Professor Carmen Amaro – Dr Carla Hernández-Cabanyero | Vibrio vulnificus and Rising Ocean Temperatures: A Threat to Human and Marine Life
Vibrio vulnificus is a bacteria found in warm aquatic ecosystems around the world, capable of infecting and killing both humans and fish. As global ocean temperatures rise, the habitat of this organism is extending, and so are the numbers of infections it causes. Dr Amaro and Dr Hernández-Cabanyero from the University of Valencia are working to understand the biology and lifecycle of this organism so that we can control its growth and virulence to protect fish stocks and human lives.
Climate Change and Food Security
Climate change is arguably the biggest challenge facing humanity. Climate change can be defined as the long-term changes seen in temperatures and weather patterns across the globe. Human activities are driving these changes by producing large amounts of heat-trapping gases in the atmosphere.
It is widely recognised that climate change is having a clear and significant impact on people’s lives worldwide. The consequences include the loss of homes and the displacement of individuals due to rising sea levels and extreme weather events like hurricanes and wildfires. Moreover, increasing temperatures are influencing water availability and public health. Climate change also has implications for global food security, as changes in temperature and rainfall patterns contribute to crop failures. Additionally, rising sea temperatures have had repercussions on fish and shellfish stocks, affecting their habitats and enabling the spread of some of its natural pathogens, such as the emerging pathogen Vibrio vulnificus (V. vulnificus), into new areas.
Vibrio vulnificus: An Emerging Zoonotic Pathogen and Marker of Climate Change
V. vulnificus is a type of bacteria found in temperate coastal ecosystems around the world. It can infect and cause disease in a range of hosts, including fish and humans. Moreover, it is considered a zoonotic pathogen because it can be transmitted between species from animals – usually infected fish – to humans.
Humans can be infected with V. vulnificus by eating raw seafood or by having an open wound that comes into contact with seafood or seawater containing the bacteria. Infection results in severe gastroenteritis or dangerous wound infections, both of which can lead to sepsis – an overwhelming immune reaction to a bloodstream infection. These infections can be life-threatening, particularly in people with high iron levels in their blood due to underlying disease. When infections lead to sepsis, there is a mortality rate of 50%, and the majority of these deaths occur within 48 hours of infection.
Due to V. vulnificus’s requirement for a warm habitat, increases in ocean temperatures caused by climate change have allowed this bacterium to spread from its traditional habitat, resulting in increased infections of both fish and humans. In the Eastern United States, wound infections caused by V. vulnificus have increased eight-fold between 1988 and 2018, and the bacteria have recently been found much further north than ever before – a phenomenon which has officially been attributed to climate change. V. vulnificus is now considered a biological marker of climate change as the number of human and animal infections increase simultaneously and proportionately with increases in seawater temperature.
Dr Carmen Amaro, Dr Carla Hernández-Cabanyero, and a team of researchers from the University of Valencia in Spain are experts in the biology, evolution and host-pathogen interactions of Vibrio bacteria, including V. vulnificus. They have conducted research into factors that influence the lifecycle of V. vulnificus as well as the mechanisms that allow V. vulnificus to infect and cause sepsis in two very different hosts: humans and fish.
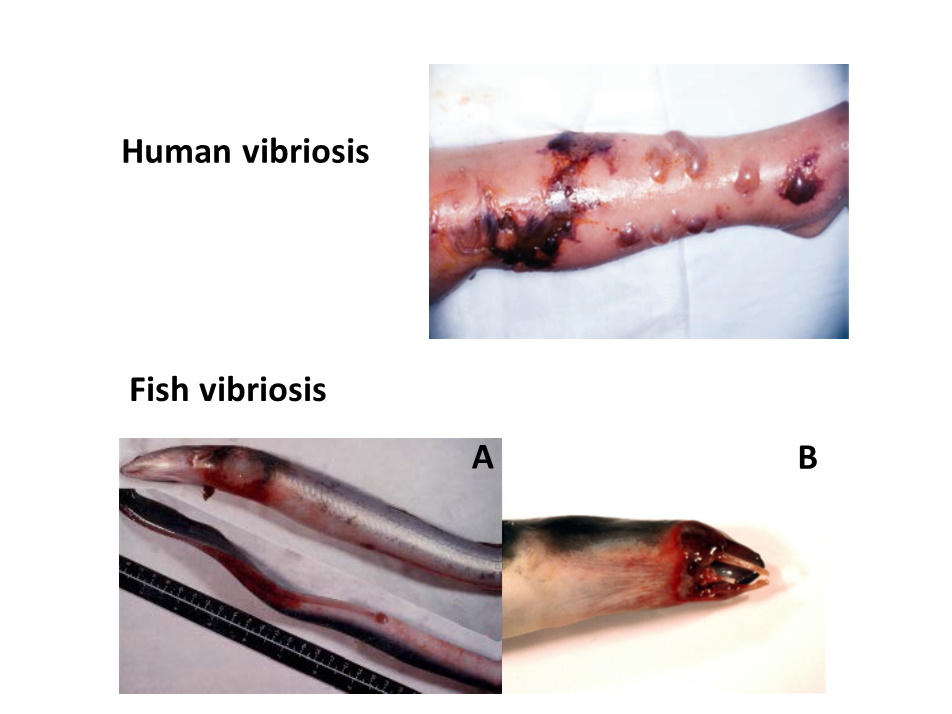
Clinical signs of human and fish vibriosis caused by V. vulnificus. Human vibriosis: A patient showing typical clinical signs of primary and secondary sepsis caused by V. vulnificus: swelling, erythema, development of vesicles or bullae and tissue necrosis. Image credit to Dr Ching-Chuan Liu, Department of Pediatrics, National Cheng Kung University Hospital, National Cheng Kung University, Tainan City, Taiwan). Eel vibriosis: haemorrhagic septicaemia caused by contact (A) or by the oral route (B). Image credit to C. Amaro and B. Fouz.
The Impact of Environment on Lifecycle
In humans, the severity of V. vulnificus infection is linked to levels of iron in the blood, and people who have abnormally high levels of blood iron (due to conditions such as haemochromatosis, cirrhosis and other liver diseases) are more likely to develop sepsis and die from the disease.
This led Dr Amaro and Dr Hernández-Cabanyero to question what impact iron was having on the biology of V. vulnificus. They conducted a series of experiments to analyse the response of V. vulnificus to changes in iron levels, whereby the bacteria were exposed to different levels of iron, and compared the expression of different genes. Analysis showed that around 25% of V. vulnificus’s genes, mostly those related to bacterial metabolic activity, motility, protection against stress, including host immunity, and virulence factors such as toxins, are impacted by iron, with different effects seen at high and low concentrations. These results confirmed that iron plays a critical role in both the environmental survival and virulence of V. vulnificus.
Iron levels serve as a distinctive indicator, enabling bacteria to identify their surroundings and activate specific genes necessary for their survival and reproduction. Environmental waters typically contain low levels of free iron, inducing the bacteria into dormant or resistant states (biofilms, for example). On the contrary, in fish farm environments, the water typically contains moderate levels of iron, facilitating bacterial mobility and their ability to locate and adhere to fish or human skin, especially in open wounds. Upon entering the bloodstream, the bacteria detect a shift in iron levels, which is different depending on the host (high in susceptible humans and low in fish), triggering the production of virulence factors responsible for causing diseases. In the case of humans, the greater the presence of iron in the blood (i.e., susceptible patients), the higher the capacity of V. vulnificus to survive, multiply, and generate virulence factors.
Medical reports and data from fish farms have suggested that the severity of V. vulnificus infection is strongly influenced by water temperature, especially in the case of fish, with bacteria recovered from warmer water causing more extreme disease. To understand the role of temperature in the generation of a virulent phenotype in V. vulnificus, the team grew bacteria at four different temperatures and then compared gene expression and different physical markers of virulence. They grew bacteria at 20°C as no infections have been linked to this temperature, at 25°C as this is the temperature that moderate outbreaks in fish farms take place, at 28°C as this is the temperature at which high levels of mortality are seen at in fish outbreaks, and 37°C to represent human infection.
The team found that higher temperatures led to an increase in the expression of cell functions which help the bacteria infect a host and cause disease. At higher temperatures, bacteria grow more quickly, which allows for higher numbers in water and greater ability to move, helping them reach suitable hosts, produce a protective layer to shield themselves from the host immune system, and produce molecules that break down host cells and tissues allowing them to colonise body sites. These results complement existing knowledge and highlight the role environmental temperature plays in determining the biology and life cycle of V. vulnificus. At low temperatures, the bacteria are not able to infect humans or fish. Increasing water temperature leads to changes in the bacteria that make it better for infecting hosts and more likely to cause severe disease.
The combination of this study of temperature and the team’s previous work on the impact of iron come together to show that both factors influence the lifestyle and virulence of V. vulnificus. Whilst some virulence factors are influenced by temperature (growth ability and motility, for example), the main toxins and enzymes that are known to lead to death from sepsis are both influenced by iron levels but not temperature. Higher ocean temperatures caused by climate change allow the bacteria to be in higher concentrations in the environment, increasing the probability of infection and, at the same time, activating traits allowing them to more effectively infect fish and humans. Then, iron concentration in the host blood (low in healthy fish and high in susceptible humans) leads to the production of factors which, together, cause sepsis.
How One Type of Bacteria Can Infect Two Very Different Hosts
Very few pathogens can cause similar diseases in evolutionary distant host species. V. vulnificus can cause sepsis in both fish and humans – two very different organisms. To understand this, Dr Amaro and Dr Hernández-Cabanyero compared the genes expressed by a zoonotic strain of V. vulnificus, known to be able to infect both eels and humans, in the blood of both species. They discovered that high levels of iron were needed for infection only in human blood, showing that different mechanisms are activated in different host organisms, and highlighting the role of iron as a marker that triggers host-specific virulence.
When in human blood containing high levels of iron (susceptible humans), V. vulnificus produces an envelope with a thick protective capsule – a layer of slime that protects the bacteria from the host immune system and produces toxins that aggravate sepsis. When in human blood without high iron levels (healthy humans), the bacteria were not able to produce this thick capsule layer and were unable to grow in blood. This is why sepsis is more likely to occur in people with high blood iron.
In contrast, when V. vulnificus infected eels, it produced an envelope which contained two outer membrane proteins specific for fish. The researchers have described these two outer membrane proteins as a ‘survival in fish blood kit’, as they protect the bacteria from the fish immune system, allowing it to replicate within the fish bloodstream, and then cause severe disease symptoms.
Other scientists’ work has shown that these zoonotic strains cause rapid death in mammals by cytokine storm sepsis, but little is known about V. vulnificus disease development in fish. Cytokines are proteins produced by immune cells, such as leukocytes, to help regulate and coordinate the body’s defence against infection. In a cytokine storm, too many cytokines are produced, and this damages tissues and can ultimately lead to organ failure.
To understand how V. vulnificus kills fish, the team again used an eel model. They found that V. vulnificus triggers a strong but unusual inflammatory response in which red blood cells seem to play an active role. As well as providing information on disease progression, this study informed the design of a diagnostic assay for the early detection of V. vulnificus sepsis in fish, which could be very helpful in preventing the spread of the disease in fish farms – frequent ‘hotspots’ for V. vulnficus outbreaks.
The Future of Vibrio vulnificus
Dr Amaro’s and Dr Hernández-Cabanyero’s work on V. vulnificus has provided important information on its survival, both in the environment and host, and its ability to cause disease. As our oceans grow warmer, V. vulnificus will be able to infect fish and humans across an even broader geographical range, with important ramifications for both food security and public health. The more we understand about this unique pathogen, the better equipped we are to prevent and treat the disease.
SHARE
DOWNLOAD E-BOOK
REFERENCE
https://doi.org/10.33548/SCIENTIA942
MEET THE RESEARCHERS

Professor Carmen Amaro
University of Valencia
Valencia
Spain
Professor Carmen Amaro is an expert in pathogenesis and microbial evolution, specialising in Vibrio vulnificus (V. vulnificus), an aquatic zoonotic pathogen related to climate change. She founded the PAFZP (Pathogens in Aquaculture, Fish and Zoonoses) team in 2004. This team conducts both fundamental academic research and applied research for the aquaculture-related industry. Professor Amaro is considered a world authority on the biology and diseases (vibriosis) caused by V. vulnificus. She ranks third in the world in the number of articles on V. vulnificus and first in the number of articles on vibriosis, according to the Scopus database. She has trained more than 50 researchers within his research team, supervising Spanish and foreign postdoctoral, predoctoral and postgraduate students. She is a permanent member of the organising committee of international congresses on Vibrio and collaborates on papers for the World Health Organization and the Food and Agriculture Organization of the United Nations.
CONTACT
W: https://www.uv.es/uvweb/college/en/profile-1285950309813.html?p2=amaro&idA=true

Dr Carla Hernández-Cabanyero
Department of Fundamental Microbiology
University of Lausanne
Switzerland
Dr Carla Hernández-Cabanyero received her PhD from the University of Valencia in 2020, working on the host-pathogen interaction of the aquatic zoonotic pathogen Vibrio vulnificus (V. vulnificus), an emerging zoonotic pathogen linked to global warming. Her PhD was financed by a grant awarded through public competition by the Spanish Ministry of Science and Innovation. The main objective of her thesis was to unravel the role of environmental factors (i.e., iron levels, temperature and infected host) on the bacterial virulence traits, as well as to determine the host immune response after infection. From 2020 to 2021, she was a postdoctoral researcher at the Institute of Biotechnology and Biomedicine of the University of Valencia, working in Vibrio spp. infection models. In January 2022, she joined the Microbiota-Targeted Interventions Group at the Department of Fundamental Microbiology (UNIL, Lausanne) as a postdoctoral scientist, where she is studying gut microbiota-host interactions and how they contribute to small intestinal inflammation and nutrient absorption. She is part of Professor Carmen Amaro’s research team as an external collaborator. Dr Hernández-Cabanyero also plays an active role in teaching students and engaging the general public in science communication outreach.
CONTACT
E: carla.hernandezcabanero@unil.ch
W: https://wp.unil.ch/vonaeschlab/group-members/
MAIN COLLABORATORS CONTRIBUTING TO THIS RESEARCH
Professor Lluis Tort Bardolet (Autonomous University of Barcelona, Spain)
Professor Luis Mercado (Pontifical Catholic University of Valparaíso, Chile)
CURRENT FUNDING
THINKINAZUL/2021/027 from MCIN (Ministerio de Ciencia e Innovación de España) with funding from European Union NextGeneration EU (PRTR-C17.I1) and GV (Generalitat Valenciana)
PID2020-120619RB-I00 funded by MCIN/AEI/10.13039/501100011033
CIAICO/2021/293 funded by Conselleria de Innovación, Universidades, Ciencia y Sociedad Digital (Generalitat Valenciana, Spain)
Dr Carla Hernández-Cabanyero has also been funded by the FPI grant BES-2015-073117 (Ministerio de Ciencia, Innovación y Universidades, Spain)
FURTHER READING
C Hernández-Cabanyero, E Sanjuán, FE Reyes-López, et al., Transcriptomic Study Reveals That Fish Vibriosis Due to the Zoonotic Pathogen Vibrio vulnificus Is an Acute Inflammatory Disease in Which Erythrocytes May Play an Important Role, Frontiers in Microbiology, 2022, 13, 852677. DOI: https://doi.org/10.3389/fmicb.2022.852677
C Hernández-Cabanyero, E Sanjuán, B Fouz, et al., The Effect of the Environmental Temperature on the Adaptation to Host in the Zoonotic Pathogen Vibrio vulnificus, Frontiers in Microbiology, 2020, 11, 489. DOI: https://doi.org/10.3389/fmicb.2020.00489
C Hernández-Cabanyero, CT Lee, V Tolosa-Enguis, et al., Adaptation to host in Vibrio vulnificus, a zoonotic pathogen that causes septicemia in fish and humans, Environmental Microbiology, 2019, 21(8), 21(8), 3118–3139. DOI: https://doi.org/10.1111/1462-2920.14714
D Pajuelo, C Hernández-Cabanyero, E Sanjuan, et al., Iron and Fur in the life cycle of the zoonotic pathogen Vibrio vulnificus, Environmental Microbiology, 2016, 18(11), 4005–4022. DOI: https://doi.org/10.1111/1462-2920.13424


REPUBLISH OUR ARTICLES
We encourage all formats of sharing and republishing of our articles. Whether you want to host on your website, publication or blog, we welcome this. Find out more
Creative Commons Licence (CC BY 4.0)
This work is licensed under a Creative Commons Attribution 4.0 International License. 
What does this mean?
Share: You can copy and redistribute the material in any medium or format
Adapt: You can change, and build upon the material for any purpose, even commercially.
Credit: You must give appropriate credit, provide a link to the license, and indicate if changes were made.
SUBSCRIBE NOW
Follow Us
MORE ARTICLES YOU MAY LIKE
Nick Martin | Data Assimilation: Overcoming AI’s Data Uncertainty Limitations for Water Resources
Water resources are essential for human life. Knowing how to manage water, both now and in the future, is necessary to continue using it as well as possible. Nick Martin and Jeremy White are examining limitations to artificial intelligence applications in water resources generated from noisy and estimated data sets. For poor quality data sets, they found that machine learning models will perform poorly relative to tools that explicitly include physics-based descriptions of physical processes; this is because physics-based calculations can use both data and physics knowledge through data assimilation techniques.
Nick Martin | The Future of Floods: Smarter Risk Tools for Sustainable Water Management in a Changing Climate
Sustainable decision-making requires balancing the costs borne by today’s society with those that will fall on future generations. Climate change is intensifying extreme weather, making floods more severe because a warmer atmosphere can hold and deliver a larger volume of water as precipitation. It may also be the case that severe floods are becoming more frequent as drought becomes more frequent, average conditions rarely occur, and weather oscillates between short duration wet and long duration dry extremes. Worryingly, traditional infrastructure (often designed using outdated, backward-looking models) risks failing under these evolving conditions.
Nick Martin from Vodanube LLC, and his colleagues have applied Probabilistic Risk Assessment (PRA) to flood inundation. Their research optimises current adaptation and future mitigation strategies, even while acknowledging PRA’s limitations. The team demonstrates how this approach can guide more resilient water resource management, and highlights opportunities for further study.
Dr James D. Burrington | Fuelling Hydrogen’s Role in a Net Zero Future
Hydrogen is often touted as the fuel of the future, but how much can it really contribute to global decarbonisation? Dr James D. Burrington, founder of NiceChemistry.com, has modelled how hydrogen technologies, particularly green hydrogen, might support worldwide net zero goals. His research applies rigorous metrics to assess energy efficiency, cost, emissions, and land use. This revealed that, while hydrogen may not directly replace electricity, it could be critical in decarbonising sectors where electrification falls short.
Dr Shigetaka Hayano | The Rubber Revolution: Cracking the Code for Tire Recycling!
Traditionally, rubber waste was nearly impossible to recycle due to crosslinked sulphur bonds. But a team of researchers led by Dr Shigetaka Hayano from Zeon Corporation, in Japan, have achieved a groundbreaking feat in rubber recycling. Using mild conditions for the reaction, scientists have overcome the unfavourable cross-linked structure and have achieved recovery of rubber’s original monomers. This process restores cyclopentene monomers with 90% efficiency, allowing old tires and industrial rubber waste to be chemically recycled into high quality materials. If scaled up, this innovation could revolutionise waste management, reduce environmental pollution, and enable a circular economy for rubber production.