Dr Uwe Grether – Targeting the Cannabinoid CB2 Receptor for Novel Anti-inflammatory Therapeutics
On the surface of many of our cells, intriguing receptors called cannabinoid receptors reach out into their environment. The CB2 subtype of this receptor family is expressed largely on our immune cells and plays an important role in inflammatory responses, making it a promising target for anti-inflammatory therapeutics. To produce effective drugs, the molecules that bind to the CB2 receptor, known as ligands, need to be characterised. Dr Uwe Grether from F. Hoffmann-La Roche Ltd has dedicated his research to discovering useful CB2 receptor ligands and utilising them in the development of novel anti-inflammatory therapeutics.
Ligands and Receptors
Receptors are important protein structures that allow cells to interact with the world around them (if they are on the cell surface) or help to control the activities within the cell (if they are intracellular). Cell-surface receptors reach out into their environment and bind to molecules that have the right complementary structure. The specific molecules that bind to a given receptor are known as its ligand. Ligands and receptors that bind strongly together are said to have a high affinity for each other and more energy is needed to pull them apart.
Although there are hundreds of different types of receptors, those on the surface can be placed into one of three groups: ligand-gated ion channels, receptor tyrosine kinases and G protein-coupled receptors (GPCR). The latter are characterised by their protein structure weaving in and out of the cell membrane seven times, creating seven protein segments. An area of the protein within the cell can bind to a G protein which gives the receptor its name. The arrival and binding of a ligand activate the G protein and induce a signalling pathway across the cell that eventually leads to a response.
The type of response depends on the type of receptor and ligand interacting. For example, around 800 GPCR in humans are believed to bind to a specific scent molecule and their response results in us smelling the associated odour. Another type is that of cannabinoid receptors, named after their ligands called cannabinoids. Many people may associate the word cannabinoid with the cannabis plant (for good reason). Plant cannabinoids such as tetrahydrocannabinol are one of the three groups of cannabinoid ligands. However, they can also be produced by mammals (called endocannabinoids) or produced artificially (synthetic cannabinoids).
Currently, there are two known cannabinoid receptors, known as CB1 and CB2, although more may await discovery. The CB1 receptor is mainly found in areas of the central nervous system like the brain but also in the kidneys, liver and lungs. In contrast, CB2 receptors are mainly found within the immune system and in the cells that multiply to create other blood cells – haematopoietic stem cells. During pathological stimuli, there is evidence that the expression of CB2 receptor increases, meaning that more of the protein is produced and placed on cell surfaces. An agonist (any ligand that activates the receptor) then initiates a signalling cascade that prevents or treats an illness. While this binding of an inverse agonist could worsen the illness, in some rare cases, the opposite has been reported for CB2.
These findings converge to suggest that the CB2 receptor is involved in certain disease processes and is, therefore, a promising drug target. Uncovering the intricate structure and function of CB2 receptors and their ligands is the work of Dr Uwe Grether, who leads his collaborative research at the biotechnology company, F. Hoffmann-La Roche, based in Basel, Switzerland.

Characterising Cannabinoid Receptor Ligands
Due to its distribution concentrated in the immune system, the CB2 receptor as a therapeutic target generated considerable interest in relation to inflammatory disease and tissue injuries. In fact, drugs for these disorders have already been widely developed and utilised. However, precisely how they work in cells is an ongoing task for Dr Grether, who is working to verify the molecular mode of action of CB2 receptor ligands as a therapy and their selectivity for the receptor. He also studied the associated pharmacokinetic properties, that is, how the drug is absorbed, distributed, metabolised and excreted over time throughout the body.
Through extensive collaboration with multiple academic and industry laboratories, Dr Grether comprehensively profiled the most widely used CB2 receptor ligands. He and his team discovered key differences in how a variety of ligands activate specific signalling pathways in a cell and their ability to interact with undesired proteins, known as off-target effects. They also found three highly selective CB2 receptor ligands to recommend in the study of the role of the CB2 receptor in biological and pathological processes: HU910, HU308 and JWH133. These ligands also have a promising pharmacokinetic profile.
Dr Grether believes that this in-depth study will set the standard for CB2 receptor experiments in the future because his team has created the largest data set of cannabinoid receptor ligands under the same experimental conditions. These novel characterisations can be used all over the world, and not just for cannabinoids. Importantly, the approach that the research team took could be used to characterise other therapeutic targets for all kinds of drug discovery research.
‘This probe is the first of its kind to preserve both a strong affinity to the CB2 receptor as well as its agnostic efficacy once bound, shown in both human and mice cells.’
Reproduced from https://doi.org/10.1039/D1SC06659E with permission from the Royal Society of Chemistry.
Ocular Inflammation Treatment
The ligands that Dr Grether investigated in this early work are described as agonists – a type of ligand that induces normal activity when bound. In the case of CB2, the natural ligands are agonists, whereas other types of ligands have opposing effects (such as inverse agonists). In his next study, Dr Grether studied a mixture of these ligands for their use in therapeutics, specifically ocular inflammation.
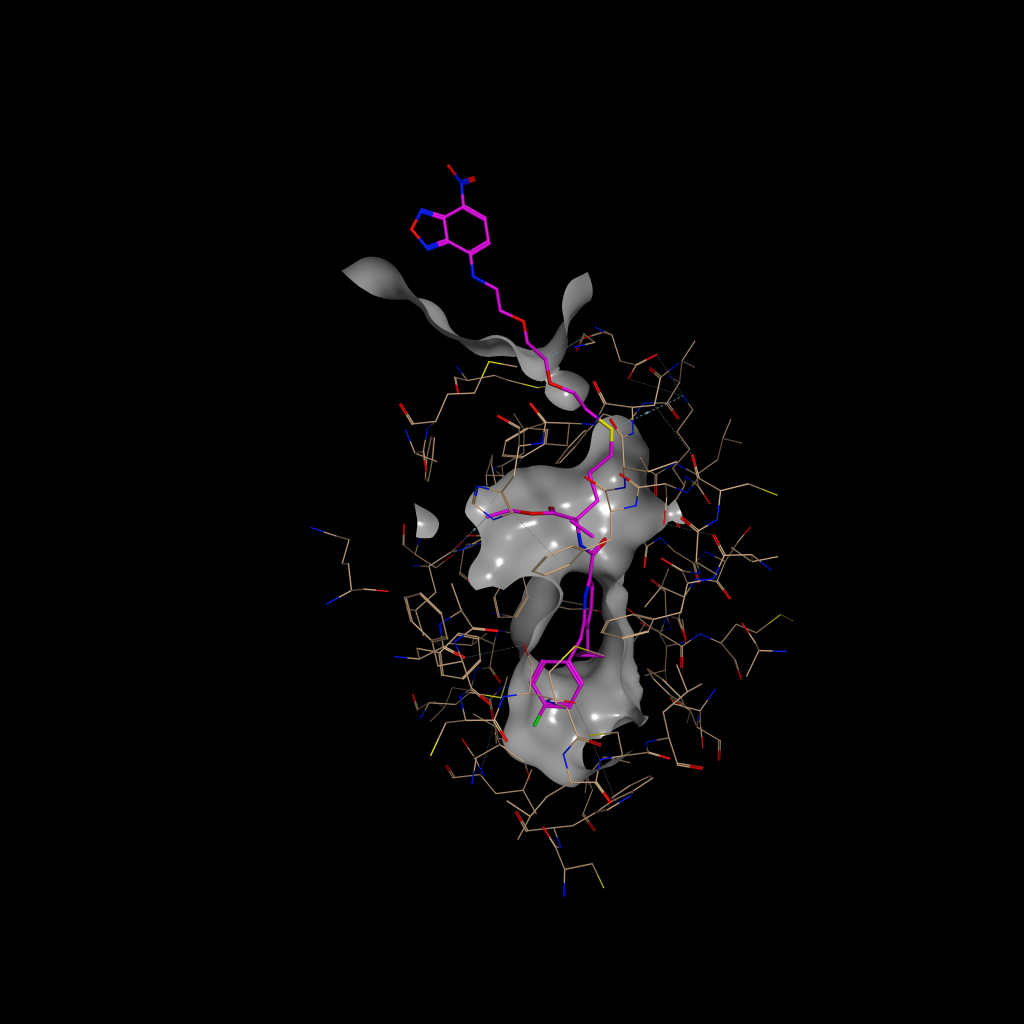
He focussed on two CB2 receptor agonists, called RO6871304 and RO6871085, and an inverse agonist, RO6851228. Although these ligands are all structurally unique, they are also highly selective for the receptor, meaning they don’t attach to other receptors to produce unwanted reactions. Using computer models (in silico) and cell-based (in vitro) tests, Dr Grether and the team studied the interactions between these ligands and the receptor including binding and cell signalling. Furthermore, they characterised the absorption, distribution, metabolism, excretion and toxicology profile of the ligands.
Dr Grether revealed that the two agonists had a strong affinity for CB2 receptors and once bound, they reduced the inflammatory effects of various immune cells in the eye. Therefore, drugs containing these ligands may be useful for targeting the CB2 receptor to enhance its anti-inflammatory activities. To treat eye inflammation, these drugs could be administered topically as well as orally to treat inflammation throughout the eye. As a result, patients could receive effective treatment and prevent consequent ocular tissue damage.
Reproduced from https://doi.org/10.1039/D1SC06659E with
permission from the Royal Society of Chemistry
Developing Novel Fluorescent Probes for CB2 Receptors
Dr Grether has also carried out investigations into the development of probes to enable other studies into CB2 receptors and their distribution and activities within both healthy and diseased cells. These probes are fluorescent, which means that when a specific wavelength of light is shone on them, its molecules absorb the light and then re-emit a different specific wavelength. This new wavelength of light can then be detected and viewed in colourful images. Because the fluorescent probes are attached to a protein of interest, the fluorescence that is detected indicates the location of that protein.
However, attaching a fluorescent probe to a protein is not as simple as it may sound. Such probes have to be carefully designed so that they can attach to a certain part of the protein of interest. In a recent study, and another collaborative effort, Dr Grether led an innovative group to design a collection of highly specific CB2 receptor fluorescent probes which were successfully used in a wide range of cell types, in different species and for a number of applications.
This is an exciting project, not least because it will allow Dr Grether and other scientists around the world to better understand the CB2 receptor, its cell signalling mechanisms and expression throughout the body, tissues and cells. Critically, Dr Grether has facilitated the continued study of the receptor, driving forward new developments in the treatment of autoimmune disorders, inflammatory diseases and even cancer.

Developing More Effective CB2 Receptor-based Drugs
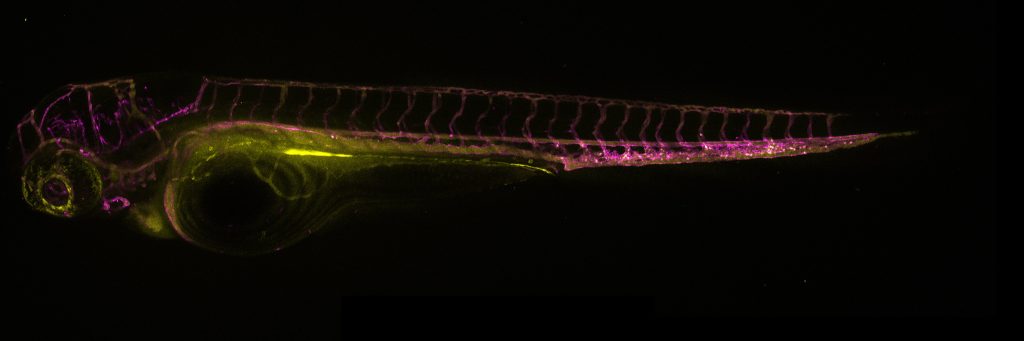
Further building on this work, one of Dr Grether’s most recent projects has resulted in another new fluorescent probe. As he explains, ‘This probe is the first of its kind to preserve both a strong affinity to the CB2 receptor as well as its agonistic efficacy once bound, shown in both human and mice cells.’ When applied in zebrafish larvae (a common model organism for scientific research), this fluorescent probe facilitated live cell imaging. Excitingly, this allowed the team to observe the functions of the CB2 receptor within the cells in real-time, allowing them to gain a better understanding of its function. The dedicated work of Dr Grether is playing a vital role in advancing knowledge of this important cannabinoid receptor in the quest to develop more effective CB2 receptor-based drugs.
SHARE
DOWNLOAD E-BOOK
REFERENCE
https://doi.org/10.33548/SCIENTIA817
MEET THE RESEARCHER

Dr Uwe Grether
F. Hoffmann-La Roche Ltd
Basel
Switzerland
Dr Uwe Grether studied undergraduate and postgraduate chemistry at the University of Karlsruhe in Germany before undertaking a postdoctoral fellowship at Oregon State University in Corvallis, USA. Since then, Dr Grether has worked at F. Hoffmann-La Roche in Basel, Switzerland. Starting as a medicinal chemist in diabetes, he went on to study dyslipidaemia, kidney and inflammatory diseases, and also ophthalmology. Currently, Dr Grether is now project leader of research programs, focussing on the central nervous system and immunology. He dedicates much of his research to the endocannabinoid system, and in particular the cannabinoid receptor CB2 and its ligands, due to their implication in inflammation disorders and consequently, their exciting potential for drug targeting.
CONTACT
W: https://www.roche.com/
Twitter: @Roche
KEY COLLABORATORS
Erick M. Carreira, Laboratorium für Organische Chemie, Eidgenössische Technische Hochschule Zürich, Vladimir-Prelog-Weg 3, 8093 Zürich, Switzerland
Melanie Kelly, Department of Pharmacology, Dalhousie University, Halifax, Nova Scotia, Canada
Mauro Maccarrone, Department of Biotechnological and Applied Clinical Sciences, University of L’Aquila (Italy) & European Center for Brain Research, Santa Lucia Foundation, 00128 Rome, Italy.
Marc Nazaré, Leibniz-Forschungsinstitut für Molekulare Pharmakologie FMP, Campus Berlin-Buch, 13125 Berlin, Germany
Pal Pacher, National Institute on Alcohol Abuse and Alcoholism, National Institutes of Health, Rockville, MD 20852, USA
Mario van der Stelt, Department of Molecular Physiology, Leiden Institute of Chemistry, Leiden University, 2333 CC, Leiden, The Netherlands
Dmitry B. Veprintsev, Faculty of Medicine & Health Sciences, University of Nottingham, Nottingham NG7 2UH, England, United Centre of Membrane Proteins and Receptors (COMPARE), University of Birmingham and University of Nottingham, UK
FURTHER READING
T Gazzi, B Brennecke, K Atz, et al., Detection of Cannabinoid Receptor Type 2 in Native Cells and Zebrafish with a Highly Potent, Cell-Permeable Fluorescent Probe, Chemical Science, 2022. DOI: https://doi.org/10.1039/D1SC06659E
A Haider, LC Gobbi, J Kretz, et al., Identification and Preclinical Development of a 2,5,6-Trisubstituted Fluorinated Pyridine Derivative as a Radioligand for the Positron Emission Tomography Imaging of Cannabinoid Type 2 Receptors, Journal of Medicinal Chemistry, 2020, 63, 18, 10287-10306.
RF Porter, A-M Szczesniak, JT Toguri et al., Selective Cannabinoid 2 Receptor Agonists as Potential Therapeutic Drugs for the Treatment of Endotoxin-Induced Uveitis, Molecules, 2019, 24(18), 3338. DOI: https://doi.org/10.3390/molecules24183338
RC Sarott, MV Westphal, P Pfaff, et al., Development of High-Specificity Fluorescent Probes to Enable Cannabinoid Type 2 Receptor Studies in Living Cells, Journal of American Chemical Society, 2020, 142(40), 16953–16964. DOI: https://doi.org/10.1021/jacs.0c05587
M Soethoudt, U Grether, J Fingerle, et al., Cannabinoid CB2 receptor ligand profiling reveals biased signalling and off-target activity, Nature Communications, 2017, 8, 13958. DOI: https://doi.org/10.1038/ncomms13958

REPUBLISH OUR ARTICLES
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence (CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
SUBSCRIBE NOW
Follow Us
MORE ARTICLES YOU MAY LIKE
Dr Ralf Adam | New Technologies Shaping the Future of Oral Hygiene
Understanding the efficiency of various toothbrush technologies is essential for achieving optimal oral health. Dr Ralf Adam, who leads a dedicated team at Procter & Gamble in Germany, is keen to investigate the complexities of these technologies. His team have provided new insights into the best toothbrush types for plaque removal and the maintenance of gum health. By highlighting the importance of informed oral care decisions and ongoing investigations, this vital research works towards ensuring everyone can achieve a brighter, healthier smile.
Dr Toby Phesse | Revealing the Mysteries of Wnt Signalling: Novel Approaches to Beating Cancer
Cancer remains a leading cause of mortality worldwide, and the need for new, more effective treatments remains an urgent challenge. Dr Toby Phesse from Cardiff University in the UK focuses on the role of the Wnt receptor found on the surface of cells and its involvement with cell communication and cancer growth, bringing fresh hopes for new therapeutic options.
Dr Vijay Reddy | The Virus World Database: An Invaluable Resource for Public Health and Healthcare
Severe viral disease presents an ongoing challenge to the health of humankind. While unparalleled developments in science and technology are improving our understanding of such viruses, this information needs to be readily accessible to researchers to ensure continued progress in public health and healthcare. Dr Vijay Reddy and his colleagues at the Hormel Institute (University of Minnesota) developed the Virus World database, an invaluable resource that details the genome, structure, and host of practically every discovered virus to date.
Professor Ralf Herwig | Deciphering the Enigma of Vitamin D and the Immune System
Vitamin D has been studied as a treatment for a large number of diseases and conditions, from cancer to autism to COVID-19. However, its mode of action is not completely understood. Professor Ralf Herwig carries out his research at HG Pharma GmbH (Austria) and Ulster University (UK). His vital work explores the role of vitamin D in the body with a view to unlocking its potential as a treatment for a variety of health conditions involving the immune system.