Dr Sudhir Jain | Understanding How Receptors and Genetic Mutations Promote Hypertension
Hypertension (high blood pressure) is a common health issue in adults that can lead to numerous co-morbidities. For hypertension to develop, a pathway called the renin-angiotensin-aldosterone system needs to be activated, and a vital part of this system involves the angiotensin 1 receptor. Dr Sudhir Jain from New York Medical College studies this receptor and how specific groups of genetic mutations, known as haplotypes, promote hypertension under different conditions.
Lifestyle Factors and Biological Mechanisms
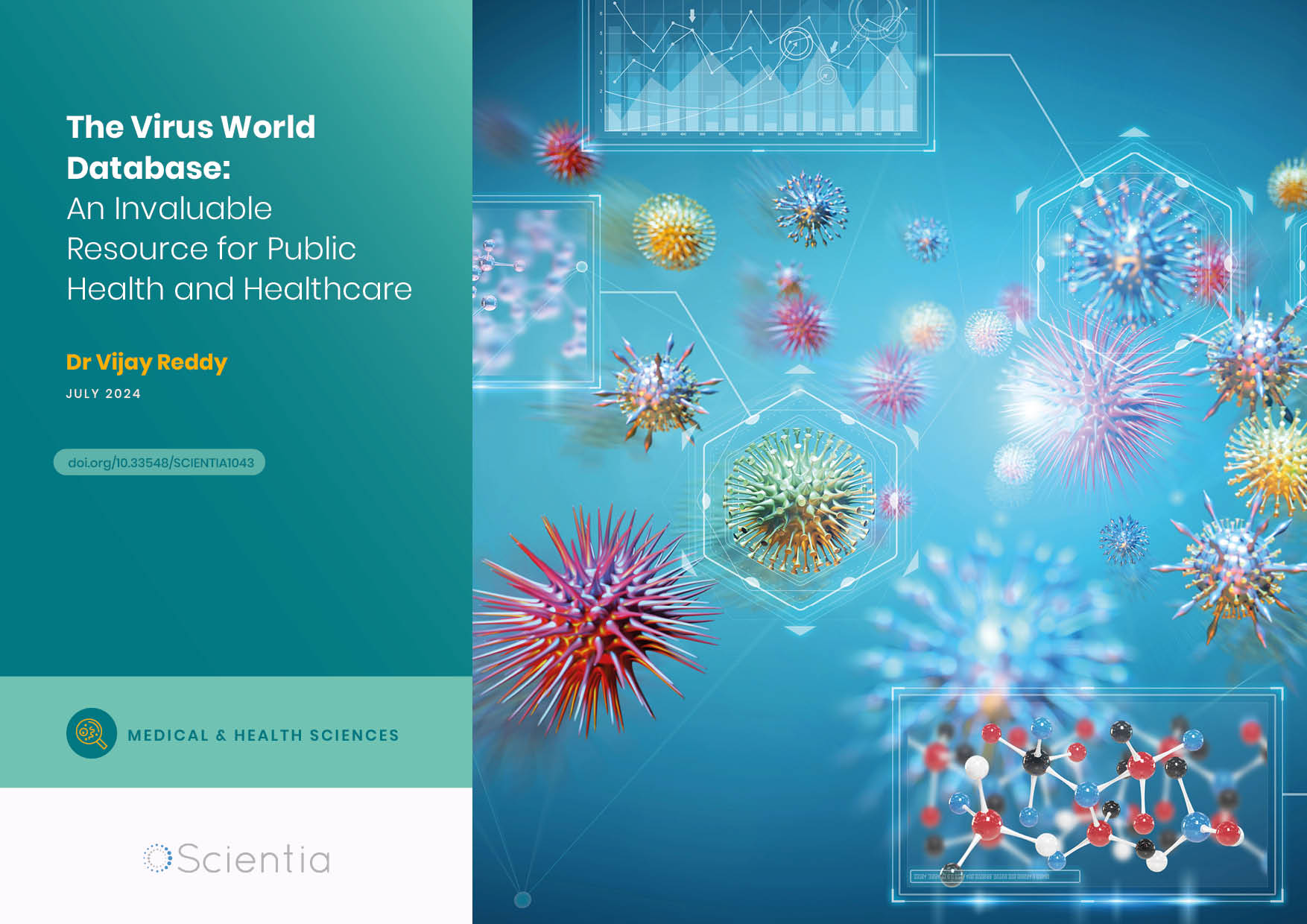
Hypertension, also known as high blood pressure, is an increasingly common condition. Astonishingly, an estimated one-half of adults in the USA have hypertension. Blood pressure is measured in millimetres of mercury (mmHg) representing systolic pressure in the arteries when the heart beats versus diastolic pressure in the arteries when the heart rests between beats. A healthy blood pressure reading for systolic versus diastolic pressure should be between 90/60 mmHg and 120/80 mmHg whilst a measurement of 140/90 mmHg or higher tends to indicate hypertension.
Many factors can increase a person’s risk of high blood pressure including but not limited to being overweight, smoking, being sleep deprived, and being over 65 years old. Because hypertension increases the strain on blood vessels as well as organs such as the heart, kidneys, eyes and brain, it can result in serious co-morbidities. Heart disease, attack or failure may occur, and strokes, kidney disease and vascular dementia are also potential risks.
Fortunately, introducing healthy lifestyle changes can bring down high blood pressure and prevent it from becoming a problem in the first place. Increasing exercise, reducing salt, alcohol and caffeine intake, increasing fruit and vegetable consumption, and stopping smoking are all key interventions. There are also a number of drugs, usually in the form of pills, that clinicians use to try to control hypertension. ACE inhibitors, calcium channel blockers, diuretics, beta-blockers, and angiotensin-II receptor blockers, are all frequently prescribed to patients.
In particular, angiotensin II receptor blockers target the AT1-receptor, thus directly blocking the vasoconstrictive effect of its ligand angiotensin II, which is a part of the renin-angiotensin-aldosterone system that regulates blood pressure. When angiotensin-II is activated, it creates vasoconstriction (the narrowing of blood vessels) which causes elevated blood pressure. It also stimulates the release of another hormone called aldosterone which encourages sodium retention within the kidneys, increasing the overall blood volume and therefore, increasing blood pressure.
The angiotensin-II activation for these processes to take place comes via receptors called angiotensin 1 receptors (AT1R for short). This receptor and the biological activities it promotes are the focus of Dr Sudhir Jain’s research at New York Medical College in the USA.
Utilising Mice Models
Dr Jain studies how genetic differences (more formally and precisely referred to as genetic heterogeneity) impact illness, specifically diseases of the cardiovascular-renal systems – those of the heart and blood vessels and the kidneys. Every human’s genetic makeup is unique and this is in part due to genetic mutations; some of these are harmless but some result in a higher risk of disease. This is because our genes are read by cellular machinery to produce proteins and a consequent wrong amount or wrong types of protein can cause issues. One of the most common mutations in humans are called single nucleotide polymorphisms (SNPs – pronounced ‘snips’). These are characterised by a single nucleotide, a DNA building block, being altered within the genetic sequence. For example, a cytosine nucleotide being swapped for a guanine nucleotide.
Dr Jain researches SNPs within the renin-angiotensin-aldosterone system because they can give insight into how and why hypertension develops. In turn, this will help him to identify novel molecular targets within the system which tailored drugs for hypertension and its co-morbidities could be aimed towards.
To this end, Dr Jain and his colleagues have genetically engineered mice models with the human angiotensinogen gene (the precursor to angiotensin-II) and human AT1R gene variants. These mice are used to investigate how SNPs that are reported in the human population may result in altered regulation of the renin-angiotensin-aldosterone system and predisposition to hypertension.
Inducing SNPs in Angiotensin 1 Receptor Genes
One of Dr Jain’s studies built on the evidence that increased AT1R expression contributes to the onset of high blood pressure. An increase in a receptor’s expression can be due to SNPs appearing on the promoter region of the gene. This is a section of DNA adjacent to the gene of interest to which the DNA transcription machinery will bind and initiate the process of protein synthesis. Therefore, Dr Jain chose a promoter region on the AT1R gene, and its common SNPs, to study in relation to hypertension.
He discovered two separate sets of four specific SNPs that always occur together and named them haplotype-I and haplotype-II; a haplotype is the name for a set of DNA variations. Previous research had revealed that haplotype-I results in increased promoter activity and is associated with hypertension within the Caucasian population. The mice that Dr Jain and his team engineered had either haplotype-I or II in their AT1R genes. They discovered that, in comparison to mice with haplotype-II, those with haplotype-I had higher levels of the mRNA that would go on to aid the synthesis of AT1R proteins. They also showed an increased expression of inflammatory markers, high oxidative stress and higher blood pressure.

Impact of a High-fat Diet
A subsequent study by Dr Jain progressed this research further to look into how diet may impact the expression of the AT1R gene. His initial hypothesis was that a high-fat diet would increase AT1R gene expression via alterations in the transcriptional environment, dependent on the haplotype present. As a result of inflammation and oxidative stress, he predicted that hypertension would occur in haplotype-I mice. Metabolic syndrome, a combination of obesity and high blood pressure, is a known consequence of an overactive renin-angiotensin-aldosterone system. Therefore, Dr Jain set out to understand how diet-induced metabolic syndrome impacts the cellular environment for transcription and its effects on AT1R expression.
Once again, he used engineered mice with haplotype I or II, and fed them a high-fat diet for 20 weeks. After this time, their blood pressure and tissue samples were analysed. In line with his previous results, Dr Jain found that haplotype-I mice showed an increased expression of the AT1R gene, an increase in inflammatory molecules and suppression of antioxidant defences, compared to haplotype-II mice. Although the high-fat diet increased blood pressure in both sets of mice, these factors resulted in more severe hypertension in the haplotype-I mice. This suggests that the presence of the haplotype-I mutations predisposes the mice, and likely humans as well, to hypertension, especially under poor diet conditions.
Older Age Is Also a Risk Factor
This time focusing on a different risk factor for hypertension, Dr Jain carried out a study on the effect of age on AT1R expression. As we age, many of our biological processes alter in some way, so Dr Jain hypothesised that this would be another factor that changed the transcriptional environment for the regulation of AT1R gene expression. And once again, he believed that the haplotype present in the AT1R gene would impact the result of this.
The mice he and the team used for this experiment were either adults aged 10–12 weeks old or aged 20–34 months old. Haplotype I and II mice were present in both of these groups. The results were as he predicted. Aged (i.e., older) mice with haplotype-I displayed increased AT1R expression with higher blood pressure, in addition to suppressed antioxidant defences and anti-ageing molecules. These factors were coupled with elevated inflammatory molecule levels and increased insulin resistance (which can lead to diabetes).
Through these three studies, Dr Jain has revealed vital information that has pushed forward our understanding of hypertension. The results from his mice model experiments strongly suggest that humans who hold a haplotype-I mutation in their AT1R genes are much more likely to experience high blood pressure than those without. In addition, consuming a high-fat diet or being older is likely to increase the risk of hypertension more for these people than others.
Continuing to Investigate Hypertension
Although Dr Jain has already uncovered exciting new findings, he continues to research this important receptor and how different physiological conditions affect its role in hypertension. For example, a recent study examined the role of ageing and high blood pressure in lung pathologies. Perhaps unsurprisingly, aged haplotype-I mice showed worsened lung damage and high mortality after infection.
Another study looked at the impact of a typically Westernised high-fat diet on mice, revealing an increase in blood pressure for both haplotypes I and II mice, but more severely in haplotype-I. Consequent biological changes also resulted in kidney damage and even kidney failure. In his most recent paper, Dr Jain demonstrated how this westernised diet also has negative consequences for the heart in haplotype I and II mice.
Dr Jain has also completed a detailed gene expression analysis of the RNA sequencing data from the heart, kidney, and lung tissues of transgenic mice. This was completed using Ingenuity Pathways Analysis software from Qiagen, which is a web-based bioinformatics application that allows researchers to upload data analysis results from high-throughput experiments such as microarray and next-generation sequencing for functional analysis, integration and further understanding.
Recent work with collaborator Dr Marcello Rotta has extended Dr Jain’s contribution to our understanding of heart pathophysiology. In one study, the researchers used an in vivo approach to explore electrical recovery and diastolic function at the cellular level, substantiating the role of sodium channel subunits. In another study, the researchers explored the alterations in heart rhythm dynamics associated with myocardial infarction in rodents, confirming the validity of this approach in researching the development and manifestation of cardiovascular conditions in humans.
Through his dedicated research, Dr Jain continues to explore the genetic markers regulated by diet, ageing and lifestyle to prevent high blood pressure and its associated health issues. By utilising a range of scientific approaches and methodologies as described here, his work is pioneering a better understanding of heart pathophysiology for the benefit of human health.
SHARE
DOWNLOAD E-BOOK
REFERENCE
https://doi.org/10.33548/SCIENTIA832
MEET THE RESEARCHER

Dr Sudhir Jain
Department of Pathology, Microbiology & Immunology
New York Medical College
Valhalla, NY
USA
Dr Sudhir Jain received his BSc in Zoology, Chemistry and Environmental Science from Delhi University in India before completing an MSc in Zoology at Sagar University. He then went on to achieve his PhD in Life Sciences from Devi Ahilya University in Indore before moving to the USA to serve in various research and teaching roles at New York Medical College. Currently, Dr Jain is an Associate Professor in the Department of Pathology, Microbiology and Immunology. His published work in genetic heterogeneity in pathological conditions associated with cardiovascular-renal systems has earned him a number of awards.
CONTACT
KEY COLLABORATOR
Marcello Rota, PhD, Department of Physiology, New York Medical College, USA
MEMBERSHIPS AND AFFILIATIONS
American Heart Association
The American Physiological Society
New York Academy of Sciences
FUNDING
Hypertension and Inflammation: Novel insights from human AT1R variants
2019-2023: Principal Investigator: RO1, National Institute of Health
Myocyte Repolarization and Cardiac Dysfunction with Age
2018-2023: Collaborator (PI: Marcello Rota): R01, National Institute of Aging
FURTHER READING
DO Cervantes, E Pizzo, H Ketkar, et al., Scn1b Expression in the Adult Mouse Heart Modulates Na+ Influx in Myocytes and Reveals a Mechanistic Link between Na+ Entry and Diastolic Function, American Journal of Physiology-Heart and Circulatory Physiology, 2022, 332(6), H975–H993. DOI: https://doi.org/10.1152/ajpheart.00465.2021
S Jain, N Puri, A Rana, N Sirianni, et al., Metabolic Syndrome Induces Over Expression of the Human AT1R: A Haplotype-Dependent Effect With Implications on Cardio-Renal Function, American Journal of Hypertension, 2018, 31(4), 495–503. DOI: https://doi.org/10.1093/ajh/hpx176
S Jain, A Rana, K Jain, et al., Age-Related Expression of Human AT1R Variants and Associated Renal Dysfunction in Transgenic Mice, American Journal of Hypertension, 2018, 31(11), 1234–1242. DOI: https://doi.org/10.1093/ajh/hpy121
S Jain, J Tulsulkar, A Rana, et al., Transgenic mice overexpressing human angiotensin I receptor gene are susceptible to stroke injury, Molecular Neurobiology, 2016, 53(3), 1533–1539. DOI: https://doi.org/10.1007/s12035-015-9109-2

REPUBLISH OUR ARTICLES
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence (CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
SUBSCRIBE NOW
Follow Us
MORE ARTICLES YOU MAY LIKE
Dr Ralf Adam | New Technologies Shaping the Future of Oral Hygiene
Understanding the efficiency of various toothbrush technologies is essential for achieving optimal oral health. Dr Ralf Adam, who leads a dedicated team at Procter & Gamble in Germany, is keen to investigate the complexities of these technologies. His team have provided new insights into the best toothbrush types for plaque removal and the maintenance of gum health. By highlighting the importance of informed oral care decisions and ongoing investigations, this vital research works towards ensuring everyone can achieve a brighter, healthier smile.
Dr Toby Phesse | Revealing the Mysteries of Wnt Signalling: Novel Approaches to Beating Cancer
Cancer remains a leading cause of mortality worldwide, and the need for new, more effective treatments remains an urgent challenge. Dr Toby Phesse from Cardiff University in the UK focuses on the role of the Wnt receptor found on the surface of cells and its involvement with cell communication and cancer growth, bringing fresh hopes for new therapeutic options.
Dr Vijay Reddy | The Virus World Database: An Invaluable Resource for Public Health and Healthcare
Severe viral disease presents an ongoing challenge to the health of humankind. While unparalleled developments in science and technology are improving our understanding of such viruses, this information needs to be readily accessible to researchers to ensure continued progress in public health and healthcare. Dr Vijay Reddy and his colleagues at the Hormel Institute (University of Minnesota) developed the Virus World database, an invaluable resource that details the genome, structure, and host of practically every discovered virus to date.
Professor Ralf Herwig | Deciphering the Enigma of Vitamin D and the Immune System
Vitamin D has been studied as a treatment for a large number of diseases and conditions, from cancer to autism to COVID-19. However, its mode of action is not completely understood. Professor Ralf Herwig carries out his research at HG Pharma GmbH (Austria) and Ulster University (UK). His vital work explores the role of vitamin D in the body with a view to unlocking its potential as a treatment for a variety of health conditions involving the immune system.