.est.1c01712
D Kamath, S Mezyk, D Minakata, Elucidating the elementary reaction pathways and kinetics of hydroxyl radical-induced acetone degradation in aqueous phase advanced oxidation processes, Environmental Science & Technology, 2018, 52(14), 7763–7774. DOI: https://doi.org/10.1021/acs.est.8b00582
D Minakata, D Kamath, S Maetzold, Mechanistic insight into the reactivity of chlorine-derived radicals in the aqueous-phase UV/chlorine advanced oxidation process: Quantum mechanical calculations, Environmental Science & Technology, 2017, 51(12), 6918–6926. DOI: https://doi.org/10.1021/acs.est.7b00507
D Minakata, SP Mezyk, JW Jones, et al., Development of linear free energy relationships for aqueous phase radical-involved chemical reactions, Environmental Science & Technology, 2014, 48, 13925–13932. DOI: https://doi.org/10.1021/es504491z
D Minakata, W Song, J Crittenden, Reactivity of aqueous phase hydroxyl radical with halogenated carboxylate anions: Experimental and theoretical studies, Environmental Science & Technology, 2011, 45, 6057–6065. DOI: https://pubs.acs.org/doi/10.1021/es200978f
D Minakata, J Crittenden, Linear Free Energy Relationships between the Aqueous Phase Hydroxyl Radical (HO•) Reaction Rate Constants and the Free Energy of Activation, Environmental Science & Technology, 2011, 45, 3479–3486. DOI: https://doi.org/10.1021/es1020313
D Minakata, K Li, P Westerhoff, Crittenden, Development of a group contribution method to predict aqueous phase hydroxyl radical (HO•) reaction rate constants, Environmental Science & Technology, 2009, 43, 6220–6227. DOI: https://doi.org/10.1021/es900956c

REPUBLISH OUR ARTICLES
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence (CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
SUBSCRIBE NOW
Follow Us
MORE ARTICLES YOU MAY LIKE
Alex Spezowka | Responsible Research Writing in the Age of AI: From Detection to Transparency
Artificial intelligence is often discussed as a future challenge for research, yet it is already shaping how many papers are written. Drawing on emerging evidence and recent analysis, work led by Alex Spezowka highlights a key shift in thinking: rather than trying to detect AI use, the focus is moving towards how it can be used responsibly. This has important implications for how research is produced, evaluated, and trusted.
Distinguished Professor Michael Zhdanov | Mapping Magma and Drilling for Oil: New Methods for Geophysical Modelling
Geophysicists use a variety of different methods to peer beneath the Earth’s surface. Seismic activity, gravitational fields , and magnetic fields each offer their own windows into the world underground, but, in isolation, are incapable of giving us the full picture. Combining data from distinct geophysical surveys, however, is its own challenge. For a number of years, a team of researchers led by Professor Michael Zhdanov has worked to develop a mathematical framework capable of generating detailed geophysical models from multiphysics data.
Through application to modelling magma chambers underneath Yellowstone and searching for oil deposits in the Barents Sea, they demonstrate that their approach can produce robust and accurate predictions
Dr David Berry | Bridging the gap between Agriculture and Automation
As automation and controlled environments reshape modern farming, the need for technicians who understand both plant science and engineering is rapidly increasing. At Virginia Western Community College (VWCC) located in Roanoke, VA, Dr David Berry is leading a programme supported by the National Science Foundation (NSF) with the aim to merge agriculture with mechatronics, preparing students for the realities of emerging industries. A distinctive example of this approach is an automated hydroponics system designed by two industry-sponsored students, demonstrating how practical experience, interdisciplinary training and strong employer partnerships can create the skilled workforce required for the future of sustainable food production.
Nick Martin | Data Assimilation: Overcoming AI’s Data Uncertainty Limitations for Water Resources
Water resources are essential for human life. Knowing how to manage water, both now and in the future, is necessary to continue using it as well as possible. Nick Martin and Jeremy White are examining limitations to artificial intelligence applications in water resources generated from noisy and estimated data sets. For poor quality data sets, they found that machine learning models will perform poorly relative to tools that explicitly include physics-based descriptions of physical processes; this is because physics-based calculations can use both data and physics knowledge through data assimilation techniques.





